
Flow chart.
DOI: https://doi.org/https://doi.org/10.57187/4790
Lung cancer remains the leading cause of cancer-related mortality in France, with 52,777 new cases in 2023. According to the French National Cancer Institute, the 5-year overall survival (OS) rate across all stages is estimated at 20%, whereas for metastatic non-small cell lung cancer (NSCLC), this rate is below 10%. The KBP-CPHG study [1] reported that for all histologies combined, the 2-year OS rate in 2020 was 47.8%, compared with 21.2% in 2000, representing a 26.6% increase over 20 years. Early mortality rates remain elevated, mainly due to metastatic patients who remain largely incurable.
In light of this public health challenge, there has been a significant increase in therapeutic innovations, particularly with the emergence of targeted therapies for oncogene-addicted cancers and immunotherapy, especially immune checkpoint inhibitors, such as anti-PD-(L)1 (i.e. programmed cell death receptor 1/programmed death ligand 1). These systemic therapies have become the cornerstone of management for metastatic NSCLC, which represents more than 60% of cases at diagnosis [1].
The advent of immunotherapy in the treatment of metastatic stages has significantly improved progression-free survival (PFS) and overall survival (OS). A study conducted by Reck et al. [2] demonstrated that patients with PD-L1 expression greater than 50% who received immunotherapy had a median PFS of 10.3 months, compared with 6 months in those receiving standard chemotherapy. However, the emergence of resistance mechanisms to anti-PD-(L)1 agents, as well as chemotherapy, remains a major research focus [3]. In this context, a therapeutic strategy combining systemic treatment with local treatment may provide an alternative approach to slowing the progression of metastatic disease while preserving options for systemic therapies for potential future lines of treatment.
The concept of oligoprogression, introduced by Hellman and Weichselbaum in 1995 [4], refers to the appearance or progression of a limited number of metastases (typically 3 to 5 metastases), reflecting at least a partial escape from systemic therapies [5]. This concept is related to the broader definition of oligometastatic disease, which refers to a limited number of metastases or metastatic sites that exhibit more indolent behaviour than polymetastatic disease [6]. Oligopersistence refers to the presence of a small number of cancer cells that remain active at a limited number of sites after treatment [7]. This condition is thus conducive to surgical or ablative interventions targeting progressing sites with the aim of increasing survival, slowing the disease, preserving systemic treatment lines, and improving patients’ quality of life.
Recent research has validated the efficacy of ablative radiotherapy in the management of oligoprogression in various cancer types, demonstrating its impact on PFS and OS [8]. Moreover, a study by Gomez et al. on local treatment of oligometastatic NSCLC at diagnosis (i.e. metastatic disease at diagnosis but with a limited number of metastases, typically 1 to 5 [4]) in the first-line treatment setting reported a significant increase in OS (41.2 months) and PFS (14.2 months) [9]. However, this phase II trial, which began in 2012, did not include immunotherapy in the management of metastatic NSCLC. Therefore, the benefit of combining immunotherapy with local treatment in these patients remains to be determined.
Given the absence of robust prospective studies and the need to investigate the benefit of combining immunotherapy and local treatment, we conducted this hypothesis-generating exploratory study as an initial step to generate real-world data. The primary objective of this study was to assess the effects of integrating local treatment on PFS and OS in patients with metastatic NSCLC receiving immunotherapy and experiencing oligoprogression. The secondary objective was to assess this strategy implemented at our centre over several years and to compare it with available data in the literature.
This monocentric retrospective study was based on the Oncoloire Thoracic multidisciplinary team meeting (MDT) database. This study was approved by Saint Etienne’s local ethics committee.
The study included patients with metastatic NSCLC (stage IV) with oligoprogression or dissociated progression while receiving immunotherapy. The included patients were treated between 1 April 2015 and 16 December 2022 with first-line metastatic immunotherapy (i.e., monotherapy based on PD-L1 status or in combination with chemotherapy) or as subsequent-line therapy. Patients with progression under immunotherapy who were not eligible for local treatment were excluded, along with patients with stage III receiving consolidative immunotherapy and patients with a histology other than NSCLC. Patients with oncogene addictions (e.g. EGFR, BRAF, ALK rearrangements, and ROS) except for KRAS mutations were also excluded to eliminate potential confounding factors, as ablative treatments such as radiotherapy are often part of the standard clinical practice for these patients. Eligible patients could receive local treatments (surgery, ablative therapy, radiotherapy) for all progressing lesions, either with or without the continuation of immunotherapy. All decisions were made after multidisciplinary discussion at the Oncoloire Thoracic MDT. No control group was defined in this study; patients who did not receive local treatment were not eligible according to our criteria. These patients either continued immunotherapy, switched to a different systemic treatment, or were placed under surveillance alone.
