Prognostic impact of systolic blood pressure trajectory among patients hospitalised
in an acute heart failure setting: insights from a real-world multinational cohort
DOI: https://doi.org/https://doi.org/10.57187/4956
Marc Carminatia,
Tamila
Abdurashidovaa,
Nisha
Soboruna,
Guillaume Stauffachera,
Barbara
Pitta-Grosa,
Sara
Schukrafta,
Ioannis Skalidisb,
Georgios
Tzimasa,
Panagiotis
Antiochosa,
Pierre
Monneya,
Hicham
Skalic,
Roger
Hullina,
Henri Lua
a Division of Cardiology, Lausanne
University Hospital (CHUV), University of Lausanne (UNIL), Lausanne,
Switzerland
b Institut
Cardiovasculaire Paris-Sud, Hôpital Jacques Cartier, Ramsay-Santé, Massy,
France
c Division of Cardiovascular Medicine,
Brigham and Women’s Hospital, Harvard Medical School, Boston MA, USA
Summary
BACKGROUND: Systolic blood pressure is a prognostic
marker in acute heart failure, but the prognostic implications of in-hospital
changes in systolic blood pressure are unclear. We assessed the association
between in-hospital systolic blood pressure changes and outcomes in a
real-world, multinational cohort of acute heart failure patients.
METHODS: We analysed consecutive patients hospitalised
for acute heart failure between 2005 and 2020 at two tertiary-care centres
(CHUV, Switzerland; NCCIM, Kyrgyzstan) with available systolic blood pressure measurements
at admission and discharge. Patients were classified into four systolic blood
pressure trajectory categories: stable normal/low (systolic blood pressure consistently
<140 mm Hg or minor increase, Δ<10), increasing (systolic blood pressure rose
≥10 mm Hg from <140 to ≥140 mm Hg), decreasing (systolic blood pressure dropped
≥10 mm Hg from ≥140 to <140 mm Hg), stable elevated (systolic blood pressure
consistently ≥140 mm Hg or minor decrease, Δ<10). The primary outcome of the
study was a composite of first heart failure hospitalisation or all-cause
mortality, assessed over a 1-year follow-up period. The association between
categories and the primary outcome was assessed with Cox models, adjusted for
relevant covariates.
RESULTS: Among 1490 patients (80% Swiss, 56% male, age
75 ± 13 years), 621 experienced the primary outcome at 1 year. Compared to
those with stable normal/low systolic blood pressure, patients with decreasing systolic
blood pressure had a significantly lower risk of the primary outcome (adjusted
HR: 0.81; 95% CI: 0.66–0.99; p = 0.040), with no significant differences for
the other systolic blood pressure trajectories. Results remained consistent
regardless of sex, age and left
ventricular ejection fraction (Pinteraction for all
>0.05).
CONCLUSION: In this real-world, multinational cohort
of 1490 acute heart failure patients, in-hospital decline in systolic blood
pressure was independently associated with improved outcomes in those with an
elevated systolic blood pressure at admission.
Abbreviations
- ACE
-
angiotensin-converting enzyme
- AFF
-
atrial fibrillation or flutter
- ARB
-
angiotensin receptor blocker
- ARNi
-
angiotensin receptor/neprilysin inhibitor
- BMI
-
body
mass index
- COPD
-
chronic obstructive pulmonary disease
- DBP
-
diastolic blood pressure
- eGFR
-
estimated glomerular filtration rate
- HbA1C
-
haemoglobin
A1C
- HFH
-
heart
failure hospitalisation
- ICD
-
implantable cardioverter-defibrillator
- LVEDD
-
left ventricular end-diastolic diameter
- LVEF
-
left
ventricular ejection fraction
- LVMi
-
left
ventricular mass index
- MRA
-
mineralocorticoid receptor antagonist
- NT-proBNP
-
N-terminal
pro-B-type natriuretic peptide
- SBPa
-
systolic blood pressure at admission
- SBPd
-
systolic blood pressure at discharge
Introduction
Despite recent significant advancements in the
management of heart failure [1], hospitalisations for acute heart failure remain
frequent and impose a considerable burden on healthcare systems. Acute heart
failure represents the leading cause of unplanned hospital admissions among
individuals aged 65 years and older, contributing substantially to high healthcare
costs associated with these admissions [2–4].
Acute heart failure remains strongly associated with
poor outcomes, including very high short-term readmission rates and high
mortality. Every year, over one million patients are hospitalised with a primary
diagnosis of acute heart failure in North America and Western Europe [4, 5].
High systolic blood pressure is a key risk factor of heart
failure, both with reduced and preserved ejection fraction [6], and has emerged
as an important prognostic marker in the context of acute heart failure [7]. Previous
studies
have suggested that, among patients hospitalised for acute heart failure, higher
systolic blood pressure at the time of hospital admission may be linked to
better outcomes compared to lower systolic blood pressure values [7, 8]. This contrasts
with the ambulatory and chronic heart failure setting, where elevated systolic
blood pressure has traditionally been associated with a poorer prognosis [1, 9, 10].
However, the
prognostic significance of systolic blood pressure trajectory during hospitalisation
remains less understood. Gaining a clearer understanding of how systolic blood
pressure changes between hospital admission and discharge could help identify
patients at higher risk of adverse clinical outcomes, who may benefit from
closer monitoring.
Using data from a prospective, large, real-world, multiethnic
cohort including close to 1500 patients in Switzerland and Kyrgyzstan, we aimed
to examine the association between in-hospital systolic blood pressure changes
and subsequent clinical outcomes.
Methods
Study design and population
Consecutive adult patients hospitalised for acute
heart failure in the Cardiology and Internal Medicine Divisions of Lausanne
University Hospital (CHUV) in Lausanne, Switzerland, and the National Center of
Cardiology and Internal Medicine (NCCIM) in Bishkek, Kyrgyzstan, between 2005
and 2020, were included in the cohort. Both the CHUV and NCCIM are tertiary
teaching hospitals offering comprehensive heart failure care, including
ambulatory services, acute heart failure hospitalisation units, interventional
procedures and device implantation.
Patients had a clinical diagnosis of acute heart
failure according to heart failure guidelines in effect at the time of the
study [11]. Only those with systolic
blood pressure (SBP) measurements available at both admission (SBPa) and
discharge (SPBd) were included in the present study. Exclusion criteria
included patients who died during hospitalisation and those presenting with
acute pulmonary embolism, acute myocardial ischaemia, severe valvular
regurgitation or stenosis requiring percutaneous or surgical intervention, stress-induced
cardiomyopathy, exacerbation of chronic obstructive pulmonary disease (COPD), complex
congenital heart disease or recent cardiac surgery. Additional exclusions
applied to patients with acute infectious, toxic or metabolic conditions, defined
as acute non-cardiac conditions (such as acute intoxications or severe
metabolic derangements, including diabetic ketoacidosis) that were considered
the primary cause of haemodynamic instability or heart failure-like
presentation, those undergoing dialysis, pregnant women or individuals with an
estimated survival of less than one year due to comorbidities.
Echocardiography was performed or reviewed by
board-certified cardiologists following applicable international guidelines [12].
Clinical
variables, comorbidities, laboratory values, vital signs at admission and
discharge, treatments and echocardiographic parameters were extracted from the
electronic medical records of each centre using standardised case report forms.
