Recommendations for early mobilisation at Swiss Stroke Centres and Stroke Units: a
practice guide and discussion paper
DOI: https://doi.org/https://doi.org/10.57187/4921
Martina
Betschartab*,
Lucie Sahlic*,
Katrien Van
den Keybus Déglond*,
Jens Wuschkee*
a Department of Health, Eastern Switzerland University of
Applied Sciences, St. Gallen, OST, Switzerland
b Institute of Therapies and Rehabilitation, Cantonal
Hospital Winterthur (KSW), Winterthur, Switzerland
c Department of Therapies, Hospital
network of Neuchâtel (RHNe), Switzerland
d Département des neurosciences
cliniques, Centre Hospitalier Universitaire Vaudois (CHUV), Lausanne,
Switzerland
e Department of Neurology and Stroke Centre, Health
Eastern Switzerland, Cantonal
Hospital St. Gallen, St. Gallen, Switzerland
* Representing the Stroke Therapies Network and the Stroke Nursing Network (working
groups of
the Swiss Stroke Society)
Summary
The present recommendation document proposes a non-binding
practical guide intended to support the standardised implementation of early
mobilisation at Swiss Stroke Centres and Stroke Units, with application planned
to begin in January 2026. It was developed by the “Early Mobilisation Working
Group”, established in November 2023, to promote a consistent, evidence-based
approach to mobilising stroke survivors in the (hyper)acute phase at Swiss Stroke
Centres and Stroke Units. The group originated from the “Stroke Therapy Network”
committee of the Swiss Stroke Society and developed into a collaboration with
the “Stroke Nursing Network” committee of the Swiss Stroke Society.
The current literature does not provide sufficient evidence
to define all aspects of the dose and intensity of early mobilisation of stroke
survivors. Therefore, this guide also draws upon clinical expertise and
considers the structural and organisational conditions specific to Swiss Stroke
Centres and Stroke Units.
Some aspects of early mobilisation remain insufficiently
defined and require ongoing discussion and research. The authors explicitly invite
stakeholders and readers to provide feedback to support the continued development
and improvement of this guide.
Introduction
The use of standardised early mobilisation protocols promotes
patient-specific care and has been shown to support recovery processes. In a
cohort study involving 1117 patients in a neurological intensive care unit, the
introduction of a standardised, progressive early mobilisation protocol led to
significantly higher levels of mobilisation as well as mobility compared to
conventional practice [1, 2]. It was also associated
with a 33% shorter hospital stay and a clinically relevant decrease in
hospital-related anxiety and depression.
A national survey conducted by the Early Mobilisation
Working Group between April and June 2025 revealed variability in the
implementation of early mobilisation across Swiss Stroke Centres and Stroke
Units [3]. Of the 25 institutions
contacted, 24 responded, with 22 reporting the presence of an internal mobilisation
protocol. Among these, 14 reported consistent adherence to their protocol. All
protocols were developed based on scientific evidence and clinical expertise.
The observed 64% adherence rate to mobilisation protocols
within Swiss Stroke Centres and Stroke Units aligns with findings from previous
literature. In a study by Rethnam et al. [4], 40% of neurology clinicians − including
physicians, nurses and therapy staff − reported relying primarily on their
clinical expertise and interpretation of existing literature, citing that
current clinical practice guidelines for stroke care are often perceived as
insufficiently specific and lacking the necessary flexibility for real-world
application.
The present work aims to synthesise existing high-level
evidence concerning early mobilisation in Swiss Stroke Centres and Stroke Units
through a narrative review, intended to serve as a practical guide and
discussion paper rather than a systematic review or the development of a new
guideline. It is important to emphasise that the primary focus was on the
timing and dosage (including time points, duration and frequency) of early
mobilisation. The guide does not provide specific recommendations regarding the
type of mobilisation (e.g. techniques or concepts).
To encourage the
application of the described procedures, this paper includes open questions and
discussion points for the reader.
Methods
To inform this practical guide, three authors (LS, KVdKD, MB)
conducted a targeted literature search in PubMed, the Cochrane Library and
selected stroke guideline websites. The literature search was conducted between
December 2023 and June 2024. Publications were included when published in
English or German, and the study design was a systematic review or meta-analysis.