Of the initial 862 patients, 813 patients were excluded, and 49 patients were included in the study (figure 1).
Clinical data, along with information regarding systemic treatment and oligoprogression, were retrospectively collected from medical records. Immunotherapy included anti-PD1 agents (e.g. nivolumab and pembrolizumab) and anti-PD-L1 agents such as atezolizumab. Local treatment could include stereotactic or conformal radiotherapy or surgical treatment.
For patient characteristics, categorical data were presented as absolute numbers (n) and percentages, and continuous data were expressed as medians with standard deviations and first (Q1) and third (Q3) quartiles. Missing data were not included in the statistical analysis.
For survival analysis, Kaplan-Meier curves were used to estimate survival functions, with median survival and its 95% confidence intervals (CI). The 5-year survival probability was also calculated. Patients lost to follow-up during the study period were right-censored in the analysis. Overall survival (OS) was calculated from the initiation of local treatment. Similarly, progression-free survival (PFS) was evaluated using the same methods, taking into account data censoring, which allowed the quantification of the time to disease progression or death, with corresponding median estimates and CI. Statistical analysis was performed using R software (URL: https://www.R-project.org/, R Foundation for Statistical Computing, Vienna, Austria) for statistical computing and graphics.
After the selection process (figure 1), a total of 49 patients treated with immunotherapy for metastatic NSCLC and who received additional local treatment for oligoprogression were enrolled in this study.

Flow chart.
In this cohort, 63% of patients were male, and 37% were female. The median age at diagnosis was 66 years (IQR: 59-72). Notably, 96% of patients had a history of smoking, with a median consumption of 40 pack-years; 40% of these patients were still active smokers at the time of diagnosis. In addition, 16% of patients had a history of vascular disease, and 37.8% had renal insufficiency. All clinical characteristics are summarised in table 1.
Table 1Patient characteristics (n = 49).
| Age at diagnosis (years) – median (IQR, SD) | 66.00, ([59.00–72.00], 9.13) | |
| Sex – no. (%) | Male | 31 (63%) |
| Female | 18 (37%) | |
| Body Mass Index (IQR, SD) | 24.30 ([21.56–28.16], 5.11) | |
| Smoking status – no. (%) | Current smoker | 20 (41%) |
| Former smoker | 27 (55%) | |
| Non-smoker | 2 (4%) | |
| Tobacco pack-years (IQR, SD) | 40.00, ([30.00–50.00], 19.20) | |
| Asbestos exposure – no. (%) | 8 (16%) | |
| Chronic alcoholism – no. (%) | 6 (12%) | |
| Cardiac (rhythmic/ischemic) history – no. (%) | 9 (18%) | |
| High blood pressure – no. (%) | 22 (45%) | |
| Diabetes | 9 (18%) | |
| Vascular disease history – no. (%) | 8 (16%) | |
| Respiratory disease history – no. (%) | 16 (62%) | |
| Previous cancer – no. (%)* | 11 (22.4%) | |
* Metastatic prostate adenocarcinoma in remission (n = 1), oropharyngeal squamous cell carcinoma (n = 2), colon cancer (n = 1), operated cervical cancer (n = 1), localised prostate cancer (n = 2), bladder cancer (n = 1), esophageal, adenocarcinoma (n = 1), renal carcinoma (n = 1), thyroid carcinoma (n = 1), urothelial carcinoma (n = 1), follicular lymphoma in remission (n = 1)
IQR: interquartile range; SD: standard deviation
Regarding NSCLC characteristics, 63% of patients had adenocarcinoma and 35% had squamous cell carcinoma. In terms of PD-L1 expression levels, 28.6% of patients exhibited expression of less than 1%, 33.3% showed expression ranging from 1% to 49%, and 38.1% demonstrated high expression levels (50–100%). The most frequently affected metastatic sites were the lymph nodes (82.9%), the lungs (42.5%), and the brain (31.9%). Before the initiation of immunotherapy, 31% of patients had an Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0, whereas 67% had a PS of 1. KRAS mutations were present in 47.7% of cases (table 2).