Mortality data were obtained from national death registries, and heart failure
rehospitalisations were retrieved from each hospital’s electronic health
system. All data were then transferred into a centralised, deidentified
research database. Quality control included double-checking of 20% of entries, yielding
a 99.7% concordance rate [13]. Sodium-glucose
cotransporter-2 inhibition was not yet recommended for heart failure management
during the study period, and was therefore categorised as oral antidiabetic
treatment. Information on 1-year all-cause mortality was extracted from
national registries in Kyrgyzstan and Switzerland (accessed in December 2021).
Data on hospitalisation for heart failure were obtained from the electronic
medical records of each hospital.
This study was conducted in accordance with the
Declaration of Helsinki and received approval from the corresponding local
ethics committees (Bishkek: Local Ethics Committee 2019-7; Lausanne: CER-VD
2019-1158).
Systolic blood pressure measurement and categorisation
Blood pressure was measured using an automated device
or a standard sphygmomanometer with an appropriately sized cuff, in both centres,
at both admission and discharge. SBPa was defined as the first systolic blood
pressure measurement available in the medical record after the start of
hospitalisation, SBPd as the last systolic blood pressure measurement available
before the end of hospitalisation.
Patients were categorised into four systolic blood
pressure trajectory groups based on SBPa and SBPd values:
- Stable normal/low: both SBPa and SBPd <140 mm
Hg, or SBPa <140 mm Hg and SBPd ≥140 mm Hg, with a difference (Δ) <10 mm Hg.
- Increasing: SBPa <140 mm Hg and SBPd ≥140
mm Hg, with a Δ ≥10 mm Hg.
- Decreasing: SBPa ≥140 mm Hg and SBPd <140 mm
Hg, with a Δ ≥10 mm Hg.
- Stable elevated: both SBPa and SBPd ≥140 mm Hg,
or SBPa ≥140 mm Hg and SBPd <140 mm Hg, with a Δ <10 mm Hg.
The threshold of 140 mm Hg for defining “high” and “normal/low”
systolic blood pressure was chosen based on the standard definition of
hypertension, applicable in both chronic and acute settings [14, 15]. The selection
of
a Δ of 10 mm Hg reflects the rationale that this level of change is clinically
significant and meaningful in an acute setting.
Study outcomes
The primary outcome was a composite of first hospitalisation
for heart failure or all-cause death occurring within one year after the index hospitalisation
for heart failure. The secondary outcomes were the individual components of the
composite, namely first hospitalisation for heart failure alone and all-cause
death alone, assessed over the same follow-up period.
Statistical analyses
Data were reported as count (percentage) for
categorical variables, mean (standard deviation) when distributed normally and
median (interquartile range) for non-normally distributed variables. To compare
global differences across the four groups, we used the chi-squared test for
binary variables, analysis of variance for normally distributed continuous
variables and the Kruskal–Wallis test for non-normally distributed continuous
variables.
The association between systolic blood pressure trajectory
categories and outcomes (all analysed as time to first events) was assessed
using Cox proportional hazards models, with the stable normal/low category
(SBPa and SBPd <140 mm Hg, Δ <10 mm Hg) serving as the reference. The
follow-up period began at the date of discharge. Analyses were stratified by
country (Kyrgyzstan or Switzerland), and were both unadjusted and adjusted for age,
sex, body mass index (BMI), prior
myocardial infarction, atrial fibrillation or flutter, prior hospitalisation
for heart failure, hypertension, diabetes, left ventricular ejection fraction
(LVEF), estimated glomerular filtration rate, length of hospital stay, use of
beta-blockers, angiotensin-converting enzyme (ACE) inhibitors, angiotensin
receptor blockers (ARB) and mineralocorticoid receptor antagonists (MRA).The selection
of these
covariates was guided by baseline differences between categories and clinical
judgement. Interaction testing was conducted to evaluate the interaction
between systolic blood pressure trajectory categories and sex, age groups
(<78 years, ≥78 years, based on the median age in the whole cohort) and LVEF
(<40%, ≥40%) in relation to the primary outcome. We also assessed the
relative change in the use of heart failure prognostic medication classes
(beta-blockers, ACE inhibitors, angiotensin receptor blockers, mineralocorticoid
receptor antagonists, angiotensin receptor/neprilysin inhibitor), as well as
loop diuretics, between admission and discharge according to systolic blood
pressure trajectory categories. The relative change for each medication class
was calculated as: (proportion at discharge – proportion at admission) /
proportion at admission. In
sensitivity analyses, we further explored the prognostic impact of SBPa and
SBPd individually, categorised as follows: <120 mm Hg, 120–139 mm Hg, 140–159
mm Hg and ≥160 mm Hg. Crude
incidence rates of events were reported using the Kaplan–Meier method. We tested for
violation of the proportional
hazards assumption for all hazard ratios that were reported, via Schoenfeld
residuals.
Additionally, we analysed the relationship between the primary outcome and continuous
SBP change (defined as SBPd − SBPa), using Poisson regression to
estimate incidence rates, and applying multivariable restricted cubic splines, with
the numbers of knots selected to minimise the resulting Akaike information
criteria (3–6 knots tested).
Finally, a
multivariable logistic regression model with a forward stepwise approach was used
to identify clinical, biological and echocardiographic parameters associated
with the SBP trajectory category with the most favourable prognosis, including
all relevant parameters at baseline in the model. To ensure comparability, continuous
variables were standardised. Statistical analyses were performed using STATA, version
18 (StataCorp, College Station, TX, USA). A two-sided p-value <0.05
was considered statistically significant.
Results
Demographics and baseline characteristics
A total of 1746 patients were recruited, including
1208 (69.2%) in Switzerland. Among these, 61 patients (3.5%) died during the
index hospitalisation and were excluded from the analyses. Among the remaining
1685 patients, systolic blood pressure measurements were available at both
admission and discharge in 1490 patients (80% from Switzerland, 56% male, mean
age: 75 ± 13 years). Of these, 757 (50.8%) had stable/low systolic blood
pressure, 77 (5.2%) had increasing systolic blood pressure, 412 (27.7%) had
decreasing systolic blood pressure and 244 (16.3%) had stable elevated systolic
blood pressure (figure 1).