Additionally, national or international guidelines published or updated after
2013 were considered. Guidelines were only included if they described the use
of a standardised framework for the development of recommendations that
included assessments of the validity of the included studies. Consequently, to
avoid redundancy, the internal and external validity of the included
publications were not analysed. Clinical trials published after the included
systematic reviews and guidelines were screened by their abstracts; however,
they were only included if they provided new or conflicting information. Search
terms were “stroke” AND “acute mobilisation” OR “early mobilisation” OR “acute
management”, AND “guidelines” or “Leitlinie”, “Mobilisation” and
“Schlaganfall”, “Neurointensiv*”. This practical guide was formulated in
accordance with the Scale for Assessment of Narrative Review Articles (SANRA) [5].
In this search, the term
mobilisation included the activity levels of sitting (in bed) and out-of-bed
mobilisation (including sitting in an armchair, standing and gait activities).
The findings were presented to the Board of the Swiss Stroke
Society in autumn 2024. The discussion emphasised medical aspects that, in the
authors’ opinion, were not adequately covered in the existing literature (e.g.
inclusion of patients after haemorrhagic stroke, intravenous thrombolysis,
thrombectomy, and the literature-based medical contraindications). Additionally,
the discussion included the presentation of a decision tree (figure 1). The
feedback was integrated into the practical guide, ensuring that the
recommendations reflect both the available scientific evidence and the
consensus of clinical experts in the Swiss context.
Results
Seven international guidelines [6–11], two consensus-based
recommendations [12, 13] and two systematic reviews [14–16] (table 1) were retained,
of
which only moderate-to-strong recommendations were extracted to create the decision
tree (figure 1). No clinical trials were included.
The green numbering in figure
1 is referred to in the following text with bold numbers in brackets “(1–5)”.
Based on the available evidence, these recommendations are tentative. All
statements in this chapter are based on the listed evidence if not otherwise
stated.
Table 1Studies and guidelines included.
| Organisation / authors |
Title & reference |
| AHA/ASA – acute ischemic stroke guideline [6] |
American Heart Association / American Stroke Association |
Guidelines for the Early Management of Patients With Acute
Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management
of Acute Ischemic Stroke: A Guideline for Healthcare Professionals from the
American Heart Association/American Stroke Association. |
| NICE – Stroke and TIA Guideline [7] |
National Institute for Health and Care Excellence |
Stroke and transient ischaemic attack (TIA) in over 16s:
diagnosis and initial management (2019). NICE guideline NG128. Accessed June
2025. |
| DGAI – S3 ICU AWMF Guideline [8] |
Deutsche Gesellschaft für Anästhesiologie und
Intensivmedizin |
Deutsche Gesellschaft für Anästhesiologie und
Intensivmedizin (DGAI). S3-Leitlinie zur Lagerungstherapie und Mobilisation
von kritisch Erkrankten auf Intensivstationen. Version 3.1, 2023. Chapter 5.
Available at: https://register.awmf.org/de/leitlinien/detail/001-015.
Accessed June 2025. |
| SAP-E Stroke Action Plan Essentials [9] |
European Stroke Organisation |
SAP-E Essentials of Stroke Care; European Stroke
Organisation Action Plan: An overview of evidence-based interventions
covering the entire chain of stroke care. Available at: https://actionplan.eso-stroke.org/wp-content/uploads/2021/07/Essential-Stroke-Care_final_20210714_CPO.pdf.
Accessed April 2025. |
| Stroke Foundation – living guideline [10] |
Stroke Foundation |
Stroke Foundation. Australian and New Zealand Living
Clinical Guidelines for Stroke Management: Chapter 5 of 8: Rehabilitation.