Table 2Oncological characteristics.
| Anatomopathology (n = 49) | Adenocarcinoma | 31 (63%) |
| Squamous cell carcinoma | 17 (35%) | |
| Others* | 1 (2%) | |
| KRAS mutation* (n = 44) | 21 (47.7%) | |
| PDL1 status (n = 42) | < 1% | 12 (28.6%) |
| 1-50% | 14 (33.3%) | |
| 50-100% | 16 (38.1%) | |
| Stage at initiation of immunotherapy*** (n = 47) | IIIA | 2 (0.4%) |
| IIIB | 2 (0.4%) | |
| IIIC | 1 (0.2%) | |
| IV | 42 (89%) | |
| Metastatic site at diagnosis** (n = 47) | Brain | 15 (31.9%) |
| Lung | 20 (42.5%) | |
| Lymph node | 39 (82.9%) | |
| Liver | 2 (4.2%) | |
| Adrenal gland | 10 (21.3%) | |
| Bone | 15 (31.9%) | |
* KRAS mutations were not investigated in squamous cell carcinomas. There were 21 KRAS mutations, including G12A, G12C, G12D, G12V, and G12R. Six adenocarcinomas did not show mutations, and there were four missing values in the adenocarcinoma group.
** Number of metastases and metastatic site at the time of diagnosis of metastatic disease.
*** Stage III cases were considered metastatic because they were not eligible for local treatment.
Regarding therapeutic management, before initiating local treatment, 53.1% of patients were receiving a combination of chemotherapy and immunotherapy, whereas 46.9% were receiving immunotherapy exclusively.
At the point of local treatment initiation, 63.3% of patients were in the first line of treatment, 30.6% were in the second line, 4.1% were in the third line, and 2.4% were in the fourth line of treatment.
A total of 71% of patients did not experience immunotherapy-related toxicity. Among those who did, the most frequently reported toxicity was rheumatological, with diffuse arthralgia being the predominant issue, affecting 10.2% of this cohort (table 3).
Table 3Local treatment characteristics.
| Line number of local treatment | L1 | 31 (63.3%) |
| L2 | 15 (30.6%) | |
| L3 | 2 (4.1%) | |
| L4 | 1 (2%) | |
| Systemic treatment after local treatment | 21 (47.7%) | |
| Chemotherapy + immunotherapy | 26 (53.1%) | |
| Immunotherapy alone | 23 (46.9%) | |
| Time to onset of oligoprogression after initiation of immunotherapy | Mean (months) | 13 |
| Median (months) | 10 | |
| Continuation of systemic treatment after local treatment | 21 (42.9%) | |
| Type of local treatment | Radiotherapy | |
| ... Stereotactic | 30 (61.2%) | |
| ... Conformal | 12 (24.5%) | |
| Surgery | 4 (8.2%) | |
| Surgery + stereotactic treatment | 3 (6.1%) | |
| Radiofrequency | 0 | |
| Number of lesions defined as oligoprogressive and treated with local therapy* | 1 | 36 (73.5%) |
| 2 | 11 (22.4%) | |
| 3 | 2 (4.1%) | |
| Metastasis site of local treatment | Lung | 15 (30.6%) |
| Lymph node | 8 (16.3%) | |
| Brain | 23 (46.9%) | |
| Adrenal gland | 2 (4.1%) | |
| Bone | 4 (8.2%) | |
| Skin | 1 (2%) | |
| Performance status at the initiation of immunotherapy (n = 48) | 0 | 15 (31.9%) |
| 1 | 32 (67%) | |
| 2 | 1 (2.1%) | |
| Performance status after first-line local treatment (n = 41) | 0 | 17 (41.5%) |
| 1 | 18 (43.9%) | |
| 2 | 4 (9.8%) | |
| 3 | 1 (2.4%) | |
| 4 | 1 (2.4%) | |
| Immunotherapy toxicities | No toxicity | 35 (71%) |
| Digestive | 2 (4.1%) | |
| Pneumopathy | 2 (4.1%) | |
| Neurological | 2 (4.1%) | |
| Cutaneous | 2 (4.1%) | |
| Rheumatological | 5 (10.2%) | |
| Hepatic | 2 (4.1%) | |
| Endocrinological | 6 (12.3%) | |
| Adverse effects of local treatment (n = 45) | No event | 39 (86.7%) |
| Radiation pneumonitis | 3 (6.7%) | |
| Asthenia | 1 (2.2%) | |
| Headache | 1 (2.2%) | |
| Oesophagitis | 2 (4.4%) | |
* One patient received local treatment for three metastases in the same organ (brain); one patient received local treatment for two pulmonary lesions and one bone lesion. Among the patients who had two metastases, six of them received local treatment in the same organ (five in the brain and one in the bone), and five received local treatment in two different organs (brain and lymph node). Thirty-six patients received local treatment for a single lesion.