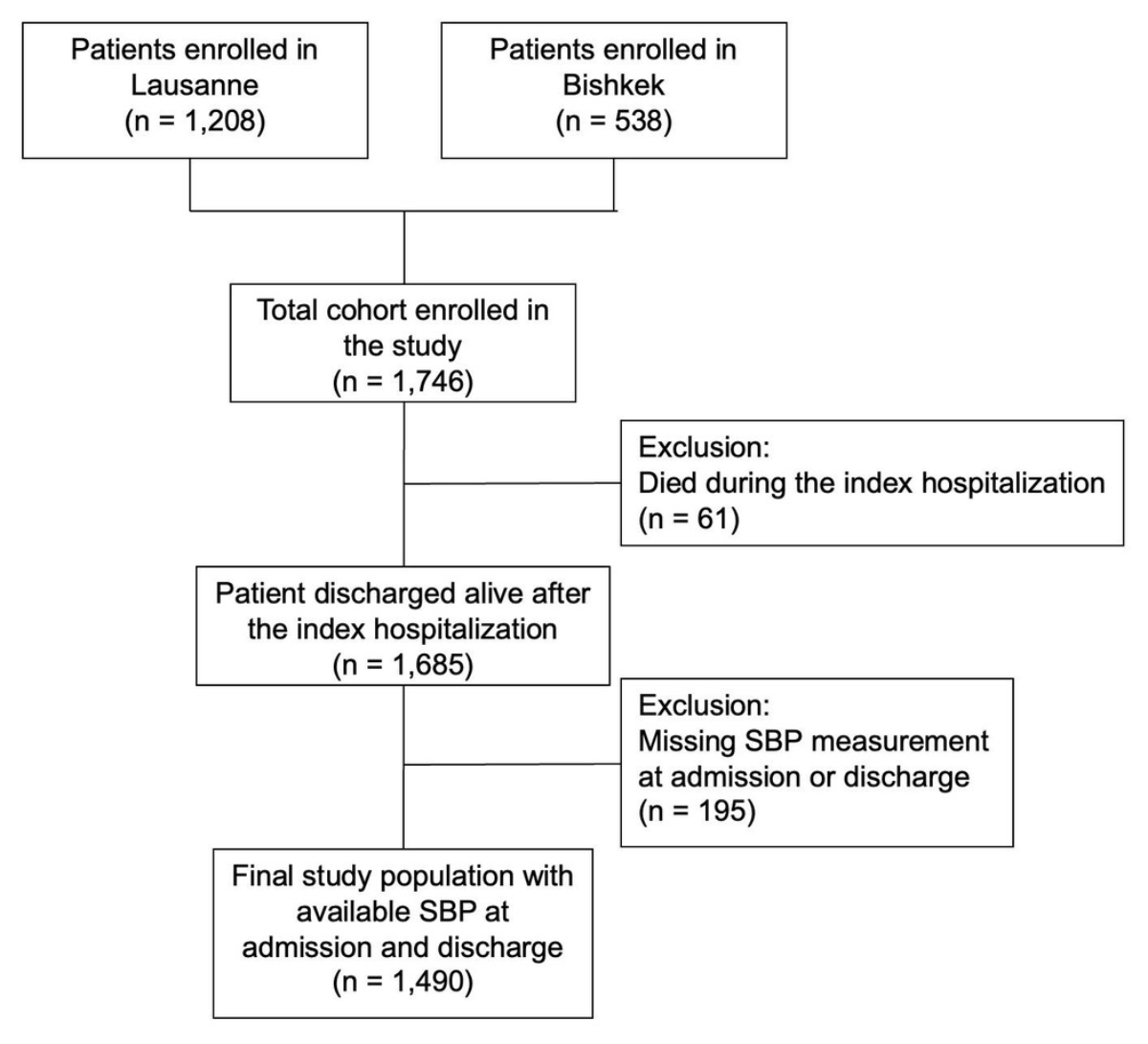
Figure 1Patient
selection flowchart. The flowchart illustrates the derivation of the final
study population from patients hospitalised for acute heart failure at the two
participating centres. “Patients enrolled” refers to patients meeting the
clinical diagnosis of acute heart failure and included in the study databases
at each centre. The flowchart details exclusions due to in-hospital death and
missing systolic blood pressure measurements at admission and/or discharge,
leading to the final analysis cohort.
The increasing systolic blood pressure category
included more women (57.1%) and older patients (82.5 years) compared to other
groups (p <0.001, table 1). Patients in this category also had a significantly
higher prevalence of atrial fibrillation or flutter, lower glomerular
filtration rates and lower haemoglobin levels. Patients with stable elevated systolic
blood pressure were less likely to have a previous episode of hospitalisation
for heart failure, but had a higher prevalence of hypertension and diabetes. N-terminal
pro–B-type natriuretic peptide (NT-proBNP, only available in 55.8% of patients)
was significantly higher in the stable normal/low systolic blood pressure
group. No significant differences were observed across systolic blood pressure
trajectory groups regarding BMI, prior myocardial infarction, COPD or tobacco
use. In terms of echocardiographic parameters, patients with stable normal/low systolic
blood pressure had significantly lower LVEF and larger LV end-diastolic
diameters. Regarding treatments at admission, patients in the stable elevated systolic
blood pressure group were more frequently prescribed beta-blockers, angiotensin
receptor blockers, and insulin, while those in the stable normal/low systolic
blood pressure group more often received mineralocorticoid receptor antagonists and
loop diuretics.
Table 1Baseline
patient characteristics by systolic blood pressure (SBP) category. Results are
expressed as n (%), mean ± standard deviation or median [interquartile range]. Groups
were compared using the chi-squared test for categorical variables, analysis
of variance (ANOVA) for normally distributed continuous variables and the
Kruskal–Wallis test for non-normally distributed continuous variables.
| Characteristics |
Stable normal/low SBP (n = 757) |
Increasing SBP (n = 77) |
Decreasing SBP (n = 412) |
Stable elevated SBP (n = 244) |
p-value |
| Demographics and vitals |
Age, years |
73.1 ±
14.3 |
82.5 ±
8.7 |
76.3 ±
12.4 |
78.5 ±
10.1 |
<0.001 |
| Female sex |
301 (39.8%) |
44 (57.1%) |
183 (44.4%) |
128 (52.5%) |
<0.001 |
| BMI, kg/m2 |
27.4 ± 6.6 |
27.1 ± 6.5 |
28.1 ± 6.2 |
27.1 ± 5.7 |
0.23 |
| Bishkek cohort |
188 (24.8%) |
1 (1.3%) |
92 (22.3%) |
18 (7.4%) |
<0.001 |
| SBPa, mm Hg |
117.0 ±
14.2 |
120.6 ±
11.6 |
159.8 ±
17.3 |
162.4 ±
21.5 |
By design |
| DBPa, mm Hg |
71.8 ±
13.0 |
70.6 ±
15.0 |
90.7 ±
17.4 |
85.3 ±
16.9 |
<0.001 |
| Heart rate, bpm |
93.4 ±
25.6 |
93.8 ±
26.3 |
95.1 ±
28.0 |
86.8 ±
23.7 |
<0.001 |
| Comorbidities |
Previous myocardial infarction |
331 (43.7%) |
32 (41.6%) |
181 (43.9%) |
112 (45.9%) |
0.90 |
| AFF |
444 (58.7%) |
47 (61.0%) |
224 (54.4%) |
115 (47.1%) |
0.011 |
| Prior heart failure hospitalisation |
583 (77.0%) |
52 (67.5%) |
290 (70.4%) |
155 (63.5%) |
<0.001 |
| Hypertension |
541 (71.5%) |
70 (90.9%) |
352 (85.4%) |
230 (94.3%) |
<0.001 |
| Diabetes |
226 (29.9%) |
29 (37.7%) |
146 (35.4%) |
106 (43.4%) |
<0.001 |
| COPD |
161 (21.3%) |
14 (18.2%) |
97 (23.5%) |
49 (20.1%) |
0.61 |
| Smoking |
334 (44.1%) |
33 (42.9%) |
174 (42.2%) |
112 (46.1%) |
0.80 |
| Dyslipidaemia |
386 (51.0%) |
37 (48.1%) |
234 (56.8%) |
145 (59.7%) |
0.040 |
| Haemoglobin, g/L |
128.4 ±
22.9 |
117.0 ±
21.9 |
129.9 ±
22.7 |
121.6 ±
21.7 |
<0.001 |
| HbA1C, % |
7.0 ±
1.5 |
7.1 ±
0.7 |
6.8 ±
1.4 |
6.9 ±
1.4 |
0.94 |
| NT-proBNP, pg/ml* |
5543 [2361 – 11,060] |
3586 [2005 – 9534] |
4402 [2377 – 8886] |
3530 [1785 – 9910] |
0.048 |
| Creatinine, µmol/l |
124.5 ±
63.4 |
130.2 ±
61.6 |
123.5 ±
88.1 |
143.2 ±
109.8 |
0.009 |
| eGFR, ml/min/1.73m2 |
56.9 ±
22.7 |
48.2 ±
20.4 |
57.0 ±