Available at: https://informme.org.au/guidelines/living-clinical-guidelines-for-stroke-management#. Accessed June 2025. |
| KNGF – Stroke Guideline for physical therapy [11] |
Royal Dutch Society for Physical Therapy |
Royal Dutch Society for Physical Therapy (Koninklijk
Nederlands Genootschap voor Fysiotherapie, KNGF) Clinical Practice Guideline
for Physical Therapy in patients with stroke. |
| ESO – motor rehabilitation framework [12] |
European Stroke Organisation |
Motor rehabilitation after stroke: European Stroke
Organisation (ESO) consensus-based definition and guiding framework. |
| NA |
Schaller et al. (2024) [13] |
Guideline on positioning and early mobilisation in the critically
ill by an expert panel. |
| Systematic reviews* |
| NA |
Rethnam et al. (2022) [14] |
Early mobilisation post-stroke: a systematic review and
meta-analysis of individual participant data. |
| NA |
Mariana de Aquino Miranda et al. (2021) [15] |
Early mobilisation in acute stroke phase: a systematic
review. |
| EBRSR - Stroke Rehabilitation Review |
Foley et al. (2018) [16] |
Evidence-Based Review of Stroke Rehabilitation (EBRSR).
(2018). Chapter 6: The elements of stroke rehabilitation. Available at: http://www.ebrsr.com/evidence-review. Accessed June 2025. |
It is crucial to emphasise that the patient’s unique
clinical condition must always be considered when applying these
recommendations. An interprofessional exchange is crucial for this purpose. However,
the current literature does not provide evidence-based guidance on which
professional group is responsible for prescribing mobilisation, determining
continuation of bed rest or coordinating interprofessional roles. In the
absence of such data, these responsibilities must be defined at the
institutional level, according to local organisational structures and
interprofessional agreements.
Furthermore, these recommendations do not supersede medical
prescriptions or contraindications. Recommendations for medical
contraindications or criteria for discontinuation are delineated in table 2.
The contraindication criteria provide cut-offs to support clinical
decision-making regarding when mobilisation should be paused, discontinued or
refrained from.
The recommendations are for people who have had an ischaemic
stroke (1). This practical guide does not provide recommendations
regarding mobilisation after endovascular thrombectomy, and on the duration of