The median time to the onset of oligoprogression following the initiation of immunotherapy was 10 months. The primary local treatment employed was radiotherapy, accounting for 85.7%, with a notable preference for stereotactic radiotherapy. The second most commonly used local treatment was surgery, comprising 14.3%, followed by a combination of surgery and stereotactic treatment at 6.1%. In 42.9% of cases, patients continued systemic treatment following local therapy. For 73.5% of patients, local treatment involved addressing a single metastasis. Among patients with two metastases, five received local treatment in two different organs, whereas six were treated within the same organ. Immunotherapy added to local treatment did not appear to be associated with an increased risk of toxicity (table 3).
The median overall survival was 28 months (95% CI: 17.4%–33%) with a 5-year overall survival rate of 12.36% (figure 2). The median progression-free survival following local treatment was 7.56 months (95% CI: 6.01%–11%) (figure 3).
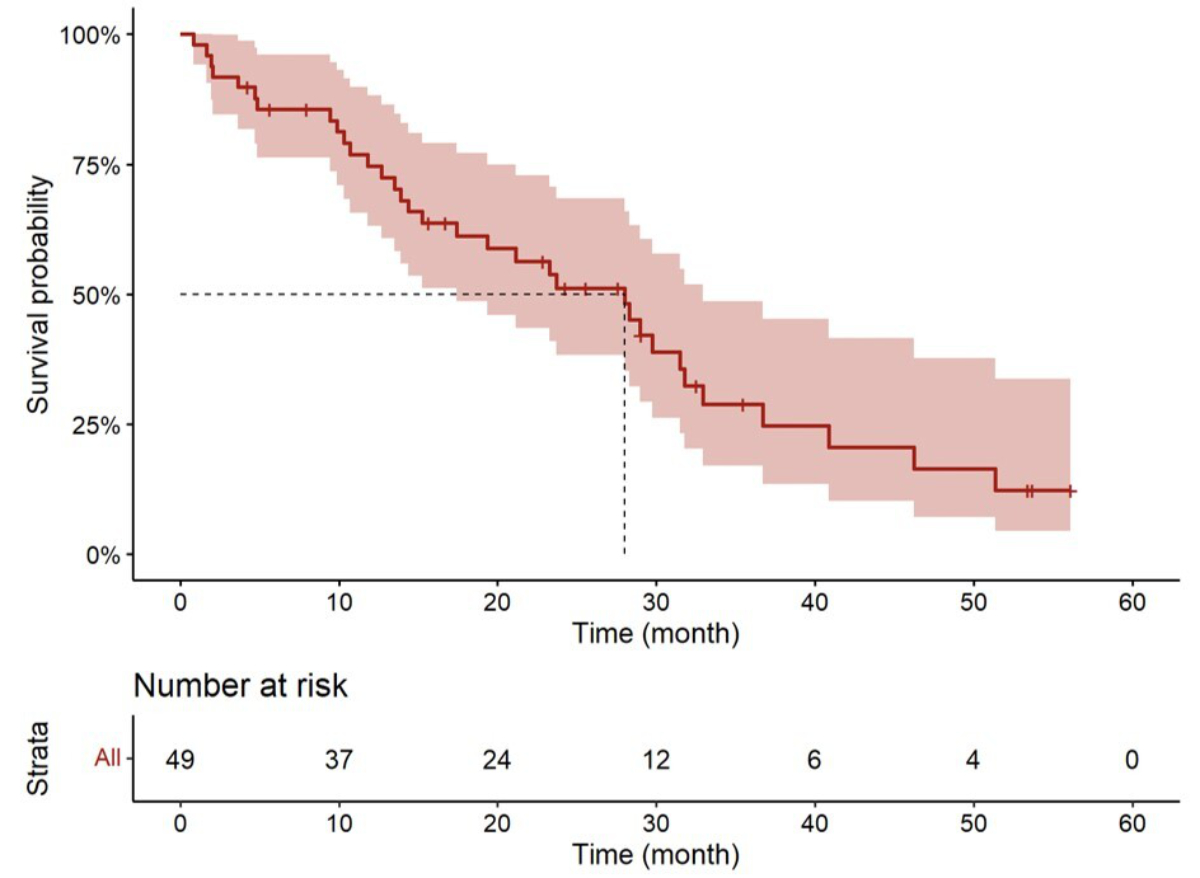
Overall survival from the time of local treatment.