21.6 |
52.1 ±
25.1 |
<0.001 |
| Echocardiographic parameters |
LVEF, % |
41.0 ±
16.3 |
51.2 ±
13.6 |
45.2 ±
15.3 |
51.5 ±
14.0 |
<0.001 |
| LVEDD, mm |
56.2 ±
11.1 |
49.8 ±
9.6 |
54.0 ±
10.0 |
50.7 ±
7.4 |
<0.001 |
| LVMi, g/m2 |
119.1 ±
43.9 |
111.2 ±
33.7 |
123.2 ±
42.0 |
113.5 ±
32.0 |
0.031 |
| Treatment |
Statin |
264 (34.9%) |
30 (39.0%) |
135 (32.8%) |
98 (40.2%) |
0.24 |
| Beta-blocker |
351 (46.4%) |
42 (54.5%) |
187 (45.4%) |
139 (57.0%) |
0.011 |
| ACE inhibitor |
224 (29.6%) |
25 (32.5%) |
129 (31.3%) |
86 (35.2%) |
0.42 |
| ARB |
135 (17.8%) |
23 (29.9%) |
105 (25.5%) |
67 (27.5%) |
<0.001 |
| ARNi |
3 (0.4%) |
0 (0.0%) |
3 (0.7%) |
0 (0.0%) |
0.50 |
| MRA |
169 (22.3%) |
5 (6.5%) |
53 (12.9%) |
24 (9.8%) |
<0.001 |
| Loop diuretic |
433 (57.2%) |
43 (55.8%) |
200 (48.5%) |
124 (50.8%) |
0.028 |
| Oral antidiabetic |
125 (16.5%) |
16 (20.8%) |
56 (13.6%) |
48 (19.7%) |
0.15 |
| Insulin therapy |
76 (10.0%) |
13 (16.9%) |
64 (15.5%) |
51 (20.9%) |
<0.001 |
| ICD |
38 (5.0%) |
1 (1.3%) |
7 (1.7%) |
4 (1.6%) |
0.004 |
| Pacemaker |
81 (10.7%) |
8 (10.4%) |
32 (7.8%) |
21 (8.6%) |
0.39 |
Association between systolic blood pressure trajectory categories and outcomes
Over a 1-year follow-up period, 621 patients experienced
a primary outcome event (table 2). Using the stable normal/low systolic blood
pressure category as the reference (64.0 events per 100 patient-years [py]), the
decreasing systolic blood pressure category was associated with a significantly
lower risk for the primary outcome, both in non-adjusted (49.4 events per 100
py, HR: 0.78, 95% CI: 0.64–0.94; figure 2A) and adjusted (adjusted HR: 0.81, 95%
CI: 0.66–0.99) analyses. No significant differences were observed for the other
systolic blood pressure trajectory categories. These findings were consistent
regardless of sex (Pinteraction = 0.91), age category (<78 and
≥78 years, Pinteraction = 0.09) and LVEF (<40% and ≥40%, Pinteraction
= 0.22).
Table 2Event counts, event rates and hazard ratios associated with categories of systolic
blood pressure trajectory. All analyses are stratified by country (Switzerland or
Kyrgyzstan). Comparisons
across systolic blood pressure trajectory groups were performed using Cox
proportional hazards regression.
| Events |
n (%) |
Event rate (95% CI), per 100 patient-years |
Unadjusted HR (95% CI) |
Adjusted HR (95% CI), Model* |
| First heart failure hospitalisation or all-cause death |
Stable normal/low SBP (n = 757) |
325 (42.9%) |
64.0 (57.5–71.4) |
1 (reference) |
1 (reference) |
| Increasing SBP (n = 77) |
36 (46.8%) |
76.5 (55.2–106.0) |
1.09 (0.77–1.54) |
1.00 (0.70–1.43) |
| Decreasing SBP (n = 412) |
150 (36.4%) |
49.4 (42.1–58.0) |
0.78 (0.64–0.94) |
0.81 (0.66–0.99) |
| Stable elevated SBP (n = 244) |
110 (45.1%) |
65.5 (54.3–79.0) |
0.97 (0.78–1.20) |
0.96 (0.76–1.21) |
| First heart failure hospitalisation |
Stable normal/low SBP (n = 757) |
232 (30.6%) |
38.1 (33.5–43.3) |
1 (reference) |
1 (reference) |
| Increasing SBP (n = 77) |
26 (33.8%) |
45.1 (30.7–66.2) |
1.11 (0.73–1.66) |
1.03 (0.68–1.56) |
| Decreasing SBP (n = 412) |
115 (27.9%) |
34.1 (28.4–41.0) |
0.90 (0.72–1.13) |
0.92 (0.73–1.16) |
| Stable elevated SBP (n = 244) |
83 (34.0%) |
44.5 (35.8–59.1) |
1.12 (0.87–1.44) |
1.07 (0.82–1.40) |
| All-cause death |
Stable normal/low SBP (n = 757) |
184 (24.3%) |
30.4 (26.3–35.1) |
1 (reference) |
1 (reference) |
| Increasing SBP (n = 77) |
18 (23.4%) |
29.2 (18.4–46.3) |
0.84 (0.52–1.37) |
0.78 (0.47–1.28) |
| Decreasing SBP (n = 412) |
63 (15.3%) |
17.3 (13.5–22.1) |
0.56 (0.42–0.75) |
0.58 (0.43–0.78) |
| Stable elevated SBP (n = 244) |
60 (24.6%) |
29.0 (22.5–37.4) |
0.86 (0.64–1.15) |
0.94 (0.69–1.28) |
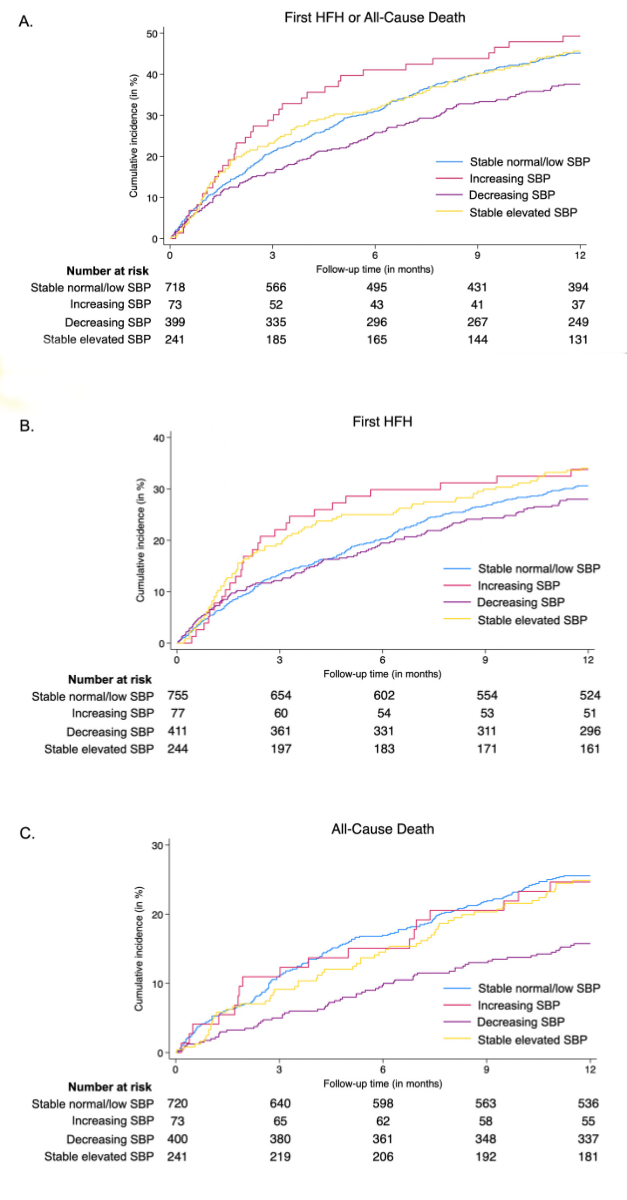
Figure 2Cumulative incidence of (A) the composite outcome of first heart failure
hospitalisation or all-cause death, (B) first heart failure hospitalisation,
(C) all-cause death according to categories of systolic blood pressure
trajectory. Cumulative incidence curves were generated using the Kaplan–Meier
method. Abbreviations: HFH: heart failure hospitalisation; SBP: systolic blood
pressure.