bed rest after intravenous thrombolysis due to a lack of recommendations in the
literature included (2). Details such as procedures, materials, provider
roles, delivery modes, personalisation strategies, and harm monitoring were not
included, as these data were insufficiently reported in the literature and
would have required a systematic expert consensus process, which was outside
the scope of this work.
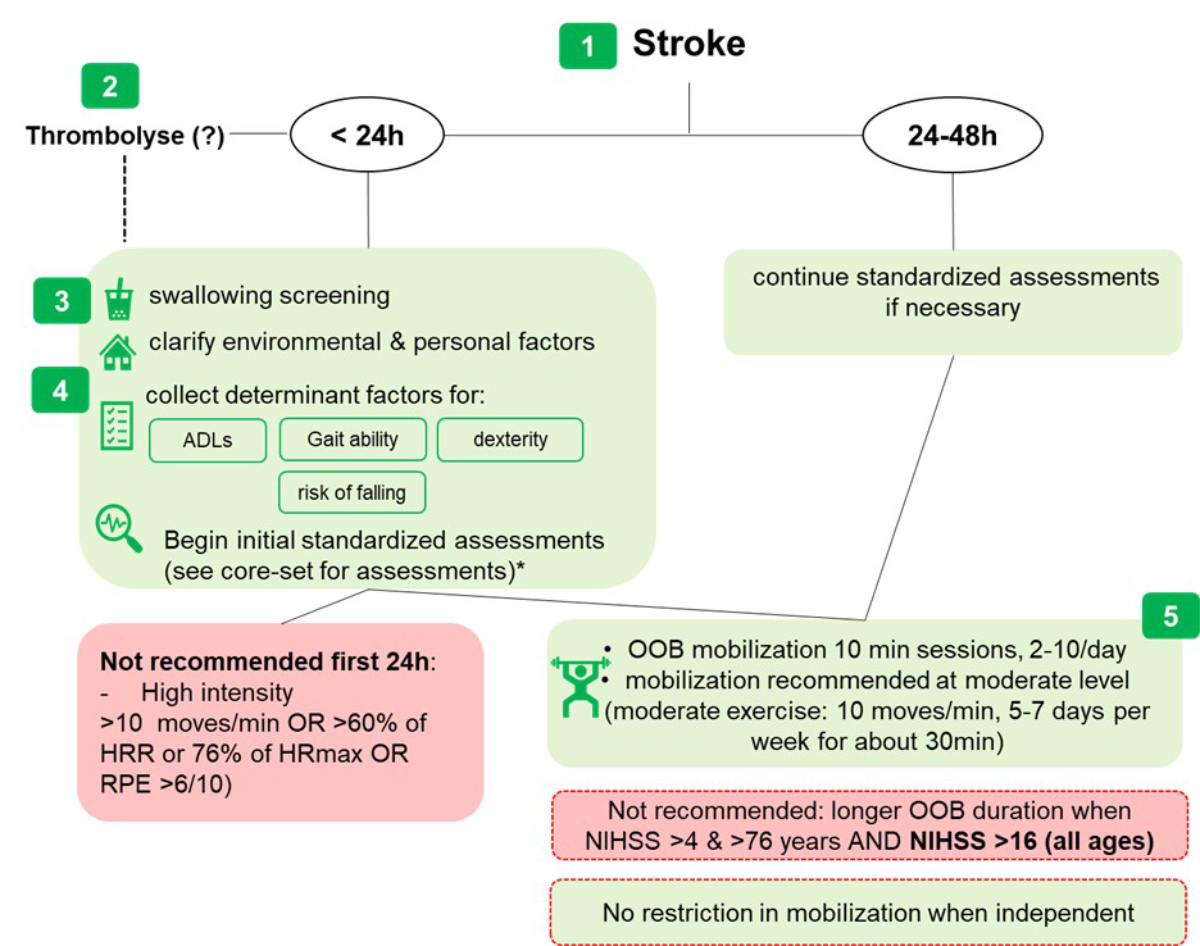
Figure 1Early mobilisation decision tree. Numbers 1–5
are placed in the “Results” section next to the corresponding explanation. (?):
no recommendations regarding Thrombolysis; *: Core-Set assessment (see
appendix); ADL: activities of daily living; HRmax: maximal
heart rate; HRR: heart rate reserve; OOB: out-of-bed; NIHSS: National
Institutes of Health Stroke Scale (ranging from 0 to 42; the higher the score,
the more severe the stroke); RPE: rate of perceived exertion measured on the
modified Borg scale ranging from 0 to 10.
Table 2Contraindications and termination criteria. Contraindications based on the
DGAI guideline, 2023, “Empfehlung 3.10” [8] and adapted from the
evaluation of a modified Early Warning Score to identify patients at risk (Subbe et
al. [17]). The criteria were
suggested as cut-offs when mobilisation should be paused, discontinued or
refrained from. Age >70 years =
higher risk of reaching unstable conditions / high dependency [17].
| Clinical parameter |
Value |
| SpO2 |
<86% |
| Heart rate increase
or heart rate (bpm) |
>30% increase from
initial value<40 or ≥130 bpm |
| Cardiac
arrhythmia |
New onset or increased
symptoms |
| Systolic blood
pressure increase |
≥40 mm Hg |
| Diastolic blood
pressure increase |
≥20 mm Hg |
| Mean arterial
pressure |
<60 or ≥110 mm Hg;
nicardipine pump |
| Respiratory rate |
>40 / min |
| Body temperature |
≤36°C or ≥38.5°C |
| Intracranial
pressure (haemorrhagic lesions only) |
≥20 cmH2O |
| Surgical
contraindications |
E.g. unstable
fractures |
| Deterioration of the
state of consciousness |
Compared to the start
of mobilisation |
| Pain |
Not amenable to
effective analgesic intervention |
First 24 hours after stroke (hyperacute phase)
According to the literature provided in table 1, mobilisation
out of bed (OOB) during the first 24 hours post-stroke can be performed in the
absence of medical contraindications [17] (figure 1 and table 1). The
only essential criterion is the aforementioned swallowing screening (3).