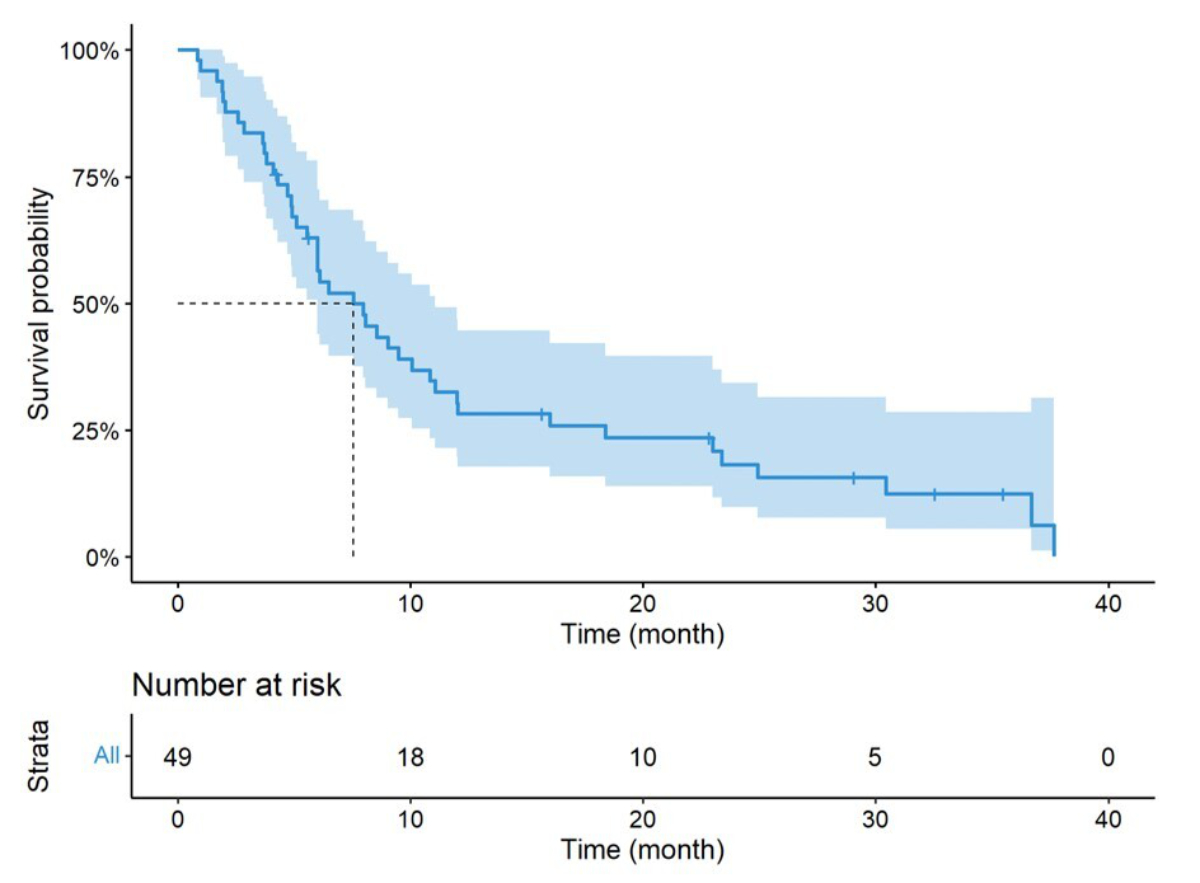
Progression-free survival from the time of local treatment.
In this exploratory study, we analysed the clinical and oncological characteristics, overall survival, and progression-free survival of a single-centre cohort of 49 patients with metastatic non-small cell lung cancer (NSCLC) with oligoprogression treated with systemic immunotherapy and additional local therapy. Our results show that incorporating local ablative therapy as part of the treatment of metastatic NSCLC treated with immunotherapy alone or in combination with chemotherapy appears to be a therapeutic strategy for slowing disease progression. Similarly, another cohort of 78 patients treated with local ablative treatment with pembrolizumab was associated with higher PFS and OS compared with pembrolizumab alone in selected patients with synchronous oligometastatic NSCLC [10]. By contrast, a recent cohort study found no significant prognostic advantage of these treatments for patients experiencing oligoprogression; however, a trend indicating enhanced local control and extended overall survival (OS) was noted [11]. In view of these contradictory results, randomised prospective studies are urgently needed.