When analysing all-cause mortality and first hospitalisation
for heart failure as separate outcomes, the decreasing systolic blood pressure
category was associated with a significant reduction in the risk of all-cause
mortality (adjusted HR: 0.58, 95% CI: 0.43–0.78; figure 2B), but there was no
significant effect on the risk of first hospitalisation for heart failure
(adjusted HR: 0.92, 95% CI: 0.73–1.16; figure 2C). No violations of the
proportional hazards assumption for systolic blood pressure categories were
identified for either the primary or secondary outcomes.
No
significant association was observed between SBPa categories and the risk of
the primary outcome (table S1 in the appendix). In contrast, patients in the
lower SBPd category (SBPd <120 mm Hg) had a significantly higher risk of the
primary outcome (adjusted HR: 1.27, 95% CI: 1.05–1.53) compared to those with
an SBPd of 120–139 mm Hg (table S2 in the appendix).
When
analysing systolic blood pressure changes between admission and discharge as a
continuous variable, a decline in systolic blood pressure was associated with a
lower rate of the primary outcome among patients with an SBPa ≥140 mm Hg (figure
3A). However, no significant association was observed between systolic blood
pressure changes and event rates in patients with an SBPa <140 mm Hg (figure
3B).
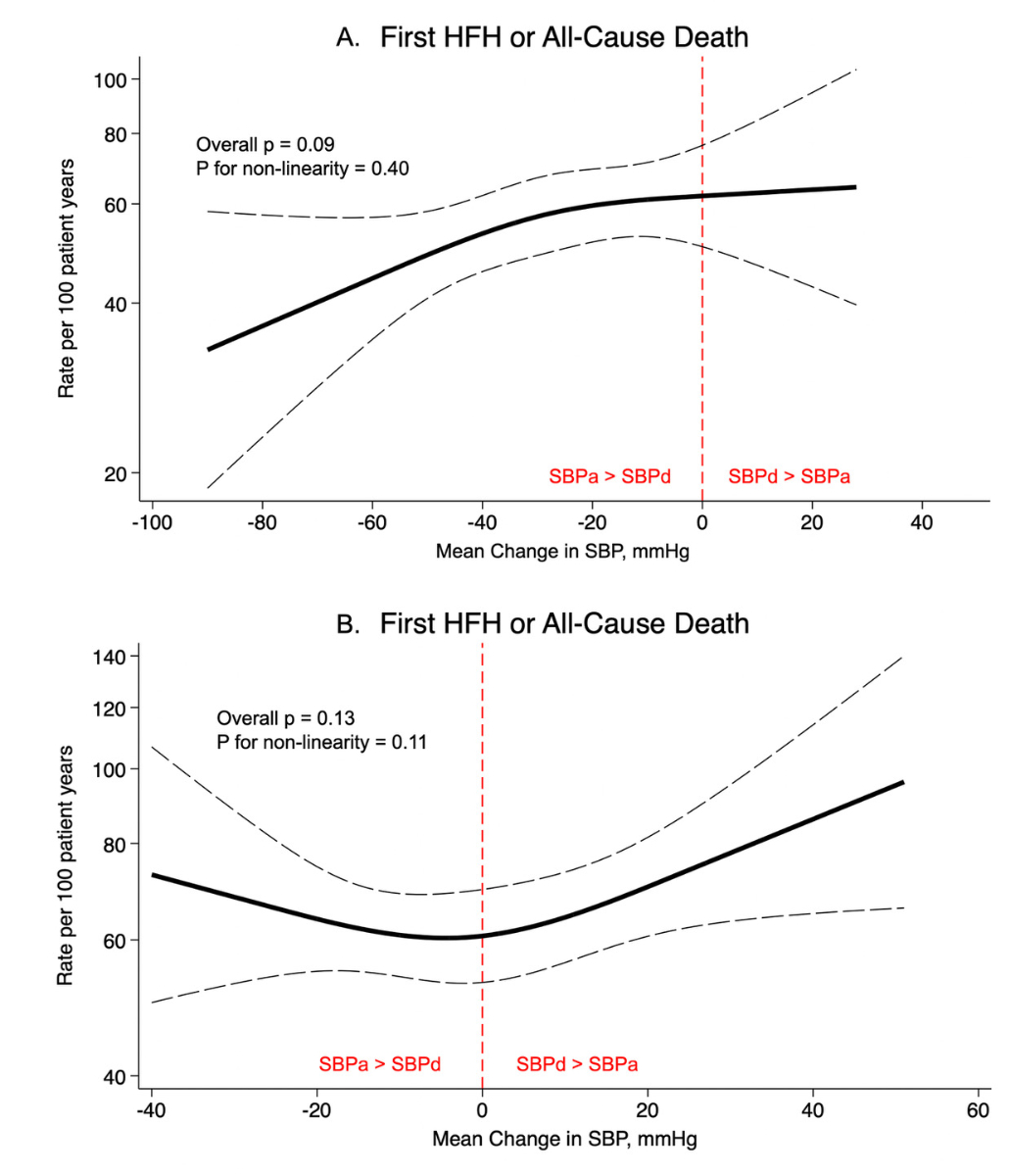
Figure 3Rate
of first heart failure hospitalisation or all-cause death according to mean change
in systolic blood
pressure (SBP at discharge – SBP at admission) (A) among patients with a
systolic blood pressure ≥140 mm Hg at admission, (B) among patients with a
systolic blood pressure <140 mm Hg at admission. Event rates were estimated
using Poisson regression models. The association between continuous systolic
blood pressure change and the primary outcome was assessed using multivariable
restricted cubic splines. Abbreviations: HFH: heart failure hospitalisation;
SBP: systolic blood pressure; SBPa: systolic blood pressure at admission; SBPd:
systolic blood pressure at discharge.