Generally, frequent but brief OOB mobilisations are recommended (e.g. 10-minute
sessions, 2–10 times per day) (5). This is particularly relevant for individuals
who are severely affected, as measured by an initial National Institutes of
Health Stroke Scale (NIHSS) score >16), or for individuals who are aged
>76 years and have an initial NIHSS score ≥4 [18]. Not recommended in the first
24 hours are intensive activities [10, 12, 18]. Sitting out of bed should be
actively promoted to reduce sedentary time, particularly in patients who are
unable to reposition themselves independently [19]. However, prolonged sitting
should be avoided, in order to minimise the risk of inactivity-related
complications. For patients who are already independent and safely mobile in
the first 24 hours, there are no arguments in favour of restrictions.
Within 24 hours of admission to the Stroke Unit or Stroke
Centre, clinical observation, including standardised assessments covering “body
function”, “activities” and “environmental and personal factors” as per the
International Classification of Functioning, Disability and Health, is
recommended [11] (4). If bed rest is
prescribed, the assessments are modified accordingly or conducted once the bed
rest has been discontinued. These observations serve two primary purposes.
First, to assess the patient’s abilities and the external contributing factors to
determine therapy needs and aftercare support recommendations. Second, they
promote safe mobilisation and therapy (4). A further working group of
the Stroke Therapy Network has compiled a core set of physiotherapeutic
assessments to serve these purposes. This Core-Set assessment is based on convergent
items from the literature and international guidelines and is available in the
supplemental files. It is not a formal consensus product, nor has it been validated
or published as guidance. Therefore, it should be viewed as a non-binding aid
rather than a strict standard.
24 hours to 48 hours
Three guidelines
strongly recommended that OOB mobilisation take place within the first 48 hours
[9–11]. During this period, the recommendation of short but more frequent mobilisation
out of bed continues to apply. Assessments should also be repeated for
follow-up documentation during the Stroke Unit or Stroke Centre stay. No specific
recommendations for 48 hours
to 72 hours were found.
Discussion
In this practical guide on mobilisation during the (hyper)acute
phase following ischaemic stroke, we aimed to follow the framework proposed by
Rethnam et al. [4], seeking a balance between the accuracy of information
and feasibility. Specifically, we addressed
key factors such as patient characteristics relevant to decision-making,
intervention dose and intensity, and known contraindications.
Our review indicates that current evidence regarding the
influence of patient characteristics and mobilisation dosage remains limited
and inconclusive. These limitations and their implications are further explored
in the subsequent section.
Inclusion of haemorrhagic strokes
While one systematic review included in our analysis
incorporated 268 individuals with haemorrhagic stroke (alongside 2362
individuals with ischaemic stroke), the evidence base remains comparatively
limited for this subgroup [14]. Further, none of the included
reviews or guidelines provided robust data on lesion size, anatomical location
or other imaging-based parameters relevant to mobilisation decisions in
haemorrhagic stroke. Haemorrhagic strokes are associated with higher mortality
rates [20]; however, clinical parameters
such as a Glasgow Coma Scale (GCS) score below nine may serve as more direct predictors
of outcomes than stroke subtype alone [21].
This subject was a main discussion point with the Board of
the Swiss Stroke Society. A consensus was reached at the Board, suggesting not
to include this patient population in the present guide. In light of these
gaps, we recommend that future guidelines cautiously incorporate this patient
group, guided by individual clinical status and known contraindications
specific to haemorrhagic events (table 2). Additionally, more research is
needed for this patient group.
Bedrest after intravenous thrombolysis
The included literature did not contain any recommendations
regarding OOB mobilisation in connection with intravenous thrombolysis. Research
indicates that mobilisation at six or twelve hours does not increase risk of
adverse outcomes [22–24]. Nevertheless,
post-thrombolysis mobilisation decisions are mainly medical; consequently, we
do not provide specific recommendations. However, developing a nationally
standardised protocol could enhance consistency in care and potentially improve
both safety and rehabilitation outcomes.
Head positioning
This guide does not provide recommendations on head
positioning due to inconsistent evidence. The international HeadPoST trial with
over 11,000 participants found no significant difference in disability outcomes
at 3 months between patients in a flat position for 24 hours and those with
head elevation >30°, but it has faced methodological criticisms [25, 26]. It did not
adequately
consider evidence suggesting head positioning matters in cases of major vessel
occlusion or high intracranial pressure, and may have had sampling bias
limiting subgroup effect detection.