Overall, the patient characteristics were consistent with those expected in the general population of patients with metastatic NSCLC. Our cohort was predominantly male, with an average age at diagnosis of 66 years. From an oncological perspective, the majority of patients (63%) had adenocarcinoma. Within our cohort, we noted a higher percentage of patients with high PD-L1 expression (greater than 50%) compared with most previous studies (38.1% vs 23–28% reported in the KEYNOTE 024 study [12]). This is not surprising given that these patients benefit most from immunotherapy in terms of objective response and survival and are therefore likely to develop oligoprogression during the disease course.
We observed a median OS of 28 months, with survival calculated from the date of initiation of local treatment. The 5-year OS rate was 12.36%. The PFS was 7.56 months, suggesting that local treatment delays the need for a change in systemic therapy. Additionally, a drop in the PFS curve to less than 8 months was observed, which may be attributed to patient selection bias and variability in the timing of immunotherapy initiation (first line or later lines). Some patients were included in the study after only two cycles of immunotherapy with almost immediate progression, while others were included outside the first-line metastatic treatment setting. Only 63.3% of patients received local treatment while receiving immunotherapy as first-line therapy.
Although the reported median OS of 28 months and PFS of 7.56 months appear encouraging, the wide confidence intervals and limited sample size greatly restrict the robustness of these findings. Larger, robust prospective studies are therefore essential to confirm these findings.
The most significant data regarding oligoprogression in NSCLC are derived from patients with oncogene-addicted NSCLC treated with tyrosine kinase inhibitors (TKIs). TKIs can induce primary resistance by selectively favouring resistant tumour clones or cause secondary resistance by altering tumour phenotypes [6,13]. Local therapy has been suggested to eliminate the evolutionary reservoir of resistant clones, enabling continuation of systemic therapy, including targeted therapy [14,15].
A retrospective study conducted by Hu, Xu et al. [16] compared survival in patients with EGFR (epidermal growth factor receptor)-mutated NSCLC with oligometastases or oligoprogression treated with either TKIs alone or TKIs combined with local therapy. The PFS in the group receiving combined therapy was significantly higher than in the TKI-only group (median PFS of 15 months vs 10 months). Similarly, the median OS was 34 months vs 21 months in favour of the TKI-plus-local therapy group. Several studies advocate for the addition of local therapy to systemic therapy in patients with NSCLC with oncogene addiction, showing promising survival results, although not achieving a cure in patients with mutations [17].
Similar outcomes might be expected in patients without mutations receiving immunotherapy combined with local treatment, warranting investigation through prospective trials. Limited data are available on the benefits of local therapy in patients with metastatic NSCLC receiving immunotherapy [13]. Some case reports have shown a long-term survival benefit with this approach [18], and small retrospective studies suggest that combining immunotherapy with local treatment may improve OS in metastatic NSCLC [13], as observed in a study by Guisier, which reported a post-local treatment PFS of 13.2 months [19].
Our study has several limitations, primarily those inherent to retrospective studies, including missing data on patient characteristics, adverse effects of local treatment, and performance status at various points during disease progression. Additionally, the small sample size of 49 patients introduces variability, reducing the precision of statistical estimates. This also increases the risk of sampling bias, making the cohort less representative of the general population. Selection bias was present, resulting in a cohort that did not accurately reflect the general NSCLC population. Indeed, the sample exhibited significant heterogeneity in terms of PD-L1 expression levels, sites of metastasis, and the stage of systemic therapy administered. The quality and precision of historical data collection also varied, potentially leading to inaccuracies or incomplete information. For example, although we were able to quantify immunotherapy-related toxicities, none of them were graded according to CTCAE (Common Terminology Criteria for Adverse Events). Finally, a major methodological issue in this study was the lack of a control group, preventing any comparative analysis. Despite these limitations, this exploratory study is original and incorporates several types of local treatment. Few studies have explored this topic [13], and no randomised studies have been conducted to date.
