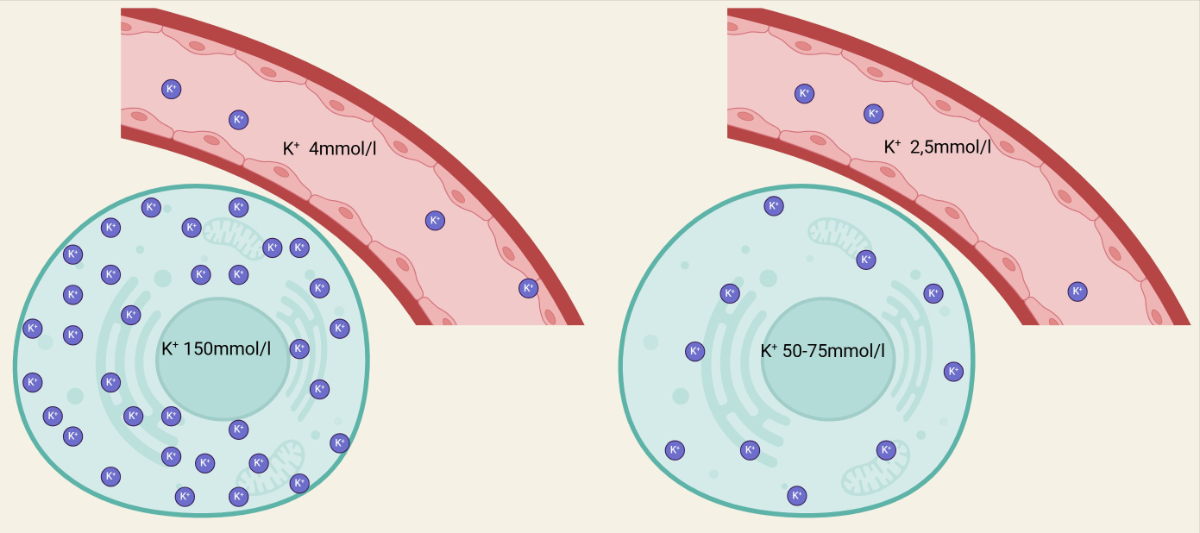
Figure 1Ratio of intra- and extracellular potassium in a physiological state (left) and in hypokalaemia (right) (Created in BioRender. Oswald S, 2025. https://BioRender.com/e47g425).
DOI: https://doi.org/https://doi.org/10.57187/4767
Electrolyte imbalances are frequently encountered among patients in the emergency department (ED), often resulting from acute illness or pharmacological interventions. These disturbances can substantially affect patient morbidity and mortality, highlighting the importance of prompt recognition, accurate diagnosis, and appropriate management [1, 2]. Notably, hypokalaemia has been shown to independently worsen outcomes even in otherwise low-risk ED populations, underscoring its clinical relevance regardless of baseline risk [1]. This article specifically addresses hypokalaemia in patients presenting to the ED.
Despite a general consensus on normal serum potassium levels (3.5 mmol/l to 4.5 mmol/l), a universal definition does not exist. Acute mild-to-moderate hypokalaemia is defined as serum potassium levels ranging from 2.5 mmol/l to 3.4 mmol/l in individuals with no history of low potassium levels [3, 4]. For research purposes, severe hypokalaemia is defined as a serum potassium level below 2.5 mmol/l [3, 4].
Several factors can lead to artificially elevated serum potassium levels. These include mechanical injury to blood samples, prolonged tourniquet use (lasting >1 minute), and fist clenching during blood collection [5]. Additional contributing factors include blood clot formation, issues during centrifugation, an elevated white blood cell count, and increased platelet levels [5-8]. Another potential factor in the variability of hypokalaemia is the collection of blood samples from sites of infusion with crystalloid or other intravenous solutions [5]. This issue may be particularly relevant in crowded EDs, where time constraints are common.
Hypokalaemia is a common electrolyte disorder frequently encountered in ED settings, affecting approximately 13–20% of patients presenting to primary care facilities [2, 9–11]. Notably, 33% of affected individuals experience recurrent episodes within one year [11]. Severe hypokalaemia affects a smaller proportion of patients, occurring in approximately 2.5% of those presenting to healthcare institutions [11].
The prevalence of severe hypokalaemia is higher in women than in men; it is thought to be attributable to factors such as lower muscle mass and a reduced capacity for potassium storage among women [3, 12]. However, age does not appear to influence the prevalence of hypokalaemia [3, 13, 14].
Diuretic use is one of the leading causes of hypokalaemia; approximately 80% of patients undergoing diuretic treatment develop this electrolyte imbalance [15, 16]. As a result, individuals with conditions associated with fluid overload, such as heart failure, as well as those with reduced renal function or hypertension, are frequently affected [15, 16]. These factors may explain the high prevalence of hypokalaemia in acute care settings such as EDs.
Potassium is a vital cation involved in numerous cellular processes, and its balance is crucial for maintaining proper physiological function. Most of the body's potassium is stored intracellularly, with only a small portion present in the extracellular space, including the blood [17]. Consequently, plasma or serum potassium levels do not accurately reflect the body’s total potassium content [17]. In the absence of stimuli that alter transcellular potassium shifts, a loss of 0.3 mmol/l in serum potassium corresponds to a total body deficit of approximately 100 mmol/l [18]. For example, a patient with a serum potassium concentration of 2.0 mmol/l may have a potassium deficit of 500 mmol/l [18]. During periods of inadequate potassium intake, such as diets lacking fruits and vegetables or during extreme fasting, intracellular potassium stores are mobilised to maintain extracellular potassium concentrations [18]. Although this compensatory mechanism prevents a rapid decline in serum potassium levels, it ultimately leads to a significant depletion of total body potassium stores over time [18].
Figure 1 presents a schematic depiction of the ratio of intra- and extracellular potassium in the physiological state (left) and in hypokalaemia (right).

Figure 1Ratio of intra- and extracellular potassium in a physiological state (left) and in hypokalaemia (right) (Created in BioRender. Oswald S, 2025. https://BioRender.com/e47g425).
Potassium homeostasis is tightly regulated, primarily by the kidneys and the endocrine system, to support various physiological functions [19]. Hypokalaemia resulting solely from insufficient intake can occur in cases of severe malnutrition; however, this is relatively uncommon because of the kidneys' ability to regulate potassium excretion. In response to low potassium levels, the juxtaglomerular cells of the kidneys secrete renin [20]. This process stimulates the production of angiotensin II while simultaneously suppressing aldosterone secretion [20]. This combination of elevated angiotensin II and reduced aldosterone levels promotes sodium chloride reabsorption (via SLC12A3 and pendrin) and reduces potassium secretion in the distal tubules [21]. This regulatory process reduces potassium elimination to a minimum of 10–30 mmol/day [22]. Unlike hyponatraemia, in which a healthy kidney can typically retain nearly all sodium from the primary urine, potassium is always excreted in small but consistent amounts [23].
