Efficacy and safety of bulevirtide in patients with chronic hepatitis D treated under
early access in Switzerland: a retrospective analysis
DOI: https://doi.org/https://doi.org/10.57187/4736
Mirjam Kolevab*,
Salomé Wolfac*,
Elizabeth Petràšac,
Véronique Erardd,
Beat Helblinge,
Beat Müllhauptf,
David Semelag,
Felix Stickelh,
Nasser Semmoa
a Department of Visceral Surgery and Medicine, Inselspital, Bern University
Hospital, Switzerland
b Graduate
School for Health Sciences, University of Bern, Switzerland
c University of Bern, Switzerland
d Department
of Infectiology and Internal Medicine, Kantonsspital Fribourg, Switzerland
e Gastroenterologie
Bethanien, Zurich, Switzerland
f Department
of Gastroenterology and Hepatology, University Hospital Zurich and University
of Zurich, Switzerland
g HOCH Health Ostschweiz, Kantonsspital St.Gallen, Universitäres Lehr- und
Forschungsspital, Klinik für Gastroenterologie und Hepatologie, St.Gallen,
Switzerland
h University Hospital of Zürich, Zurich, Switzerland
* Equal contribution as first authors
Summary
BACKGROUND AND AIMS: Bulevirtide 2 mg/day was approved in
Switzerland in February 2025 for the treatment of chronic hepatitis D virus (HDV)
infection. We present real-world data on efficacy and safety in patients
treated under an early access programme.
METHODS: This retrospective, multicentre Swiss cohort study included patients
with compensated HDV-associated cirrhosis in whom bulevirtide therapy (2
mg/day) was initiated between January 2020 and August 2024 under a
compassionate use programme. Virological response was defined as a HDV RNA level
that was undetectable or declined ≥2 log10 IU/ml from baseline. Biochemical
response was defined as normalisation of ALT. Combined response was defined as
achieving both virological and biochemical response. Liver-related events and
adverse events were assessed.
RESULTS: Fourteen patients with compensated HDV-related cirrhosis
received bulevirtide for a median duration of 1.85 years (1.1–2.1). Median age
was 51.3 years (43.9–58.5), and 71.4% were men. Baseline ALT was 81 U/l (55.8–88.8),
platelet count 102.5 × 109/l
(67.3–141.3) and liver stiffness 15.3 kPa (11.8–22.1). Baseline HDV RNA was
4.82 log10 IU/ml (4.52–6.23). Biochemical, virological and combined
responses were observed in 50%, 64.3% and 35.7% at 6 months; 66.7%, 75% and
58.3% at 12 months; and 62.5% for all three response types at 24 months. Two
patients (14.3%) developed de novo hepatocellular carcinoma, and one (7.14%) patient
underwent liver transplantation. No serious adverse events were reported. Mild
transient pruritus occurred in two (14.3%) patients.
CONCLUSIONS: In this real-world cohort of patients with compensated HDV
cirrhosis, bulevirtide demonstrated favourable efficacy and safety. These
findings support the integration of bulevirtide into routine care for patients
with HDV and compensated cirrhosis in Switzerland following its reimbursement
status as of 2025. Longer-term follow-up is warranted to assess the impact on
liver-related outcomes.
Abbreviations
- ALT
-
alanine
aminotransferase
- ANA
-
anti-nuclear antibodies
- EMA
-
European Medicines Agency
- HBV
-
hepatitis B
virus
- HCV
-
hepatitis C
virus
- HDV
-
hepatitis D
virus
- HIV
-
human
immunodeficiency virus
- IFN
-
interferon
- INR
-
international
normalised ratio
Introduction
Hepatitis D virus (HDV) is a single-stranded ribonucleic acid (RNA) defective virus
that requires the hepatitis B surface antigen (HBsAg) to enter hepatocytes, complete
its life cycle and cause liver damage [1]. It is estimated to affect approximately
12–20 million people worldwide, corresponding to 4.5–13% of all HBsAg-positive carriers
[2, 3]. Acute infection can cause acute liver failure, while persistent infection
typically causes the most severe form of chronic viral hepatitis, which is associated
with rapid and frequent progression to cirrhosis and its end-stage complications,
hepatic decompensation and hepatocellular carcinoma [1]. Until recently, no effective
antiviral therapy was available. In past decades, interferon-α (IFN-α)-based treatment
has been used to treat HDV, which was effective in only 25–30% of patients [4]. Apart
from low efficacy rates, IFN-α-associated side effects and contraindications resulted
in limited IFN-α treatment applicability, especially in advanced liver disease [1].