An analysis of the PFS curve within this cohort may potentially identify two types of population: those who exhibit early progression following local treatment, raising doubts about the efficacy of this approach, and those with a likely more indolent form of the disease, who demonstrate extended survival and may benefit from the integration of local treatment with ongoing immunotherapy. Beyond potential patient selection bias, further analyses of these subgroups could be of interest, such as univariate analyses on certain criteria like PD-L1 expression levels and whether systemic therapy was continued. Additional univariate analyses on factors such as the metastatic site of local treatment or the number of treated metastases could also be of interest. Unfortunately, the small sample size of our cohort did not allow for such analyses.
Currently, the selection of patients and the identification of predictive and prognostic biomarkers of primary and secondary resistance to immunotherapy remain unresolved [13]. Several prospective clinical trials are ongoing to evaluate the addition of local treatment, such as radiotherapy, in patients with oligoprogressive NSCLC treated with immunotherapy (NCT03158883, NCT04549428) [13], including the GFPC 08-2021 OLIMMUNE study (Correspondance en Onco-Thoracique, June 2024), a non-randomised, phase II prospective trial evaluating the addition of local treatment in oligoprogressive NSCLC treated with immunotherapy.
The addition of local treatment, such as surgery or stereotactic/conventional radiotherapy, in patients with metastatic NSCLC treated with immunotherapy, with or without chemotherapy, and experiencing oligoprogression appears to be a promising therapeutic option. This strategy has been implemented in our centre since immunotherapy became available for NSCLC, mirroring practices in other centres across France. Despite the promising data of this exploratory study, the results of this retrospective study remain preliminary and must be interpreted with caution. Numerous prospective clinical trials are currently underway to investigate the incorporation of local treatments, such as radiotherapy, for patients with oligoprogressive NSCLC who are receiving immunotherapy. In perspective, randomised controlled trials with robust patient selection criteria are urgently needed to confirm the place of local therapies in the management of oligometastatic patients.
The datasets used and/or analysed in the current study are available from the corresponding author upon reasonable request.
This study received no funding.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflict of interest related to the content of this manuscript was disclosed.
1. Debieuvre D, Locher C, Falchero L, Duval Y, Molinier O, Morel H, et al. Vingt ans d’épidémiologie des cancers broncho-pulmonaires diagnostiqués dans les hôpitaux non-universitaires français, l’étude KBP-2020-CPHG comparée aux éditions 2000 et 2010. Rev Malad Respir Actual. 2022 Jan;14(1):24–5. doi: https://doi.org/10.1016/j.rmra.2021.11.038
2. Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, et al.; KEYNOTE-024 Investigators. Pembrolizumab versus Chemotherapy for PD-L1-Positive Non-Small-Cell Lung Cancer. N Engl J Med. 2016 Nov;375(19):1823–33. doi: https://doi.org/10.1056/NEJMoa1606774
3. Tsai CJ, Yang JT, Shaverdian N, Patel J, Shepherd AF, Eng J, et al.; CURB Study Group. Standard-of-care systemic therapy with or without stereotactic body radiotherapy in patients with oligoprogressive breast cancer or non-small-cell lung cancer (Consolidative Use of Radiotherapy to Block [CURB] oligoprogression): an open-label, randomised, controlled, phase 2 study. Lancet. 2024 Jan;403(10422):171–82. doi: https://doi.org/10.1016/S0140-6736(23)01857-3
4. Hellman S, Weichselbaum RR. Oligometastases. J Clin Oncol. 1995 Jan;13(1):8–10. doi: https://doi.org/10.1200/JCO.1995.13.1.8
5. Planchard D, Popat S, Kerr K, Novello S, Smit EF, Faivre-Finn C, et al.; ESMO Guidelines Committee. Metastatic non-small cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018 Oct;29 Suppl 4:iv192–237. doi: https://doi.org/10.1093/annonc/mdy275