Serum hypokalaemia can result from inadequate potassium intake, excessive potassium loss through the urinary or gastrointestinal systems, or a combination of these factors. Additionally, various mechanisms can shift potassium intracellularly, thereby reducing extracellular potassium levels.
As mentioned above, the most common cause of renal potassium loss is the use of diuretics [15]. Loop diuretics are frequently prescribed for conditions such as heart failure and renal insufficiency, which are commonly encountered in EDs [16]. Furthermore, thiazide and thiazide-like diuretics are strongly associated with the development of hypokalaemia, with both the specific drug and its dosage playing significant roles in determining the incidence of this electrolyte imbalance [24].
In addition to diuretics, various conditions can contribute to an excess of mineralocorticoids, resulting in hypokalaemia. These include primary hyperaldosteronism (due to an adrenal adenoma), Cushing's syndrome, renin-secreting tumours, and renovascular hypertension resulting from renal artery stenosis or vasculitis [25-28]. Beta-lactam antibiotics may cause hypokalaemia by acting as non-reabsorbable anions that promote renal potassium wasting. Across clinical studies, the incidence of β-lactam-associated hypokalaemia differs substantially between individual agents, underscoring a clinically relevant but highly variable effect (e.g. a 42% incidence of hypokalaemia in patients treated with flucloxacillin and a 14% incidence of hypokalaemia in patients treated with ceftriaxone) [29].
Acute kidney injury (AKI) is associated with the development of hypokalaemia in up to 11% of affected patients [30]. The occurrence of acute hypokalaemia is an independent risk factor for mortality, regardless of age, gender, or serum creatinine levels [30].
Certain genetic conditions can also lead to persistent renal potassium loss, including Gitelman syndrome, Liddle syndrome, Bartter syndrome, congenital adrenal hyperplasia, and the syndrome of apparent mineralocorticoid excess [23, 31]. Among these, Gitelman syndrome is the most common [31].
Hypomagnesaemia frequently occurs concomitantly with hypokalaemia [32]. In addition to magnesium being lost in a manner similar to potassium, hypomagnesaemia appears to contribute to potassium loss by promoting increased urinary potassium excretion [32]. Consequently, it is recommended to correct magnesium deficiency concurrently with potassium replacement in patients presenting with hypokalaemia [32-34].
Severe acute or chronic diarrhoea is the most common extrarenal cause of hypokalaemia (including cases of prolonged laxative abuse) [35]. Additionally, hypokalaemia may occur in individuals with prolonged vomiting, coeliac disease, or intestinal villous adenomas [36, 37].
Furthermore, potassium can be lost through the skin via sweating, particularly during intense and prolonged exercise, as observed in professional athletes and individuals with anorexia or exercise addiction [38]. Additionally, burns covering a large body surface area can result in rapid volume depletion and significant electrolyte imbalances, including severe hypokalaemia [39].
Several mechanisms facilitate the movement of potassium into the intracellular space. These mechanisms are essential for preventing hyperkalaemia after a potassium-rich meal. Some of these mechanisms are used in medical practice to temporarily lower serum potassium levels in cases of hyperkalaemia. However, these same mechanisms can inadvertently contribute to the development of hypokalaemia.
One mechanism involves insulin, a hormone that promotes the shift of potassium into the intracellular space [40]. An overdose of exogenous insulin often results in hypokalaemia [41]. Patients with diabetic ketoacidosis (DKA) are often expected to have hyperkalaemia on admission because of a transcellular potassium shift driven by acidosis [42]. However, studies have shown that up to 5.6% of patients with DKA exhibit hypokalaemia even before insulin therapy is initiated; therefore, checking serum potassium levels before treatment initiation is essential [43]. With fluid resuscitation and insulin treatment, these patients are at further risk of developing acute hypokalaemia because of the compounded intracellular shift of potassium [40].
Another mechanism that induces potassium movement into the intracellular space is β-adrenergic stimulation, which is commonly used in the treatment of obstructive pulmonary diseases, such as salbutamol inhalation [44].
Finally, metabolic alkalosis enhances intracellular potassium uptake, which can result in a substantial shift in potassium distribution [45, 46]. An increase in pH by 0.1 can reduce serum potassium levels by approximately 0.4 mmol/l [46].
Mild hypokalaemia is often asymptomatic [47]. The severity of hypokalaemia-related symptoms correlates with the rate and extent of the decrease in potassium levels; however, it is highly variable [48]. The most notable clinical manifestations of hypokalaemia stem from its impact on the cardiac and skeletal muscle, as well as the kidneys [48].
The primary symptoms that warrant the consideration of hypokalaemia in the differential diagnosis include muscle weakness, cramps, spasms, fatigue, constipation, and abdominal pain [19]. However, these symptoms are nonspecific and can be associated with a range of other conditions, making it challenging to directly link them to this particular electrolyte imbalance [47]. Because these symptoms vary and are often difficult to pinpoint during history-taking in time-sensitive settings such as EDs, hypokalaemia is frequently overlooked as a potential diagnosis.
A serious consequence of potassium imbalance is the potential development of cardiac arrhythmias [49]. Severe hypokalaemia or rapid declines in serum potassium levels can trigger potentially fatal cardiac arrhythmias [49]. Hypokalaemia reduces repolarisation reserve by impairing K+ conductance and inhibiting Na+-K+ ATPase activity, leading to an intracellular accumulation of Na+ and Ca2+. This ion buildup increases the risk of arrhythmias, including increased automaticity and the occurrence of early and delayed afterdepolarisations [49].
In syndromes associated with primary mineralocorticoid excess, such as the syndrome of apparent mineralocorticoid excess, Liddle syndrome, and Cushing's syndrome, hypokalaemia resulting from renal potassium wasting may present with difficult-to-control hypertension in younger patients [31, 50]. By contrast, hypokalaemia due to renal salt wasting, such as in Bartter syndrome or Gitelman syndrome, is often associated with hypotension [51].
In rare cases, severe hypokalaemia may trigger rhabdomyolysis through impaired muscle perfusion and myocyte injury, manifesting as muscle weakness, cramps, and elevated CPK [52].
A comprehensive diagnostic approach is essential to identify the underlying cause of hypokalaemia.
This evaluation includes a thorough clinical history, physical examination, and a range of laboratory investigations.