More recently, the HDV entry inhibitor bulevirtide, a linear 47-amino-acid chemically
synthesised lipopeptide that specifically binds to the HBV/HDV entry receptor Na+-taurocholate-co-transporting
polypeptide (NTCP), was developed, thus preventing hepatocyte infection. In July 2020,
bulevirtide 2 mg received conditional marketing authorisation by the European Medicines
Agency (EMA) for the treatment of chronic hepatitis D virus infection (CHD), after
positive results regarding safety and efficacy in phase II and III trials [5–8]. Recently
published data on real-world effectiveness and safety of bulevirtide monotherapy for
up to 96 weeks in patients with HDV-related cirrhosis, from the SAVE-D study, confirmed
the positive results from the registration trials [9].
In Switzerland, bulevirtide was accessible through a
compassionate use programme before official approval in February 2025. This
study presents real-world data from Switzerland on the efficacy and safety of bulevirtide
in patients with compensated HDV-associated cirrhosis included in this programme.
Patients and methods
This was a multicentre retrospective cohort study conducted in five centres across
Switzerland. Consecutive patients with compensated HDV-related cirrhosis starting
bulevirtide 2 mg/day between January 2020 and August 2024 with a follow-up of at least
six months were included. Follow-up was calculated from baseline to the date of liver-related
event, death or last follow-up. Chronic hepatitis D infection was defined as HDV RNA
positivity for at least 6 months. Data was collected at the start of treatment, after
six months and yearly thereafter. The compassionate use programme was only available
for patients with compensated liver cirrhosis. Diagnosis of cirrhosis was based on
liver biopsy, non-invasive tests such as liver stiffness measurement (LSM) >12.5 kPa
[1, 10], imaging findings (nodular liver, splenomegaly) or laboratory parameters (thrombocytopenia).
All patients had detectable HDV RNA and HBsAg positivity. The primary outcome was
the rate of virological and/or biochemical response. Virological response was defined
as an HDV RNA level that was undetectable or declined ≥2 log10 IU/ml from baseline.
Biochemical response was defined as normalisation of alanine aminotransferase (ALT).
Secondary outcomes were the occurrence of side effects, liver-related outcomes and
assessment of change in liver stiffness measurement during the observation period.
Data on clinical (including adverse events), biochemical, virological and imaging
parameters were retrospectively collected from electronic patient records. The present
manuscript was drafted in accordance with STROBE cohort study reporting guidelines.
Treatment regimen
Bulevirtide was administered at a dose of 2 mg/day
subcutaneously. In some cases, bulevirtide was combined with pegylated
interferon-α, based on
physician discretion and patient-specific factors. Additionally, all patients were
receiving a nucleos(t)ide analogue as part of standard therapy for chronic
hepatitis B. The first patient was initiated on bulevirtide in January 2020.
Subsequently, additional patients were enrolled and treated under the
compassionate use programme. This programme remained active until 31 January 2025.
As of 1 February 2025, bulevirtide has become reimbursable through the Swiss
health insurance system and is now available as part of standard clinical care.
All patients continued therapy under this reimbursement framework. Duration of bulevirtide
therapy and any treatment modifications were documented. Adherence was
monitored through regular clinical follow-up.
Statistical analysis
Categorical variables are expressed as counts and
percentages; continuous variables are expressed as medians and interquartile
ranges (IQR, 25th–75th
percentile). Fisher’s exact test was used to compare categorical data between
two groups. The Wilcoxon rank-sum test was used to compare quantitative data
between two groups. p-values <0.05 (two-tailed) were considered significant
in all analyses. Statistical analysis was performed using R version 4.2.
Ethical approval
The study was approved by the Cantonal Ethics Committee of
Bern, Switzerland (2024-01399). Written informed consent was given by all
patients.
Results
Study population and baseline characteristics
A total of 14 patients were enrolled with a median follow-up of 1.85 years (1.1–2.1).