A head elevation of 30–45° at all times post-stroke is
recommended to support lung function, reduce intracranial pressure and minimise
aspiration risk, especially in brainstem lesions [8].
Mobilisation intensity
Despite the recognised importance of intensity in
neurorehabilitation, the included literature provides little guidance on
mobilisation intensity during the acute phase of stroke. The only specific and
quantifiable parameter identified was the number of movement repetitions per
minute (figure 1, 5). No data were reported regarding the progression or
regression of intensity. Although cadence is an improvement over time-based
measures, it may not fully capture the physical and cognitive demands, which
can be significant even in brief activities, such as five steps or sit-to-stand
transitions. The American Congress of Rehabilitation Medicine Stroke Movement
Interventions Subcommittee defines intensity as: “The amount of physical or
mental work put forth by the client during a particular movement or series of
movements, exercise or activity during a defined period of time” (from ESBR,
page 35) [16]. We therefore recommend
combining cadence with training principles and monitoring intensity using heart
rate or subjective exertion scales like Borg [27]. The reference values for
moderate intensity in neurology are defined as 40–60% of heart rate reserve
(HRR) or 64–76% of maximal heart rate (HRmax), and a perceptual exertion level
of 4–5 on the modified Borg Scale, which ranges from 0 to 10 [28]. Activities or exercises
that
result in a heart rate exceeding 60% of HRR or 76% of HRmax, and a perceived
exertion level greater than 6, are classified as high intensity. These general
reference values for high intensity were incorporated into the decision tree as
extra guidance.
Standardised assessments
There is no international consensus on standardised
physiotherapeutic assessments for patients in Stroke Centres or Stroke Units.
Only the Dutch guideline specifies goals for physiotherapists in these units [11]
(figure 2).

Figure 2Role of physiotherapy in Stroke Centres or Stroke Units (own illustration based on
the Stroke Practice Guideline of the Dutch
Physiotherapy Association [11], produced with kind permission).
The Core-Set assessment presented in the supplemental files matches
the Dutch guideline but excludes assessments like the Barthel Index and
Frenchay Arm Test; as in Switzerland, nursing and occupational therapy mainly
handle these areas.
Take-home message
In general, early out-of-bed mobilisation is recommended
within the first 24 hours after stroke. However, physically intensive therapy
should be avoided during this period.
Patients should be mobilised within the first 48 hours,
ideally at a moderate intensity.
For patients aged over 76 years with moderate stroke
severity, or for severely affected patients of any age (NIHSS >16),
prolonged mobilisation sessions (>13 minutes) should be avoided.
All recommendations must be applied with careful
consideration of individual medical contraindications.
Acknowledgments
We want to thank all the members of the Stroke Therapies
Network and the contributors from the Stroke Nursing Network (Tim Horn,
Kantonsspital Baden; Gabriela Klaiber, Spital Wetzikon; and Alexandra Loewer,
Centre Hôpitalier Universitaire Vaudois, Lausanne), as well as the Board of the
Swiss Stroke Society, for their valuable feedback. We want to thank all
colleagues from the various Stroke Centres and Stroke Units for taking the time
to answer our questionnaire. We greatly appreciate the provision of the Core-Set
assessment by its authors.
Author contributions: MB, LS and KVdKD conceptualised the idea and carried out the literature review. MB
and LS
drafted the manuscript. MB, JW, LS and KVKD contributed significantly to its
finalisation.