The clinical history should focus on identifying potential aetiologies, such as recent diuretic use, laxative use, or the presence of gastrointestinal losses (e.g. vomiting or diarrhoea). Additionally, symptoms of muscle weakness, arrhythmias, or syncope should prompt suspicion of hypokalaemia-induced complications. A history of endocrine disorders, renal disease, or medication use should also be explored, as these may contribute to the development of hypokalaemia.
The patient’s volume status should be assessed during the physical examination. Signs of heart failure or arrhythmias should be carefully evaluated. An abdominal examination may reveal reduced bowel sounds or tenderness. If the patient reports muscle weakness, a neurological examination may show either diminished or heightened reflexes, along with paraesthesia. In severe cases, laboured breathing may occur because of impaired chest expansion.
In cases of underlying systemic or genetic conditions, symptoms of hypokalaemia may present alongside features specific to the condition (e.g., abdominal striae, moon facies, or abdominal fat accumulation in Cushing’s disease) [53].
Blood pressure, heart rate, and oxygen saturation should be measured in all patients to assess haemodynamic stability.
The presence of hypertension is indicative of mineralocorticoid excess or related genetic disorders, such as Liddle syndrome or the syndrome of apparent mineralocorticoid excess [25–27, 31]. Gitelman syndrome, the most prevalent of these genetic disorders, is characterised by renal sodium and potassium wasting, hypomagnesaemia, hypocalciuria, and hypotension [31].
Because hypokalaemia can lead to potentially life-threatening arrhythmias, an ECG should be performed in patients suspected of having hypokalaemia. Typical ECG findings include a decreased T-wave amplitude, ST segment depression, a prolonged QT interval, and the appearance of U-waves (most prominent in the lateral precordial leads V4-V6) [54]. A decreased T-wave amplitude is often the earliest sign, and a U-wave is characteristic in cases of severe acute hypokalaemia [54]. Although the likelihood of ECG changes and arrhythmias increases as serum potassium levels decrease, these findings are not always reliable, as some patients with severe hypokalaemia may not show any ECG abnormalities [55]. Additionally, when ECG changes do occur, they may be nonspecific and not solely indicative of hypokalaemia [55]. In cases of severe hypokalaemia, particularly when ECG changes resulting from hypokalaemia are observed, continuous monitoring of vital signs and ECG is warranted.
Figure 2 shows an ECG with U-waves (indicated by circles) in a patient with severe hypokalaemia after prolonged diarrhoea.
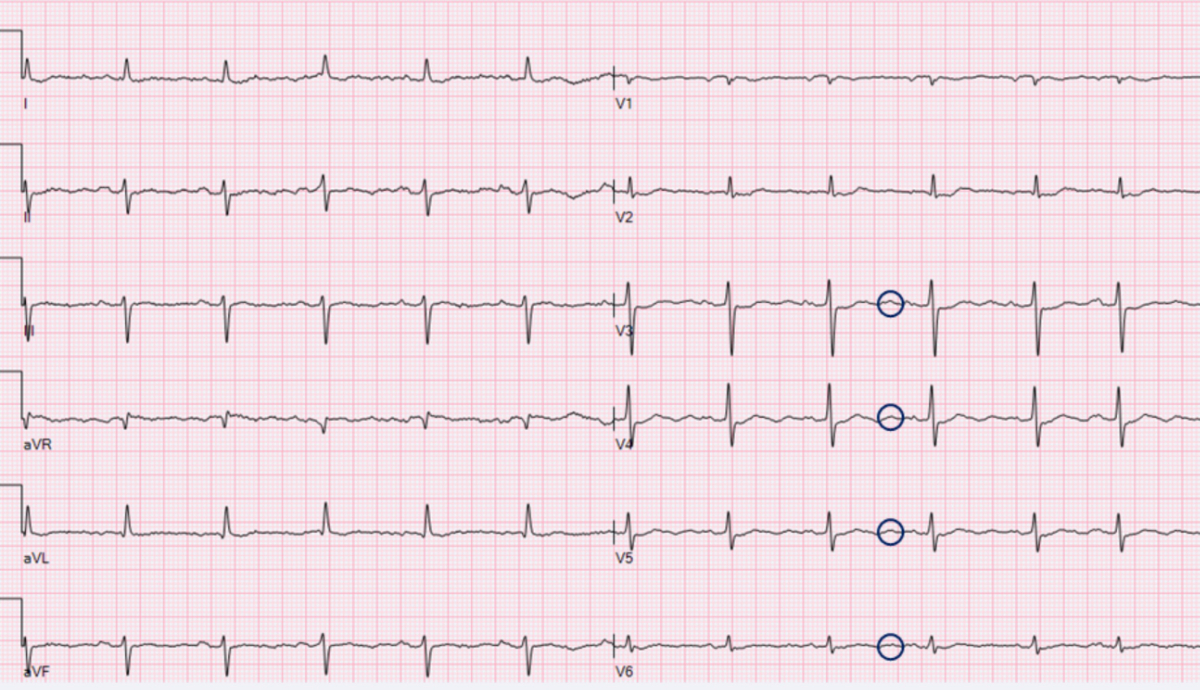
Figure 2ECG with U-waves (indicated by circles) in a patient with severe hypokalaemia after prolonged diarrhoea (ECG recorded at Kepler Universitätsklinikum Linz, 2025).
Spot urine analysis is a practical tool for narrowing the differential diagnosis of hypokalaemia, although its reliability can be affected by the patient's volume status [21, 56]. Although a 24-hour urine collection offers the most accurate assessment of renal potassium handling, its practicality is limited in the ED because of the need for a controlled environment and prolonged data collection. In cases of hypokalaemia with inappropriate kaliuresis (urinary potassium >40 mmol/24 h), renal potassium wasting is suggested; however, this requires extended monitoring, making it less feasible in urgent settings [56].
The urine potassium-to-serum creatinine ratio (UK/SCr) is an invaluable, fast test for distinguishing between renal and extrarenal causes of hypokalaemia [56]. This ratio is particularly useful in cases with an unclear history or an atypical presentation:
In addition to the UK/SCr ratio, the assessment of urinary sodium and chloride concentrations can provide further diagnostic insights [56]. Elevated urinary sodium levels in the context of potassium loss suggest a tubulopathy, whereas low urinary sodium levels typically indicate extrarenal potassium loss [56].
In the evaluation of hypokalaemia, a basic laboratory panel should be obtained, including measurements of serum sodium, potassium, chloride, magnesium, bicarbonate, glucose, liver enzymes, and renal function.
Additionally, arterial or venous blood gas analysis can provide valuable information about metabolic and respiratory acidosis or alkalosis, helping to further elucidate the underlying cause of hypokalaemia [21]. Metabolic acidosis is commonly observed in conditions such as renal tubular acidosis and certain types of secretory diarrhoea [21]. By contrast, metabolic alkalosis may be associated with the use of laxatives or diuretics, vomiting, chloride-secreting diarrhoea, or disorders such as Gitelman syndrome and Bartter syndrome [17].