Patients were recruited from Bern (n = 8, 57%), St Gallen (n = 2, 14%), Zurich (n
= 2, 14%), Fribourg (n = 1, 7%) and a private practice in Zurich (n = 1, 7%). Baseline
demographic, virological and clinical characteristics are summarised in table 1. The
presumed route of HDV transmission was intravenous drug use in 5 patients (36%), perinatal
in 1 (7%) and unknown in 7 (50%). One additional patient (7%) had either a history
of drug use or transfusion. All patients with a history of intravenous drug use were
of Swiss origin. Half of the patients were of Swiss origin (n = 7, 50%), with others
originating from Mongolia (n = 3, 21%), Turkey (n = 2, 14%) and Africa (n = 2, 14%).
All patients had compensated liver cirrhosis (Child-Pugh A)
at baseline. Five patients (36%) had endoscopically confirmed oesophageal
varices; two (14%) received non-selective beta-blockers, one (7%) underwent
ligation and one (7%) had beta-blocker intolerance. One patient (7%) had a history
of treated hepatocellular carcinoma prior to bulevirtide initiation. Comorbidities
included arterial hypertension (n = 3), diabetes mellitus (n = 1), chronic
kidney disease (n = 1), chronic pancreatitis (n = 1), coronary heart disease (n
= 1) and previous cancer (n = 2; cervical cancer and non-invasive bladder
cancer).
Two patients (14%) reported current low-level alcohol intake
(≤5 g/week), while three (21%) had a history of past alcohol use. Four patients
(29%) reported active nicotine use. No patient reported ongoing illicit drug
use at the time of treatment initiation.
Table 1Patient baseline characteristics at treatment start with
bulevirtide. Categorical variables are presented as n (%). Continuous data
are presented as median and interquartile range (IQR, 25th–75th percentile).
| Baseline variable |
Overall (n = 14) |
Missing data, n (%) |
| Demographics |
Age at treatment start, years |
51.3 (43.9–58.5) |
0 (0%) |
| Age at diagnosis of HBV, years |
33.3 (31.7–40.1) |
1 (7%) |
| Age at diagnosis of HDV, years |
37.1 (33–45) |
1 (7%) |
| Male sex, n (%) |
10 (71%) |
0 (0%) |
| Caucasian, n (%) |
7 (50%) |
0 (0%) |
| Treated HCV coinfection, n (%) |
4 (29%) |
0 (0%) |
| Treated HIV coinfection, n (%) |
1 (7%) |
0 (0%) |
| MASLD, n (%) |
2 (14%) |
0 (0%) |
| Body mass index, kg/m2 |
23.6 (21.8–26.5) |
0 (0%) |
| Biochemistry |
ALT, × ULN |
1.65 (1.5–1.98) |
0 (0%) |
| AST, × ULN |
1.5 (1.1–1.73) |
0 (0%) |
| Alkaline phosphatase, × ULN |
0.8 (0.7–0.95) |
1 (7%) |
| Bilirubin total, µmol/l
(REF: <17 µmol/l) |
8.9 (7.25–12.5) |
0 (0%) |
| Albumin, g/l
(REF: 35–52 g/l) |
34.5 (33.15–38.2) |
0 (0%) |
| INR
(REF: <1.2) |
1.1 (1.02–1.2) |