Martina Betschart, Pt., PhD
Ostschweizer Fachhochschule
CH-9001 St. Gallen
martina.betschart[at]ost.ch
References
1. Klein KE, Bena JF, Mulkey M, Albert NM. Sustainability of a nurse-driven early progressive
mobility protocol and patient clinical and psychological health outcomes in a neurological
intensive care unit. Intensive Crit Care Nurs. 2018 Apr;45:11–7. 10.1016/j.iccn.2018.01.005
2. Klein K, Mulkey M, Bena JF, Albert NM. Clinical and psychological effects of early
mobilization in patients treated in a neurologic ICU: a comparative study. Crit Care
Med. 2015 Apr;43(4):865–73. 10.1097/CCM.0000000000000787
3. Wuschke J, Sahli L, van den Keybus Deglon K, Klaiber G, Horn T, Loewer A, Betschart M.
Use of mobilisation protocols across Swiss Stroke Centers and Units; a national survey
from 2025. forthcoming 2026.
4. Rethnam V, Hayward KS, Bernhardt J, Churilov L. Early Mobilization After Stroke: Do
Clinical Practice Guidelines Support Clinicians’ Decision-Making? Front Neurol. 2021 Feb;12:606525.
10.3389/fneur.2021.606525
5. Baethge C, Goldbeck-Wood S, Mertens S. SANRA-a scale for the quality assessment of
narrative review articles. Res Integr Peer Rev. 2019 Mar;4(1):5. 10.1186/s41073-019-0064-8
6. Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, et al. Guidelines
for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the
2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for
Healthcare Professionals From the American Heart Association/American Stroke Association.
Stroke. 2019 Dec;50(12):e344–418. 10.1161/STR.0000000000000211
7. National Institute for Health and Care Excellence. Stroke and transient ischaemic
attack in over 16s: diagnosis and initial management: NICE guideline Reference number:NG128.
April 2022. https://www.nice.org.uk/guidance/ng128. Accessed Juni 2025.
8. AWMF. S3-Leitlinie Lagerungstherapie und Mobilisation von kritisch Erkrankten auf
Intensivstation. 2023-2027. https://register.awmf.org/de/leitlinien/detail/001-015. Accessed June 2025.
9. Steiner T, Turc G, Dawson J, Sunnerhagen K, Christensen H. SAP-E Essentials of Stroke
Care; European Stroke Organisation Action Plan: An overview of evidence-based interventions
covering the entire chain of stroke care. https://actionplan.eso-stroke.org/wp-content/uploads/2021/07/Essential-Stroke-Care_final_20210714_CPO.pdf. Accessed April 2025.
10. Australian and New Zealand LIving Clinical Guidelines for Stroke Managment. New Zealand
LIving Clinical Guidelines for Stroke Managment: Chapter 5 of 8: Rehabilitation. 28.07.2023.
https://app.magicapp.org/#/guideline/6841. Accessed June 2025.
11. Veerbeek J, van Wegen E, van Peppen R, Hendriks H, Rietberg MB, van der Wees P, et
al. KNGF Clinical Practice Guideline for Physical Therapy in patients with stroke:
Practice Guidelines. 2014. https://www.dsnr.nl/wp-content/uploads/2012/03/stroke_practice_guidelines_2014.pdf
12. Kwakkel G, Stinear C, Essers B, Munoz-Novoa M, Branscheidt M, Cabanas-Valdés R, et
al. Motor rehabilitation after stroke: european Stroke Organisation (ESO) consensus-based
definition and guiding framework. Eur Stroke J. 2023 Dec;8(4):880–94. 10.1177/23969873231191304
13. Schaller SJ, Scheffenbichler FT, Bein T, Blobner M, Grunow JJ, Hamsen U, et al. Guideline
on positioning and early mobilisation in the critically ill by an expert panel. Intensive
Care Med. 2024 Aug;50(8):1211–27. 10.1007/s00134-024-07532-2
14. Rethnam V, Langhorne P, Churilov L, Hayward KS, Herisson F, Poletto SR, et al. Early
mobilisation post-stroke: a systematic review and meta-analysis of individual participant
data. Disabil Rehabil. 2022 Apr;44(8):1156–63. 10.1080/09638288.2020.1789229
15. Mariana de Aquino Miranda J, Mendes Borges V, Bazan R, José Luvizutto G, Sabrysna
Morais Shinosaki J. Early mobilization in acute stroke phase: a systematic review.