A diagnostic algorithm (figure 3) is provided to assist ED clinicians in identifying the underlying cause of hypokalaemia based on the clinical presentation and results of the initial investigations. Using a structured approach, it is possible to differentiate between extrarenal and renal causes of potassium depletion, allowing for the prompt initiation of appropriate treatment.
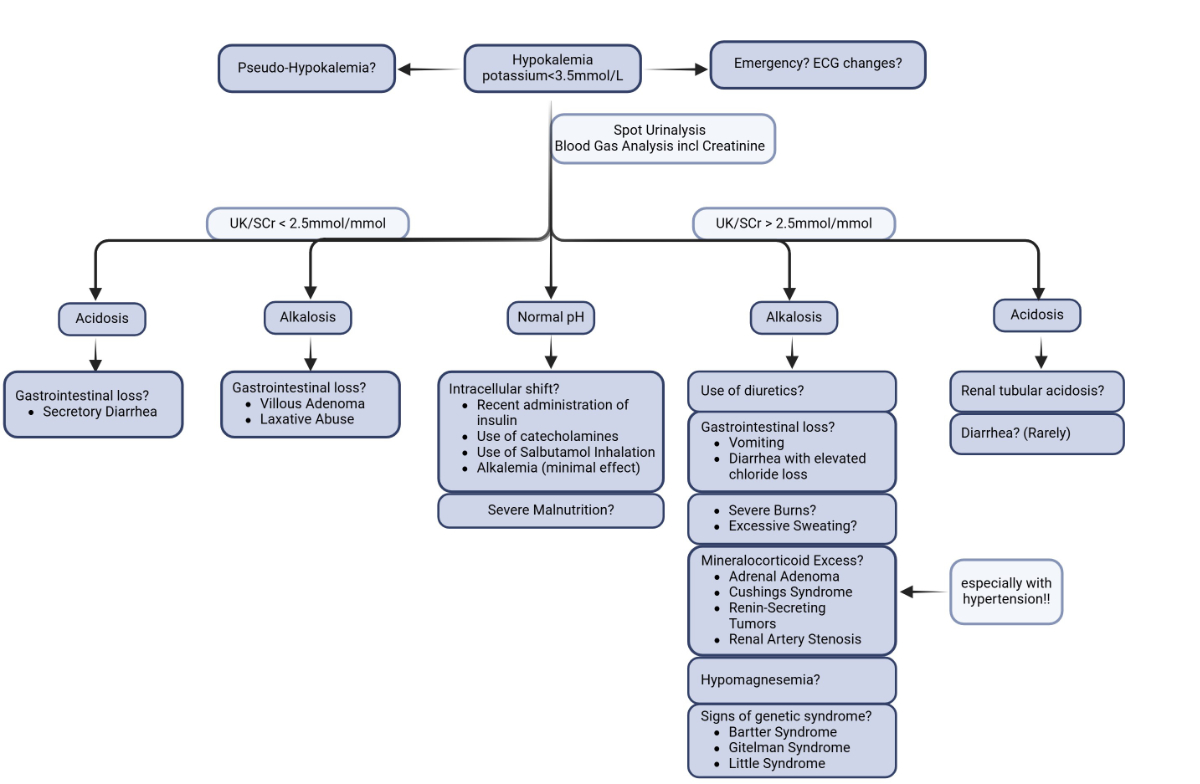
Figure 3A simple diagnostic algorithm to determine the cause of hypokalaemia for use in the emergency department (created in BioRender. Oswald S, 2025) https://BioRender.com/j66q296).
In the management of acute severe hypokalaemia, it is crucial to identify and address the underlying cause while simultaneously correcting potassium levels. For instance, diuretics should be discontinued or their dosage reduced, if possible. Additionally, laxative use should be halted, and any conditions contributing to prolonged diarrhoea or vomiting should be treated. In cases in which arrhythmias are observed on the electrocardiogram (ECG), or if there is a heightened cardiac risk, such as hypomagnesaemia, long QT syndrome, or recent myocardial infarction, continuous ECG monitoring is strongly recommended [57].
The route of potassium administration should be chosen based on the severity of hypokalaemia and the potential for adverse events. Given the potential delay in the increase in serum potassium levels as the body restores its potassium stores, careful monitoring of potassium levels is essential [58]. Transient hyperkalaemia may occur, particularly with intravenous potassium replacement [58]. However, potassium levels typically stabilise following cellular reabsorption. Therefore, it is advisable to target a potassium level within the high-normal range of 4–5 mmol/l [58].
Hypomagnesaemia, which occurs concurrently in up to 40% of patients with hypokalaemia, should always be treated in parallel [33, 59]. In the presence of cardiac arrhythmias, magnesium should be administered intravenously, typically at a rate of 8–16 mmol over 2–4 hours [32]. Although some studies have suggested administering magnesium alongside potassium regardless of magnesium levels, this approach does not show a significant benefit and may instead lead to an increased risk of hypermagnesaemia [60].
For mild-to-moderate hypokalaemia without significant complications, oral potassium supplementation is generally preferred [61]. Although rare, gastrointestinal mucosal irritation leading to bleeding or ulceration may occur [62]. As chloride loss often accompanies the most common causes of hypokalaemia, it is advisable to replace chloride in conjunction with potassium in a combined formulation [61]. A typical dosage of 100–150 mmol/day of potassium chloride is typically sufficient until normal potassium levels are achieved [61]. Oral potassium supplementation for hypokalaemia often includes slow-release tablets (≈10 mmol K⁺ per tablet) and effervescent tablets (≈30 mmol K⁺ per tablet). In patients with concomitant metabolic acidosis, such as renal tubular acidosis, alkalinising potassium formulations, such as potassium citrate or potassium bicarbonate, should be used [63]. In malnourished patients, the consumption of potassium-rich foods, such as bananas, artichokes, grapes, or pineapple, may also be beneficial [64].