0 (0%) |
| Creatinine, μmol/l
(REF: 59–104 μmol/l) |
75 (66–82) |
1 (7%) |
| Platelets, 109/l
(REF: 150–450 109/l) |
102.5 (67.3–141.3) |
0 (0%) |
| Bile acids, μmol/l
(REF: 0–10 μmol/l) |
10.8 (9.9–13.2) |
7 (50%) |
| Virology |
HBsAg, IU/ml |
7469 (2373–14,287) |
2 (14%) |
| HBsAg-positive, n (%) |
1 (7%) |
4 (29%) |
| HBV DNA detectable, n (%) |
9 (64%) |
0 (0%) |
| HBV DNA, IU/ml |
16 (14–23) |
0 (0%) |
| HDV RNA, IU/ml |
66,718 (33,250–1,714,924) |
0 (0%) |
| HDV RNA, log10 IU/ml |
4.82 (4.52–6.23) |
0 (0%) |
| Assessment of liver disease |
Child A, n (%) |
14 (100%) |
0 (0%) |
| MELD score |
8 (7–8) |
0 (0%) |
| METAVIR F3–F4, n (%) |
9 (69%) |
1 (7%) |
| Liver stiffness measurement, kPa |
15.3 (11.8–22.1) |
1 (7%) |
| Cirrhotic aspect on imaging, n (%) |
8 (57%) |
0 (0%) |
| Spleen diameter, cm |
11.2 (10–14) |
1 (7%) |
| Oesophageal varices, n (%) |
5 (36%) |
5 (36%) |
| Clinical events |
History of hepatocellular carcinoma, n (%) |
1 (7%) |
0 (0%) |
| Treatment |
Previous IFN treatment, n (%) |
8 (57%) |
0 (0%) |
| Concurrent IFN treatment*, n (%) |
1 (7%) |
0 (0%) |
| NUC treatment for HBV, n (%) |
14 (100%) |
0 (0%) |
| Duration of NUC treatment before bulevirtide start, years |
3.24 (0.44–5.05) |
1 (7%) |
Virological and biochemical response
Treatment with bulevirtide resulted in a significant
reduction in HDV RNA levels over time. Median HDV RNA declined from 4.82 (4.52–6.2)
log10 IU/ml at baseline to 3.45 at month 6 (2.0–4.14, p <0.001), 2.63
at month 12 (2.0–3.57, p <0.001) and 2.28 at month 24 (2.0–3.48, p = 0.016).
Individual responses are shown in figure 1. In parallel, median ALT levels
decreased from 81 U/l (55.8–88.8) at baseline to 46 U/l (33–58.3) at 6 months (p
= 0.006), and remained within the normal range thereafter. Biochemical and virological
response rates over time are summarised in figure 2.
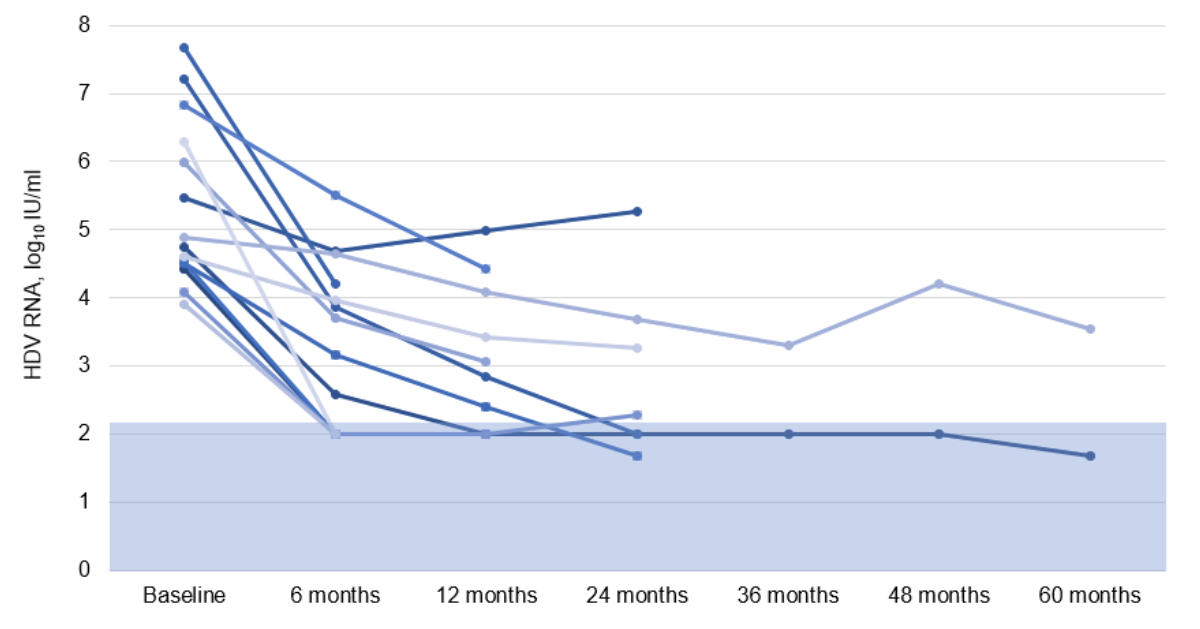
Figure 1HDV RNA levels during bulevirtide 2 mg/day
therapy up to 60 months. The apparent decline in patient numbers over
the 60-month observation period does not reflect treatment discontinuation or
loss to follow-up. Rather, it results from staggered treatment initiation
across the cohort, with only two patients having initiated therapy as early as
2020. All patients – except one with former intravenous drug use and the
transplanted patient – remained on treatment and under observation until the
end of the study period. This temporal context should be considered when
interpreting longitudinal response data. The blue area indicates that the viral
load is below the test’s lower limit of quantification. HDV RNA: hepatitis D
RNA.