Top Stroke Rehabil. 2023 Mar;30(2):157–68. 10.1080/10749357.2021.2008595
16. Foley N, Hussein N, Saikaley M, Iruthayarajah J, Wiener J, Speechley M, et al. The
Elements of Stroke Rehabilitation: Chapter 6. http://www.ebrsr.com/evidence-review/6-elements-stroke-rehabilitation
17. Subbe CP, Kruger M, Rutherford P, Gemmel L. Validation of a modified Early Warning
Score in medical admissions. QJM. 2001 Oct;94(10):521–6. 10.1093/qjmed/94.10.521
18. Bernhardt J, Churilov L, Ellery F, Collier J, Chamberlain J, Langhorne P, et al.;
AVERT Collaboration Group. Prespecified dose-response analysis for A Very Early Rehabilitation
Trial (AVERT). Neurology. 2016 Jun;86(23):2138–45. 10.1212/WNL.0000000000002459
19. Norvang OP, Hokstad A, Taraldsen K, Tan X, Lydersen S, Indredavik B, et al. Time spent
lying, sitting, and upright during hospitalization after stroke: a prospective observation
study. BMC Neurol. 2018 Sep;18(1):138. 10.1186/s12883-018-1134-0
20. Waziry R, Heshmatollah A, Bos D, Chibnik LB, Ikram MA, Hofman A, et al. Time Trends
in Survival Following First Hemorrhagic or Ischemic Stroke Between 1991 and 2015 in
the Rotterdam Study. Stroke. 2020 Mar;51(3):STROKEAHA119027198. 10.1161/STROKEAHA.119.027198
21. Namale G, Kamacooko O, Makhoba A, Mugabi T, Ndagire M, Ssanyu P, et al. Predictors
of 30-day and 90-day mortality among hemorrhagic and ischemic stroke patients in urban
Uganda: a prospective hospital-based cohort study. BMC Cardiovasc Disord. 2020 Oct;20(1):442.
10.1186/s12872-020-01724-6
22. Arnold SM, Dinkins M, Mooney LH, Freeman WD, Rawal B, Heckman MG, et al. Very early
mobilization in stroke patients treated with intravenous recombinant tissue plasminogen
activator. J Stroke Cerebrovasc Dis. 2015 Jun;24(6):1168–73. 10.1016/j.jstrokecerebrovasdis.2015.01.007
23. Momosaki R, Yasunaga H, Kakuda W, Matsui H, Fushimi K, Abo M. Very Early versus Delayed
Rehabilitation for Acute Ischemic Stroke Patients with Intravenous Recombinant Tissue
Plasminogen Activator: A Nationwide Retrospective Cohort Study. Cerebrovasc Dis. 2016;42(1-2):41–8.
10.1159/000444720
24. Silver B, Hamid T, Khan M, Di Napoli M, Behrouz R, Saposnik G, et al. 12 versus 24 h
bed rest after acute ischemic stroke thrombolysis: a preliminary experience. J Neurol
Sci. 2020 Feb;409:116618. 10.1016/j.jns.2019.116618
25. Anderson CS, Arima H, Lavados P, Billot L, Hackett ML, Olavarría VV, et al.; HeadPoST
Investigators and Coordinators. Cluster-Randomized, Crossover Trial of Head Positioning
in Acute Stroke. N Engl J Med. 2017 Jun;376(25):2437–47. 10.1056/NEJMoa1615715
26. Alexandrov AW, Tsivgoulis G, Hill MD, Liebeskind DS, Schellinger P, Ovbiagele B, et
al. HeadPoST: rightly positioned, or flat out wrong? Neurology. 2018 May;90(19):885–9.
10.1212/WNL.0000000000005481
27. Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc. 1982;14(5):377–81.
doi: https://doi.org/10.1249/00005768-198205000-00012
28. MacKay-Lyons M, Billinger SA, Eng JJ, Dromerick A, Giacomantonio N, Hafer-Macko C,
et al. Aerobic Exercise Recommendations to Optimize Best Practices in Care After Stroke:
AEROBICS 2019 Update. Phys Ther. 2020 Jan;100(1):149–56. 10.1093/ptj/pzz153
Appendix
The appendix is available in the pdf version of the article at https://doi.org/10.57187/4921.