In cases of severe hypokalaemia or when debilitating symptoms and ECG changes are present, intravenous potassium administration is required [65]. The four main types of potassium preparations are potassium chloride, potassium phosphate, potassium bicarbonate, and potassium citrate. Potassium phosphate is used specifically in hypophosphataemia, whereas potassium bicarbonate or citrate are used for metabolic acidosis [66, 67]. In most other cases, potassium chloride is the preferred solution. Potassium should always be diluted in saline, not dextrose, as the sugars in dextrose induce insulin secretion, which could cause an unwanted shift of potassium into cells and temporarily increase the risk of arrhythmias [40]. For potassium administration via peripheral veins, a maximum of 60 mmol is recommended in a 1000 ml non-dextrose solution, and a maximum of 10 mmol is recommended in a 100 or 200 ml non-dextrose solution [65, 68]. The recommended maximum rate of intravenous potassium administration is 10–20 mmol/h [65, 68]. Local pain and phlebitis may occur, particularly with a flow rate exceeding 10 mmol/h [65, 68]. However, in life-threatening situations, up to 40 mmol/h can be infused [65, 68]. The infusion rate should be reduced as soon as possible to minimise complications such as rebound hyperkalaemia [65]. Administering potassium via a central line rather than a peripheral line can reduce pain and may be advantageous when prolonged potassium substitution is necessary [65]. While administering intravenous potassium, serum potassium levels should be monitored every 1–2 hours until they stabilise within the range of 4–5 mmol/l [65]. Potassium levels should be rechecked 12 and 24 hours after the cessation of intravenous supplementation [65]. If hypokalaemia persists despite intravenous treatment, continuous renal or intestinal losses should be reassessed and addressed if possible.
Figure 4 presents a simple therapeutic approach to hypokalaemia, summarising the concepts mentioned above.
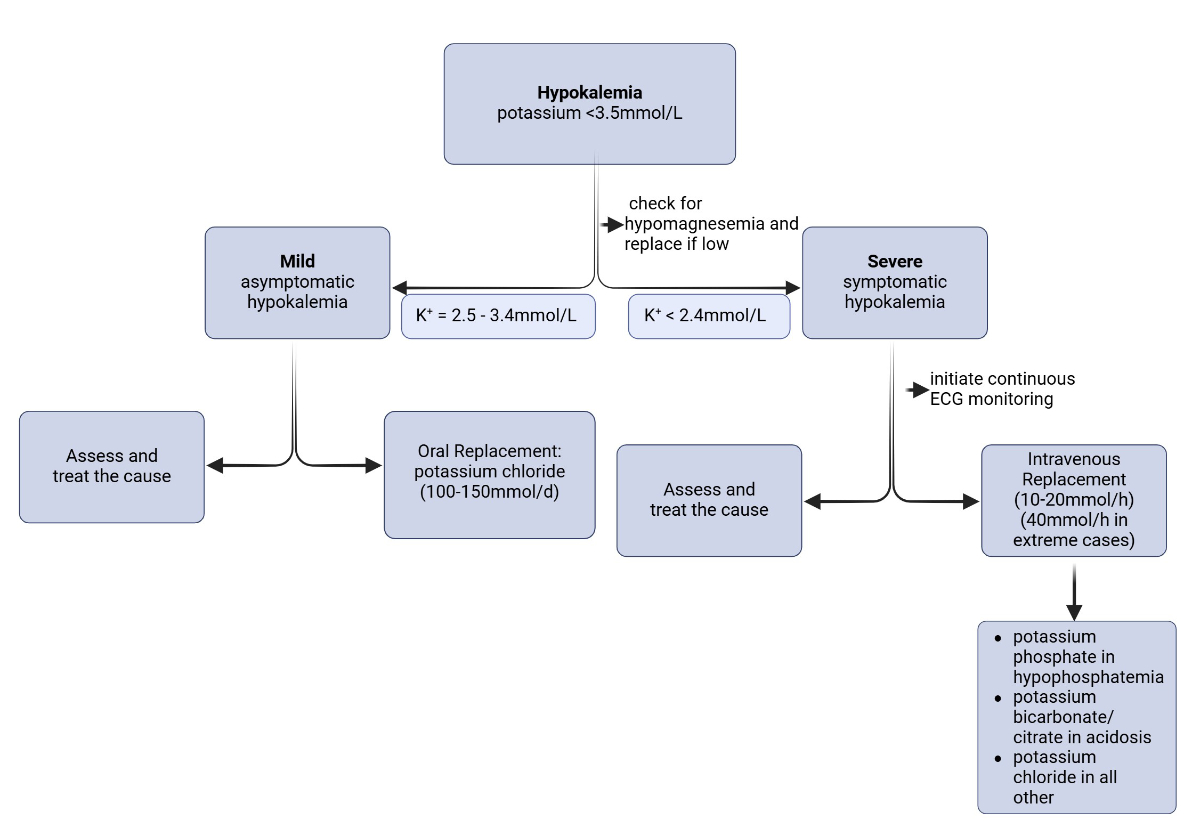
Figure 4Simple therapeutic approach to hypokalaemia (created in BioRender. Oswald S, 2025. https://BioRender.com/y77b088).
For chronic hypokalaemia, potassium-sparing diuretics, such as spironolactone, eplerenone, or amiloride, can be considered [69].
Table 1 presents a summary of important facts and key considerations for ED physicians.
Table 1Key messages for ED physicians regarding the diagnosis, workup, and treatment of hypokalaemia.
| When should hypokalaemia be considered? | Diuretic use |
| Severe vomiting and/or diarrhoea | |
| Malnutrition | |
| Primary hyperaldosteronism, renovascular hypertension | |
| Genetic conditions (Bartter, Liddle, or Gitelman syndrome) | |
| β-adrenergic activity | |
| Insulin administration | |
| Respiratory/metabolic alkalosis | |
| What symptoms are associated with hypokalaemia? | CAVE: often asymptomatic |
| Muscle weakness | |
| Cramps | |
| Fatigue | |
| Constipation | |
| Cardiac arrhythmias | |
| What diagnostic workup should be initiated in the ED? | Vital signs |
| ECG (ECG changes due to hypokalaemia?) | |
| Blood gas analysis (acid/base state?) | |
| Laboratory tests (sodium, potassium, renal function, and magnesium) | |
| Urinalysis (spot versus 24-hour collection) | |
| How should hypokalaemia be managed in the ED? | Oral replacement in mild, asymptomatic hypokalaemia without contraindication to oral potassium replacement (e.g. 100 mmol/day) |
| Intravenous replacement in symptomatic or severe hypokalaemia (e.g. 10 mmol/h; maximum 40 mmol/h) | |
| Concomitant administration of magnesium in cases of hypomagnesaemia | |
| Monitoring of vital signs and ECG in cases of severe, symptomatic hypokalaemia | |
| Other therapeutic measures? | Withhold provoking medications |
| Refer patients for further evaluation in cases of severe, recurrent hypokalaemia or suspicion of a genetic cause | |
| Increase intake of potassium-rich foods | |
| Admit or discharge? | Admit patients with severe or symptomatic hypokalaemia in need of intravenous supplementation and/or monitoring |
As emphasised throughout this article, it is crucial to address the underlying cause of acute hypokalaemia through targeted treatment. This includes a thorough review of the patient's current medications, with adjustments made where necessary. Additionally, further diagnostic investigations should be conducted to assess possible gastrointestinal issues. The potential causes of malnutrition or excessive laxative use should be explored, as these may require psychological intervention or lifestyle modifications.