Relative changes in the use of prognostic heart failure medication classes
and diuretics
Relative changes in the use of beta-blockers (+58.8%),
ACE inhibitors (+52.7%), angiotensin receptor blockers (+4.7%), mineralocorticoid
receptor antagonists (+112.5%) and loop diuretics (+76.1%) were greatest in the
decreasing systolic blood pressure category compared to other systolic blood
pressure trajectory categories (table 3). At discharge, the proportion of
patients on these medications in the decreasing systolic blood pressure
category was the highest or second highest among the four categories.
Table 3Number, proportion and relative changes in heart failure prognostic medications
by systolic blood pressure trajectory categories. Results are expressed as n
(%) unless otherwise specified. The relative change for each medication class
was calculated as: (proportion at discharge – proportion at admission) /
proportion at admission.
| Medication |
Stable
normal/low SBP (n = 757) |
Increasing
SBP (n = 77) |
Decreasing
SBP (n = 412) |
Stable
elevated SBP (n = 244) |
p-value |
| Beta-blocker |
At
admission |
351 (46.4%) |
42 (54.5%) |
187 (45.4%) |
139 (57.0%) |
0.011 |
| At
discharge |
510 (67.4%) |
42 (54.5%) |
297 (72.1%) |
163 (66.8%) |
0.020 |
| Relative
change (in %) |
+45.3% |
0.0% |
+58.8% |
+17.2% |
<0.001 |
| ACE inhibitor |
At
admission |
224 (29.6%) |
25 (32.5%) |
129 (31.3%) |
86 (35.2%) |
0.42 |
| At discharge |
308 (40.7%) |
32 (41.6%) |
197 (47.8%) |
121 (49.6%) |
0.028 |
| Relative
change (in %) |
+37.5% |
+28.0% |
+52.7% |
+40.9% |
<0.001 |
| Angiotensin receptor blocker |
At
admission |
135 (17.8%) |
23 (29.9%) |
105 (25.5%) |
67 (27.5%) |
<0.001 |
| At
discharge |
107 (14.1%) |
16 (20.8%) |
110 (26.7%) |
66 (27.0%) |
<0.001 |
| Relative
change (in %) |
-20.8% |
-30.4% |
+4.7% |
-1.8% |
<0.001 |
| Angiotensin receptor/neprilysin inhibitor |
At
admission |
3 (0.4%) |
0 (0.0%) |
3 (0.7%) |
0 (0.0%) |
0.50 |
| At
discharge |
5 (0.7%) |
0 (0.0%) |
2 (0.5%) |
0 (0.0%) |
0.55 |
| Relative
change (in %) |
+75.0% |
0.0% |
-28.6% |
0.0% |
<0.001 |
| Mineralocorticoid
receptor antagonist |
At
admission |
169 (22.3%) |
5 (6.5%) |
53 (12.9%) |
24 (9.8%) |
<0.001 |
| At
discharge |
334 (44.1%) |
7 (9.1%) |
113 (27.4%) |
37 (15.2%) |
<0.001 |
| Relative
change (in %) |
+97.8% |
+40.0% |
+112.5% |
+55.1% |
<0.001 |
| Loop
diuretic |
At
admission |
433 (57.2%) |
43 (55.8%) |
200 (48.5%) |
124 (50.8%) |
0.028 |
| At
discharge |
664 (88.2%) |
60 (78.9%) |
352 (85.4%) |
203 (83.2%) |
0.049 |
| Relative
change (in %) |
+54.2% |
+41.4% |
+76.1% |
+63.8% |
<0.001 |
Factors associated with high systolic blood pressure at admission and subsequent
decline during hospitalisation
Factors at baseline associated with the decreasing systolic
blood pressure category are presented in table 4. They include a known
comorbidity of hypertension at admission (OR: 1.61, 95% CI: 1.64–2.24), higher
BMI (OR: 1.15 per 6.4 kg/m2 change, 95% CI: 1.02–1.30), higher heart
rate at admission (OR: 1.19 per 26.1 bpm, 95% CI: 1.06–1.34) and higher eGFR at
admission (1.15 per 22.8 ml/min/1.73 m2, 95% CI: 1.01–1.30).
Table 4Factors associated with high systolic blood pressure at admission and subsequent
decline during hospitalisation. Sample size: n = 1490. There were no missing
data in any category. Odds ratios and 95% confidence intervals were obtained
using a multivariable logistic regression model with forward stepwise
selection.
| Factor |
Odds ratio |
95% CI |
p-value |
| Known hypertension |
1.61 |
1.16–2.24 |
0.005 |
| BMI, per
6.4 kg/m2 (1 SD) |
1.15 |
1.02–1.30 |
0.019 |
| Heart
rate, per 26.1 bpm (1 SD) |
1.19 |
1.06–1.34 |
0.003 |
| eGFR, per
22.8 ml/min/m2 (1 SD) |
1.15 |
1.01–1.30 |
0.034 |
Discussion
In this analysis of nearly 1500
patients hospitalised for acute heart failure, we found that: (1) In-hospital decline
in systolic blood pressure was
associated with improved outcomes in those with elevated SBPa. This result was primarily
driven by a lower risk of
all-cause mortality and was consistent irrespective of sex, LVEF and age. (2) No
significant association was found between SBPa and the risk of the primary
outcome, while lower SBPd was associated with a higher risk. (3) Relative
changes in the use of heart failure prognostic medication classes and loop
diuretics were greatest in the decreasing systolic blood pressure category
compared to other systolic blood pressure trajectory categories. (4) Factors associated
with the decreasing systolic blood
pressure category included known hypertension, higher BMI, higher heart rate
and higher eGFR at admission.
To the best of our knowledge, while
many previous studies have examined the prognostic impact of systolic blood
pressure in an acute heart failure setting [7, 8, 16], our study is the first to comprehensively
assess the
prognostic significance of the systolic blood pressure trajectory during
hospitalisation. Despite being an observational analysis, the study is
strengthened by the inclusion of a large, real-world, well-characterised, multinational
cohort of acute heart failure patients and the detailed reporting of patient
vital signs at both admission and discharge.
High systolic blood pressure
in heart failure serves both as a marker and a mediator of cardiovascular risk.
It is associated with increased afterload, arterial stiffness and impaired
ventricular-vascular coupling, which exacerbate myocardial stress and adverse remodelling
[6, 17, 18]. Among chronic heart failure patients with
hypertension, lowering systolic blood pressure to a target range of 120–130 mm
Hg may provide long-term benefits [1, 9]. In the acute setting, however, systolic
blood
pressure may have a different prognostic significance. Higher systolic blood
pressure in acute heart failure has previously been shown to be associated with
more favourable cardiovascular outcomes, possibly reflecting preserved cardiac
output, a less advanced heart failure state and more stable haemodynamics [6]. These
patients may be less prone to end-organ
hypoperfusion or cardiogenic shock, and more likely to tolerate therapies like
vasodilators or diuretics [2]. Furthermore, hospitalisations in this group may be
more often due to hypertensive pulmonary oedema rather than chronic heart
failure with systemic and neurohormonal activation [1]. High systolic blood pressure
in acute heart failure
may also facilitate the in-hospital initiation or up-titration of prognostic heart
failure treatments, which has been shown to be associated with a better
prognosis [19, 20].