6. Franceschini D, De Rose F, Cozzi S, Franzese C, Rossi S, Finocchiaro G, et al. The use of radiation therapy for oligoprogressive/oligopersistent oncogene-driven non small cell lung cancer: state of the art. Crit Rev Oncol Hematol. 2020 Apr;148:102894. doi: https://doi.org/10.1016/j.critrevonc.2020.102894
7. Guckenberger M, Lievens Y, Bouma AB, Collette L, Dekker A, deSouza NM, et al. Characterisation and classification of oligometastatic disease: a European Society for Radiotherapy and Oncology and European Organisation for Research and Treatment of Cancer consensus recommendation. Lancet Oncol. 2020 Jan;21(1):e18–28. doi: https://doi.org/10.1016/S1470-2045(19)30718-1
8. Palma DA, Olson R, Harrow S, Gaede S, Louie AV, Haasbeek C, et al. Stereotactic Ablative Radiotherapy for the Comprehensive Treatment of Oligometastatic Cancers: Long-Term Results of the SABR-COMET Phase II Randomized Trial. J Clin Oncol. 2020 Sep;38(25):2830–8. doi: https://doi.org/10.1200/JCO.20.00818
9. Gomez DR, Tang C, Zhang J, Blumenschein GR Jr, Hernandez M, Lee JJ, et al. Local Consolidative Therapy Vs. Maintenance Therapy or Observation for Patients With Oligometastatic Non-Small-Cell Lung Cancer: Long-Term Results of a Multi-Institutional, Phase II, Randomized Study. J Clin Oncol. 2019 Jun;37(18):1558–65. doi: https://doi.org/10.1200/JCO.19.00201
10. Lee HI, Choi EK, Kim SS, Shin YS, Park J, Choi CM, et al. Local Ablative Therapy Combined With Pembrolizumab in Patients With Synchronous Oligometastatic Non-Small Cell Lung Cancer: A Recursive Partitioning Analysis. Int J Radiat Oncol Biol Phys. 2024 Nov;120(3):698–707. doi: https://doi.org/10.1016/j.ijrobp.2024.05.015
11. Morinaga D, Sakakibara-Konishi J, Kamada R, Kashima M, Tsuji K, Ito S, et al. Frequency and Prognostic Impact of Local Ablation Therapy for Oligoprogression in Non-Small Cell Lung Cancer. Thorac Cancer. 2025 Jul;16(13):e70119. doi: https://doi.org/10.1111/1759-7714.70119
12. Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, et al. Updated Analysis of KEYNOTE-024: Pembrolizumab Versus Platinum-Based Chemotherapy for Advanced Non-Small-Cell Lung Cancer With PD-L1 Tumor Proportion Score of 50% or Greater. J Clin Oncol. 2019 Mar;37(7):537–46. doi: https://doi.org/10.1200/JCO.18.00149
13. Prelaj A, Pircher CC, Massa G, Martelli V, Corrao G, Lo Russo G, et al. Beyond First-Line Immunotherapy: Potential Therapeutic Strategies Based on Different Pattern Progressions: Oligo and Systemic Progression. Cancers (Basel). 2021 Mar;13(6):1300. doi: https://doi.org/10.3390/cancers13061300
14. Kim C, Hoang CD, Kesarwala AH, Schrump DS, Guha U, Rajan A. Role of Local Ablative Therapy in Patients with Oligometastatic and Oligoprogressive Non-Small Cell Lung Cancer. J Thorac Oncol. 2017 Feb;12(2):179–93. doi: https://doi.org/10.1016/j.jtho.2016.10.012
15. Gerlinger M, Rowan AJ, Horswell S, Math M, Larkin J, Endesfelder D, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012 Mar;366(10):883–92. doi: https://doi.org/10.1056/NEJMoa1113205
16. Hu F, Xu J, Zhang B, Li C, Nie W, Gu P, et al. Efficacy of Local Consolidative Therapy for Oligometastatic Lung Adenocarcinoma Patients Harboring Epidermal Growth Factor Receptor Mutations. Clin Lung Cancer. 2019 Jan;20(1):e81–90. doi: https://doi.org/10.1016/j.cllc.2018.09.010
17. Fallet V, Matton L, Schernberg A, Canellas A, Cornelis FH, Cadranel J. Local ablative therapy in oncogenic-driven oligometastatic non-small cell lung cancer: present and ongoing strategies-a narrative review. Transl Lung Cancer Res. 2021 Jul;10(7):3457–72. doi: https://doi.org/10.21037/tlcr-20-1152
18. Griswold CR, Kerrigan K, Patel SB. Combination of Local Ablative Therapy and Continuation of Immune Checkpoint Inhibitor (ICI) Therapy Provides Durable Treatment Response Past Oligometastatic Progression in NSCLC: A Case Report. Case Rep Oncol. 2019 Nov;12(3):866–71. doi: https://doi.org/10.1159/000504473
19. Guisier F, Gervais R, Husseini KE, Assié JB, Geier M, Decroisette C, et al. Local ablative treatment and treatment beyond progression for oligo-progression in stage IV non-small cell lung cancer after tumour response to anti-PD1 treatment. Ann Oncol. 2019 Oct;30:v620–1. doi: https://doi.org/10.1093/annonc/mdz260.034