The recurrence rate of hypokalaemia is notably high, at 33% [11]. Therefore, it is recommended that patients with concomitant risk factors be referred for regular laboratory monitoring and adjustment to their management plan as needed.
In conclusion, hypokalaemia is a common and potentially hazardous electrolyte imbalance observed in patients presenting to the ED. A comprehensive understanding of the underlying mechanisms, clinical manifestations, and management strategies is essential for ED physicians.
For this review, we conducted a comprehensive literature search primarily using PubMed, focusing on peer-reviewed articles relevant to the aetiology, diagnosis, and management of hypokalaemia in emergency care settings. Keywords included “hypokalaemia”, “emergency department”, “electrolyte imbalance”, “potassium replacement”, and related terms. We prioritised high-quality studies, clinical guidelines, and recent reviews published within the last 10–15 years. In addition, this article integrates clinical insights and management strategies previously established and applied by the supervising physician, ensuring that the review reflects both evidence-based knowledge and practical, real-world experience in emergency medicine.
This work received no external funding.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflict of interest related to the content of this manuscript was disclosed.
1. Abensur Vuillaume L, Ferreira JP, Asseray N, Trombert-Paviot B, Montassier E, Legrand M, et al. Hypokalemia is frequent and has prognostic implications in stable patients attending the emergency department. PLoS One. 2020 Aug;15(8):e0236934. doi: https://doi.org/10.1371/journal.pone.0236934
2. Singer AJ, Thode HC Jr, Peacock WF. A retrospective study of emergency department potassium disturbances: severity, treatment, and outcomes. Clin Exp Emerg Med. 2017 Jun;4(2):73–9. doi: https://doi.org/10.15441/ceem.16.194
3. Marti G, Schwarz C, Leichtle AB, Fiedler GM, Arampatzis S, Exadaktylos AK, et al. Etiology and symptoms of severe hypokalemia in emergency department patients. Eur J Emerg Med. 2014 Feb;21(1):46–51.
4. Kardalas E, Paschou SA, Anagnostis P, Muscogiuri G, Siasos G, Vryonidou A. Hypokalemia: a clinical update. Endocr Connect. 2018 Apr;7(4):R135–46. doi: https://doi.org/10.1530/EC-18-0109
5. Seimiya M, Yoshida T, Sawabe Y, Sogawa K, Umemura H, Matsushita K, et al. Reducing the incidence of pseudohyperkalemia by avoiding making a fist during phlebotomy: a quality improvement report. Am J Kidney Dis. 2010 Oct;56(4):686–92. doi: https://doi.org/10.1053/j.ajkd.2010.06.014
6. Alhaj Moustafa M, Malkovska V, Elmahdy S, Catlett J. A Challenging Case of Pseudohyperkalemia in Chronic Lymphocytic Leukemia. J Investig Med High Impact Case Rep. 2017 Dec;5(4):2324709617746194. doi: https://doi.org/10.1177/2324709617746194
7. Šálek T. Pseudohyperkalemia - Potassium released from cells due to clotting and centrifugation - a case report. Biochem Med (Zagreb). 2018 Feb;28(1):011002. doi: https://doi.org/10.11613/BM.2018.011002
8. Wiederkehr MR, Moe OW. Factitious hyperkalemia. Am J Kidney Dis. 2000 Nov;36(5):1049–53. doi: https://doi.org/10.1053/ajkd.2000.19084
9. Udensi UK, Tchounwou PB. Potassium Homeostasis, Oxidative Stress, and Human Disease. Int J Clin Exp Physiol. 2017;4(3):111–22. doi: https://doi.org/10.4103/ijcep.ijcep_43_17
10. Dhondup T, Qian Q. Acid-Base and Electrolyte Disorders in Patients with and without Chronic Kidney Disease: an Update. Kidney Dis (Basel). 2017 Dec;3(4):136–48. doi: https://doi.org/10.1159/000479968
11. Nilsson E, Gasparini A, Ärnlöv J, Xu H, Henriksson KM, Coresh J, et al. Incidence and determinants of hyperkalemia and hypokalemia in a large healthcare system. Int J Cardiol. 2017 Oct;245:277–84. doi: https://doi.org/10.1016/j.ijcard.2017.07.035
12. Adamczak M, Chudek J, Zejda J, Bartmańska M, Grodzicki T, Zdrojewski T, et al. Prevalence of hypokalemia in older persons: results from the PolSenior national survey. Eur Geriatr Med. 2021 Oct;12(5):981–7. doi: https://doi.org/10.1007/s41999-021-00484-6
13. Gilligan S, Raphael KL. Hyperkalemia and Hypokalemia in CKD: Prevalence, Risk Factors, and Clinical Outcomes. Adv Chronic Kidney Dis. 2017 Sep;24(5):315–8. doi: https://doi.org/10.1053/j.ackd.2017.06.004
14. Lindner G, Pfortmüller CA, Leichtle AB, Fiedler GM, Exadaktylos AK. Age-related variety in electrolyte levels and prevalence of dysnatremias and dyskalemias in patients presenting to the emergency department. Gerontology. 2014;60(5):420–3. doi: https://doi.org/10.1159/000360134
15. Alfonzo AV, Isles C, Geddes C, Deighan C. Potassium disorders—clinical spectrum and emergency management. Resuscitation. 2006 Jul;70(1):10–25. doi: https://doi.org/10.1016/j.resuscitation.2005.11.002
16. Arampatzis S, Funk GC, Leichtle AB, Fiedler GM, Schwarz C, Zimmermann H, et al. Impact of diuretic therapy-associated electrolyte disorders present on admission to the emergency department: a cross-sectional analysis. BMC Med. 2013 Mar;11(1):83. doi: https://doi.org/10.1186/1741-7015-11-83
17. Blanchard A. Pathophysiologic approach in genetic hypokalemia: an update. Ann Endocrinol (Paris). 2023 Apr;84(2):298–307. doi: https://doi.org/10.1016/j.ando.2022.11.005
18. Sterns RH, Cox M, Feig PU, Singer I. Internal potassium balance and the control of the plasma potassium concentration. Medicine (Baltimore). 1981 Sep;60(5):339–54. doi: https://doi.org/10.1097/00005792-198109000-00002
19. Castro D, Sharma S. Hypokalemia. StatPearls. Treasure Island (FL): StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC.; 2025.