Interestingly, systolic blood
pressure at admission did not demonstrate any predictive value in our analyses. Patients
with high systolic blood pressure at admission who achieved lower systolic
blood pressure values during hospitalisation experienced better outcomes
compared to those who remained hypertensive at discharge. This may be partly
driven by the fact that a higher proportion of patients in the decreasing systolic
blood pressure trajectory received heart failure prognostic medications and
loop diuretics during hospitalisation, compared to other systolic blood
pressure trajectory categories. It is well established that patients who
respond to initiation or up-titration of heart failure therapy and achieve
effective decongestion have improved prognoses in an acute heart failure
setting [21]. Moreover, our results suggest that the trajectory of
systolic blood pressure during hospitalisation appears to be a more reliable
prognostic indicator than a single systolic blood pressure measurement at
admission. The reason why the improved outcomes associated with decreasing systolic
blood pressure were primarily driven by all-cause death (and not hospitalisation
for heart failure) in our cohort remains uncertain and merits further
investigation.
In contrast, low systolic
blood pressure may signal more severe heart failure or a decompensated state, characterised
by reduced stroke volume, lower cardiac output and peripheral organ dysfunction,
both in a chronic and acute heart failure setting [22]. Consistent with this hypothesis,
our findings show
that better kidney function at admission (higher eGFR) was associated with a
higher probability of being in the decreasing systolic blood pressure category.
Additionally, higher BMI was also associated with this group, aligning with the
“obesity paradox”, where obesity is postulated to confer a better prognosis in heart
failure patients [23], although this concept is debated [24]. Notably, the better
prognosis associated with the
decreasing systolic blood pressure category remained robust after thorough
adjustment (including eGFR and BMI) and was consistent across sex and heart
failure subtypes (heart failure with reduced or preserved EF).
From a practical standpoint, our
findings underscore the importance of paying close attention to systolic blood
pressure trajectories in patients hospitalised for acute heart failure.
Persistently low or high systolic blood pressure may reflect more severe or
advanced heart failure, persistent congestion or inadequate response to
therapies, highlighting the need for closer monitoring and tailored management
strategies. Further research is warranted to assess the
effectiveness of incorporating in-hospital systolic blood pressure trajectories
into clinical decision-making and its potential impact on patient outcomes.
Limitations
Our study has several
potential limitations. First, as an observational study, causality cannot be
established, and our findings should be interpreted as hypothesis-generating.
Although we adjusted for multiple covariates, residual bias not accounted for
in the analysis may still be present. Second, BP measurements were based on
single readings, which are subject to variability; however, the reported values
reflect standard clinical practice in hospitalised settings, where single
measurements are often used. Third, only systolic blood pressure values at
admission and discharge were available, limiting the ability to perform a more
detailed longitudinal analysis of systolic blood pressure trajectories. Fourth,
body weight at discharge was not systematically available, limiting the ability
to assess decongestion using weight change as a surrogate during hospitalisation.
Finally, the study spanned the period from 2005
to 2020 and included two centres from different countries, introducing the
possibility of temporal and spatial heterogeneity in patient management and
treatment practices.
Conclusions
In a real-world, multinational cohort of 1490 acute
heart failure patients, a decline in systolic blood pressure during hospitalisation
was independently associated with improved outcomes in those with elevated systolic
blood pressure at admission, underscoring the potential prognostic significance
of systolic blood pressure trajectories during the acute management of acute
heart failure. This observation may, in part, be driven by the higher rates of
initiation of heart failure prognostic medications in this group during
hospitalisation. Further research is needed to explore the underlying
pathophysiological mechanisms behind these findings and their practical
implications.
Data sharing statement
Deidentified data may be made available from the
corresponding author upon reasonable request.
Henri Lu,
MD
Heart
Failure and Transplant Unit
Division of
Cardiology
Lausanne
University Hospital
Rue du Bugnon 46
CH-1011 Lausanne
henri.lu[at]chuv.ch
References
1. McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al.; ESC Scientific
Document Group. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic
heart failure. Eur Heart J. 2021 Sep;42(36):3599–726. 10.1002/ejhf.2333 doi: https://doi.org/10.1093/eurheartj/ehab368
2. Arrigo M, Jessup M, Mullens W, Reza N, Shah AM, Sliwa K, et al. Acute heart failure.
Nat Rev Dis Primers. 2020 Mar;6(1):16. 10.1038/s41572-020-0151-7
3. Deniau B, Costanzo MR, Sliwa K, Asakage A, Mullens W, Mebazaa A. Acute heart failure:
current pharmacological treatment and perspectives. Eur Heart J. 2023 Nov;44(44):4634–49.
10.1093/eurheartj/ehad233 doi: https://doi.org/10.1093/eurheartj/ehad617
4. Crespo-Leiro MG, Anker SD, Maggioni AP, Coats AJ, Filippatos G, Ruschitzka F, et al.;
Heart Failure Association (HFA) of the European Society of Cardiology (ESC). European
Society of Cardiology Heart Failure Long-Term Registry (ESC-HF-LT): 1-year follow-up
outcomes and differences across regions. Eur J Heart Fail. 2016 Jun;18(6):613–25.
10.1002/ejhf.772 doi: https://doi.org/10.1002/ejhf.566
5. Ambrosy AP, Fonarow GC, Butler J, Chioncel O, Greene SJ, Vaduganathan M, et al. The
global health and economic burden of hospitalizations for heart failure: lessons learned
from hospitalized heart failure registries. J Am Coll Cardiol. 2014 Apr;63(12):1123–33.
10.1016/j.jacc.2013.11.053
6. Messerli FH, Rimoldi SF, Bangalore S. The Transition From Hypertension to Heart Failure:
contemporary Update. JACC Heart Fail. 2017 Aug;5(8):543–51. 10.1016/j.jchf.2017.03.004 doi: https://doi.org/10.1016/j.jchf.2017.04.012
7. Grand J, Miger K, Sajadieh A, Køber L, Torp-Pedersen C, Ertl G, et al. Systolic Blood
Pressure and Outcome in Patients Admitted With Acute Heart Failure: An Analysis of
Individual Patient Data From 4 Randomized Clinical Trials. J Am Heart Assoc. 2021 Sep;10(18):e022288.
10.1002/ehf2.13734 doi: https://doi.org/10.1161/JAHA.121.022288
8. Gheorghiade M, Abraham WT, Albert NM, Greenberg BH, O’Connor CM, She L, et al.; OPTIMIZE-HF
Investigators and Coordinators. Systolic blood pressure at admission, clinical characteristics,
and outcomes in patients hospitalized with acute heart failure. JAMA. 2006 Nov;296(18):2217–26.
10.1001/jama.296.18.2217
9. Lu H, Kondo T, Claggett BL, et al. Systolic Blood Pressure and Pulse Pressure in Heart
Failure: Pooled Participant-Level Analysis of 4 Trials. J Am Coll Cardiol 2024:S0735-109710420-2.
doi:10.1016/j.jacc.2024.04.020
10. Arundel C, Lam PH, Gill GS, Patel S, Panjrath G, Faselis C, et al. Systolic Blood
Pressure and Outcomes in Patients With Heart Failure With Reduced Ejection Fraction.