20. Kamel KS, Schreiber M, Halperin ML. Renal potassium physiology: integration of the renal response to dietary potassium depletion. Kidney Int. 2018 Jan;93(1):41–53. doi: https://doi.org/10.1016/j.kint.2017.08.018
21. Palmer BF, Clegg DJ. Physiology and Pathophysiology of Potassium Homeostasis: core Curriculum 2019. Am J Kidney Dis. 2019 Nov;74(5):682–95. doi: https://doi.org/10.1053/j.ajkd.2019.03.427
22. Squires RD, Huth EJ. Experimental potassium depletion in normal human subjects. I. Relation of ionic intakes to the renal conservation of potassium. J Clin Invest. 1959 Jul;38(7):1134–48. doi: https://doi.org/10.1172/JCI103890
23. Lindner G, Schwarz C, Ravioli S. Pathophysiology, Evaluation, and Treatment of Hypokalemia. Nephrology Self-Assessment Program; 2024. doi: https://doi.org/10.58483/nsap.00252024
24. Ravioli S, Bahmad S, Funk GC, Schwarz C, Exadaktylos A, Lindner G. Risk of Electrolyte Disorders, Syncope, and Falls in Patients Taking Thiazide Diuretics: Results of a Cross-Sectional Study. Am J Med. 2021 Sep;134(9):1148–54. doi: https://doi.org/10.1016/j.amjmed.2021.04.007
25. Mulatero P, Stowasser M, Loh KC, Fardella CE, Gordon RD, Mosso L, et al. Increased diagnosis of primary aldosteronism, including surgically correctable forms, in centers from five continents. J Clin Endocrinol Metab. 2004 Mar;89(3):1045–50. doi: https://doi.org/10.1210/jc.2003-031337
26. Newell-Price J, Trainer P, Besser M, Grossman A. The diagnosis and differential diagnosis of Cushing’s syndrome and pseudo-Cushing’s states. Endocr Rev. 1998 Oct;19(5):647–72.
27. Corvol P, Pinet F, Plouin PF, Bruneval P, Menard J. Renin-secreting tumors. Endocrinol Metab Clin North Am. 1994 Jun;23(2):255–70. doi: https://doi.org/10.1016/S0889-8529(18)30096-3
28. Rajgopal R, Khan A, Syed AA. Renal artery stenosis presenting as hypertension with hypokalemia. CMAJ. 2022 Sep;194(36):E1248–9. doi: https://doi.org/10.1503/cmaj.220091
29. Jansen MN, Safi W, Matyukhin I, Stasche F, Tennigkeit J, Ritter O, et al. Beta-lactam-associated hypokalemia. J Int Med Res. 2024 Aug;52(8):3000605241253447. doi: https://doi.org/10.1177/03000605241253447
30. Ravioli S, Pluess E, Funk GC, Walter P, Schwarz C, Exadaktylos AK, et al. Dyskalemias in patients with acute kidney injury presenting to the emergency department are common and independent predictors of adverse outcome. Int J Clin Pract. 2021 Jan;75(1):e13653. doi: https://doi.org/10.1111/ijcp.13653
31. Cruz DN, Perazella MA. Hypertension and hypokalemia: unusual syndromes. Conn Med. 1997 Feb;61(2):67–75.
32. Huang CL, Kuo E. Mechanism of hypokalemia in magnesium deficiency. J Am Soc Nephrol. 2007 Oct;18(10):2649–52. doi: https://doi.org/10.1681/ASN.2007070792
33. Agus ZS. Hypomagnesemia. J Am Soc Nephrol. 1999 Jul;10(7):1616–22. doi: https://doi.org/10.1681/ASN.V1071616
34. Van Laecke S. Hypomagnesemia and hypermagnesemia. Acta Clin Belg. 2019 Feb;74(1):41–7. doi: https://doi.org/10.1080/17843286.2018.1516173
35. Agarwal R, Afzalpurkar R, Fordtran JS. Pathophysiology of potassium absorption and secretion by the human intestine. Gastroenterology. 1994 Aug;107(2):548–71. doi: https://doi.org/10.1016/0016-5085(94)90184-8
36. Orchard MR, Hooper J, Wright JA, McCarthy K. A systematic review of McKittrick-Wheelock syndrome. Ann R Coll Surg Engl. 2018 Oct;100(8):1–7. doi: https://doi.org/10.1308/rcsann.2018.0184
37. Wolf I, Mouallem M, Farfel Z. Adult celiac disease presented with celiac crisis: severe diarrhea, hypokalemia, and acidosis. J Clin Gastroenterol. 2000 Apr;30(3):324–6. doi: https://doi.org/10.1097/00004836-200004000-00026
38. Sato K, Feibleman C, Dobson RL. The electrolyte composition of pharmacologically and thermally stimulated sweat: a comparative study. J Invest Dermatol. 1970 Dec;55(6):433–8. doi: https://doi.org/10.1111/1523-1747.ep12260581
39. Unwin RJ, Luft FC, Shirley DG. Pathophysiology and management of hypokalemia: a clinical perspective. Nat Rev Nephrol. 2011 Feb;7(2):75–84. doi: https://doi.org/10.1038/nrneph.2010.175
40. Cohen P, Barzilai N, Lerman A, Harel H, Szylman P, Karnieli E. Insulin effects on glucose and potassium metabolism in vivo: evidence for selective insulin resistance in humans. J Clin Endocrinol Metab. 1991 Sep;73(3):564–8. doi: https://doi.org/10.1210/jcem-73-3-564
41. Johansen NJ, Christensen MB. A Systematic Review on Insulin Overdose Cases: Clinical Course, Complications and Treatment Options. Basic Clin Pharmacol Toxicol. 2018 Jun;122(6):650–9. doi: https://doi.org/10.1111/bcpt.12957
42. Yamada H, Funazaki S, Kakei M, Hara K, Ishikawa SE. Diabetic ketoacidosis producing extreme hyperkalemia in a patient with type 1 diabetes on hemodialysis. Endocrinol Diabetes Metab Case Rep. 2017;2017. doi: https://doi.org/10.1530/EDM-17-0068
43. Arora S, Cheng D, Wyler B, Menchine M. Prevalence of hypokalemia in ED patients with diabetic ketoacidosis. Am J Emerg Med. 2012 Mar;30(3):481–4. doi: https://doi.org/10.1016/j.ajem.2011.01.002
44. Hung CH, Hua YM, Lee MY, Tsai YG, Yang KD. Evaluation of different nebulized bronchodilators on clinical efficacy and hypokalemia in asthmatic children. Acta Paediatr Taiwan. 2001;42(5):287–90.
45. Rizvi K, Abraham S, Gong J, Nogar J. Severe Alkalemia and hypokalemia after chronic exposure to alkaline water. Am J Emerg Med. 2024;76:273.e5-.e7. doi: https://doi.org/10.1016/j.ajem.2023.11.039
46. Adrogué HJ, Madias NE. Changes in plasma potassium concentration during acute acid-base disturbances. Am J Med. 1981 Sep;71(3):456–67. doi: https://doi.org/10.1016/0002-9343(81)90182-0
47. Lim S. Approach to hypokalemia. Acta Med Indones. 2007;39(1):56–64.
48. Phillips SL, Polzin DJ. Clinical disorders of potassium homeostasis. Hyperkalemia and hypokalemia. Vet Clin North Am Small Anim Pract. 1998 May;28(3):545–64. doi: https://doi.org/10.1016/S0195-5616(98)50055-1
49. Weiss JN, Qu Z, Shivkumar K. Electrophysiology of Hypokalemia and Hyperkalemia. Circ Arrhythm Electrophysiol. 2017 Mar;10(3):e004667. doi: https://doi.org/10.1161/CIRCEP.116.004667
50. Churilla T, Baker JB, Chang A, Finer G. A 16-Year-Old With a Hypertension Emergency: A Quiz. Am J Kidney Dis. 2025 Oct;86(4):A13–6. doi: https://doi.org/10.1053/j.ajkd.2025.05.007
51. Peters M, Jeck N, Reinalter S, Leonhardt A, Tönshoff B, Klaus G G, et al. Clinical presentation of genetically defined patients with hypokalemic salt-losing tubulopathies. Am J Med. 2002 Feb;112(3):183–90. doi: https://doi.org/10.1016/S0002-9343(01)01086-5
52. Pecnik P, Müller P, Vrabel S, Windpessl M. Two cases of hypokalaemic rhabdomyolysis: same but different. BMJ Case Rep. 2018;2018. doi: https://doi.org/10.1136/bcr-2017-223609
53. Savas M, Mehta S, Agrawal N, van Rossum EF, Feelders RA. Approach to the Patient: Diagnosis of Cushing Syndrome. J Clin Endocrinol Metab. 2022 Nov;107(11):3162–74. doi: https://doi.org/10.1210/clinem/dgac492
54. Diercks DB, Shumaik GM, Harrigan RA, Brady WJ, Chan TC. Electrocardiographic manifestations: electrolyte abnormalities. J Emerg Med. 2004 Aug;27(2):153–60. doi: https://doi.org/10.1016/j.jemermed.2004.04.006
55. Montague BT, Ouellette JR, Buller GK. Retrospective review of the frequency of ECG changes in hyperkalemia. Clin J Am Soc Nephrol. 2008 Mar;3(2):324–30. doi: https://doi.org/10.2215/CJN.04611007
56. Li J, Ma H, Lei Y, Wan Q. Diagnostic value of parameters from a spot urine sample for renal potassium loss in hypokalemia. Clin Chim Acta. 2020 Dec;511:221–6. doi: https://doi.org/10.1016/j.cca.2020.10.024
57. Drew BJ, Califf RM, Funk M, Kaufman ES, Krucoff MW, Laks MM, et al.; American Heart Association; Councils on Cardiovascular Nursing, Clinical Cardiology, and Cardiovascular Disease in the Young. Practice standards for electrocardiographic monitoring in hospital settings: an American Heart Association scientific statement from the Councils on Cardiovascular Nursing, Clinical Cardiology, and Cardiovascular Disease in the Young: endorsed by the International Society of Computerized Electrocardiology and the American Association of Critical-Care Nurses. Circulation. 2004 Oct;110(17):2721–46. doi: https://doi.org/10.1161/01.CIR.0000145144.56673.59
58. Kovesdy CP, Matsushita K, Sang Y, Brunskill NJ, Carrero JJ, Chodick G, et al.; CKD Prognosis Consortium. Serum potassium and adverse outcomes across the range of kidney function: a CKD Prognosis Consortium meta-analysis. Eur Heart J. 2018 May;39(17):1535–42. doi: https://doi.org/10.1093/eurheartj/ehy100
59. Whang R, Flink EB, Dyckner T, Wester PO, Aikawa JK, Ryan MP. Magnesium depletion as a cause of refractory potassium repletion. Arch Intern Med. 1985 Sep;145(9):1686–9. doi: https://doi.org/10.1001/archinte.1985.00360090162024
60. Tuttle A, Fitter S, Hua H, Moussavi K. The Effects of Magnesium Coadminstration During Treatment of Hypokalemia in the Emergency Department. J Emerg Med. 2022 Sep;63(3):399–413. doi: https://doi.org/10.1016/j.jemermed.2022.06.007
61. Cohn JN, Kowey PR, Whelton PK, Prisant LM. New guidelines for potassium replacement in clinical practice: a contemporary review by the National Council on Potassium in Clinical Practice. Arch Intern Med. 2000 Sep;160(16):2429–36. doi: https://doi.org/10.1001/archinte.160.16.2429
62. Gueta I, Markovits N, Halkin H, Loebstein R. Concomitant oral potassium chloride and anticholinergic therapy is associated with upper gastrointestinal bleeding: A cohort study. Br J Clin Pharmacol. 2021 Apr;87(4):2064–9. doi: https://doi.org/10.1111/bcp.14616
63. Boyer O, Ould Rabah M, Preka E. Recent Developments in the Treatment of Pediatric Distal Renal Tubular Acidosis. Paediatr Drugs. 2024 Nov;26(6):649–57. doi: https://doi.org/10.1007/s40272-024-00651-9
64. Samadian F, Dalili N, Jamalian A. Lifestyle Modifications to Prevent and Control Hypertension. Iran J Kidney Dis. 2016 Sep;10(5):237–63.
65. Hamill RJ, Robinson LM, Wexler HR, Moote C. Efficacy and safety of potassium infusion therapy in hypokalemic critically ill patients. Crit Care Med. 1991 May;19(5):694–9. doi: https://doi.org/10.1097/00003246-199105000-00016
66. Kraft MD, Btaiche IF, Sacks GS, Kudsk KA. Treatment of electrolyte disorders in adult patients in the intensive care unit. Am J Health Syst Pharm. 2005 Aug;62(16):1663–82. doi: https://doi.org/10.2146/ajhp040300
67. Hunter RW, Bailey MA. Horses for Courses: What is the Best Oral Potassium Supplementation Strategy? Clin J Am Soc Nephrol. 18. United States2023. p. 1250-3.
68. Kruse JA, Carlson RW. Rapid correction of hypokalemia using concentrated intravenous potassium chloride infusions. Arch Intern Med. 1990 Mar;150(3):613–7. doi: https://doi.org/10.1001/archinte.1990.00390150101019
69. Horisberger JD, Giebisch G. Potassium-sparing diuretics. Ren Physiol. 1987;10(3-4):198–220.