J Am Coll Cardiol. 2019 Jun;73(24):3054–63. 10.1016/j.jacc.2019.02.026 doi: https://doi.org/10.1016/j.jacc.2019.04.022
11. Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JG, Coats AJ, et al.; ESC Scientific
Document Group. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic
heart failure: the Task Force for the diagnosis and treatment of acute and chronic
heart failure of the European Society of Cardiology (ESC)Developed with the special
contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016 Jul;37(27):2129–200.
10.1093/eurheartj/ehw128
12. Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations
for cardiac chamber quantification by echocardiography in adults: an update from the
American Society of Echocardiography and the European Association of Cardiovascular
Imaging. J Am Soc Echocardiogr. 2015 Jan;28(1):1–39.e14. 10.1016/j.echo.2015.10.029 doi: https://doi.org/10.1016/j.echo.2014.10.003
13. Abdurashidova T, Müller M, Schukraft S, Soborun N, Pitta-Gros B, Kikoïne J, et al. European
Society of Cardiology guidelines and 1 year outcomes of acute heart failure treatment
in Central Asia and Europe. ESC Heart Fail. 2024 Feb;11(1):483–91. 10.1002/ehf2.13904 doi: https://doi.org/10.1002/ehf2.14591
14. McEvoy JW, McCarthy CP, Bruno RM, Brouwers S, Canavan MD, Ceconi C, et al.; ESC Scientific
Document Group. 2024 ESC Guidelines for the management of elevated blood pressure
and hypertension. Eur Heart J. 2024 Oct;45(38):3912–4018. 10.1093/eurheartj/ehad123 doi: https://doi.org/10.1093/eurheartj/ehae178
15. Collins SP, Levy PD, Martindale JL, Dunlap ME, Storrow AB, Pang PS, et al. Clinical
and Research Considerations for Patients With Hypertensive Acute Heart Failure: A
Consensus Statement from the Society of Academic Emergency Medicine and the Heart
Failure Society of America Acute Heart Failure Working Group. J Card Fail. 2016 Aug;22(8):618–27.
10.1016/j.cardfail.2016.04.008 doi: https://doi.org/10.1016/j.cardfail.2016.04.015
16. Vidán MT, Bueno H, Wang Y, Schreiner G, Ross JS, Chen J, et al. The relationship between
systolic blood pressure on admission and mortality in older patients with heart failure.
Eur J Heart Fail. 2010 Feb;12(2):148–55. 10.1002/ejhf.76 doi: https://doi.org/10.1093/eurjhf/hfp195
17. Borlaug BA, Kass DA. Ventricular-vascular interaction in heart failure. Heart Fail
Clin. 2008 Jan;4(1):23–36. 10.1016/j.hfc.2008.01.003 doi: https://doi.org/10.1016/j.hfc.2007.10.001
18. Mitchell GF. Arterial stiffness and hypertension: chicken or egg? Hypertension. 2014 Aug;64(2):210–4.
10.1161/HYPERTENSIONAHA.114.03350 doi: https://doi.org/10.1161/HYPERTENSIONAHA.114.03449
19. Njoroge JN, Teerlink JR. Pathophysiology and Therapeutic Approaches to Acute Decompensated
Heart Failure. Circ Res. 2021 May;128(10):1468–86. 10.1161/CIRCRESAHA.121.318504 doi: https://doi.org/10.1161/CIRCRESAHA.121.318186
20. Mebazaa A, Davison B, Chioncel O, Cohen-Solal A, Diaz R, Filippatos G, et al. Safety,
tolerability and efficacy of up-titration of guideline-directed medical therapies
for acute heart failure (STRONG-HF): a multinational, open-label, randomised, trial.
Lancet. 2022 Dec;400(10367):1938–52. 10.1016/S0140-6736(22)01742-4 doi: https://doi.org/10.1016/S0140-6736(22)02076-1
21. Mullens W, Damman K, Harjola VP, Mebazaa A, Brunner-La Rocca HP, Martens P, et al. The
use of diuretics in heart failure with congestion - a position statement from the
Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail.
2019 Feb;21(2):137–55. 10.1002/ejhf.1371 doi: https://doi.org/10.1002/ejhf.1369
22. Crespo-Leiro MG, Metra M, Lund LH, Milicic D, Costanzo MR, Filippatos G, et al. Advanced
heart failure: a position statement of the Heart Failure Association of the European
Society of Cardiology. Eur J Heart Fail. 2018 Nov;20(11):1505–35. 10.1002/ejhf.1314 doi: https://doi.org/10.1002/ejhf.1236
23. Horwich TB, Fonarow GC, Clark AL. Obesity and the Obesity Paradox in Heart Failure.
Prog Cardiovasc Dis. 2018;61(2):151–6. 10.1016/j.pcad.2017.12.003 doi: https://doi.org/10.1016/j.pcad.2018.05.005
24. Butt JH, Petrie MC, Jhund PS, Sattar N, Desai AS, Køber L, et al. Anthropometric measures
and adverse outcomes in heart failure with reduced ejection fraction: revisiting the
obesity paradox. Eur Heart J. 2023 Apr;44(13):1136–53. 10.1093/eurheartj/eha doi: https://doi.org/10.1093/eurheartj/ehad083
Appendix
Table S1Event numbers, event
rates, and Hazard Ratios associated with categories of systolic blood pressure
at admission. All analyses are stratified by country (Switzerland or
Kyrgyzstan).
| First heart
failure hospitalisation or
all-cause death |
Events, n (%) |
Event rate (95% CI),
per 100 patient-years |
Unadjusted
HR (95% CI) |
Adjusted HR (95% CI), model* |
| SBPa <120 mmHg (n = 392) |
177 (45.2) |
70.9
(61.2–82.2) |
1.20 (0.97–1.47) |
1.19
(0.96–1.47) |
| SBPa 120–139 mmHg (n = 442) |
184 (41.6) |
60.3
(52.2–69.7) |
1
(reference) |
1
(reference) |
| SBPa 140–159 mmHg (n = 355) |
139 (39.2) |
54.1
(45.8–63.8) |
0.89
(0.72–1.12) |
0.88
(0.71–1.11) |
| SBPa ≥160 mmHg (n = 301) |
121 (40.2) |
56.5 (47.3–67.5) |
0.93
(0.74–1.18) |
1.00
(0.79–1.27) |
Table S2Event numbers, event
rates, and Hazard Ratios associated with categories of systolic blood pressure
at discharge. All analyses are stratified by country (Switzerland or
Kyrgyzstan). Median follow-up time: 1 year.
| First heart
failure hospitalisation or
all-cause death |
Events, n (%) |
Event rate (95% CI),
per 100 patient-years |
Unadjusted
HR (95% CI) |
Adjusted HR (95% CI), Model* |
| SBPd <120 mmHg (n = 629) |
258 (41.0) |
61.2 (54.2–69.1) |
1.12
(0.93–1.34) |
1.27
(1.05–1.53) |
| SBPd 120–139 mmHg (n = 548) |
221 (40.3) |
55.4
(48.6–63.2) |
1
(reference) |
1
(reference) |
| SBPd 140–159 mmHg (n = 243) |
114 (46.9) |
72.3
(60.2–86.9) |
1.22
(0.97–1.53) |
1.22
(0.97–1.54) |
| SBPd ≥160 mmHg (n = 70) |
28 (40.0) |
58.6
(40.4–84.8) |
0.99
(0.67–1.47) |
1.07
(0.71–1.60) |