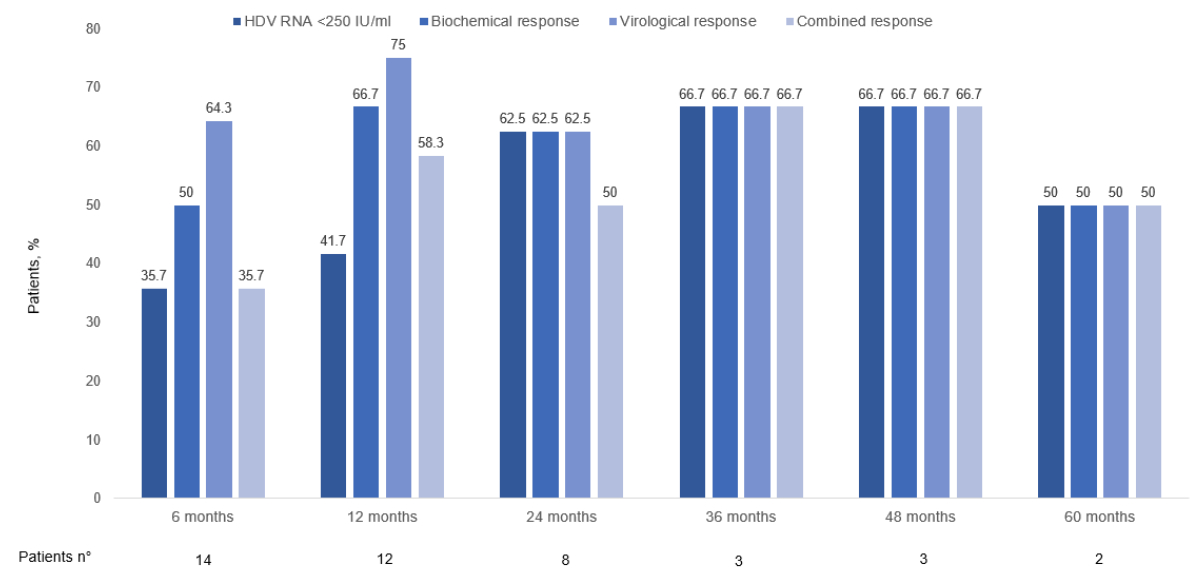
Figure 2Rates of biochemical, virological and
combined responses to bulevirtide treatment for up to 60 months.The apparent decline
in patient numbers
over the 60-month observation period does not reflect treatment discontinuation
or loss to follow-up. Rather, it results from staggered treatment initiation
across the cohort, with only two patients having initiated therapy as early as
2020. All patients – except one with former intravenous drug use and the
transplanted patient – remained on treatment and under observation until the
end of the study period. This temporal context should be considered when
interpreting longitudinal response data.
Histological changes
Thirteen of the fourteen patients (93%) underwent a liver
biopsy prior to treatment initiation, with a median interval of 2.88 years (0.73–2.98)
between biopsy and bulevirtide initiation. At baseline, 5 patients (38%) were
staged as F4, 4 (31%) as F3, 1 (8%) as F2, 2 (15%) as F1 and 1 (8%) as F0
according to METAVIR fibrosis staging. Inflammatory activity was classified as A2
in 8 patients (73%) and A1 in 3 patients (27%), with data missing in 2 cases. During
follow-up, 7 patients underwent a repeat liver biopsy at a median of 1.16 years
(1.06–1.90) after treatment initiation. Figure 3 illustrates the individual changes
in METAVIR fibrosis stages and inflammatory activity grades in these patients.
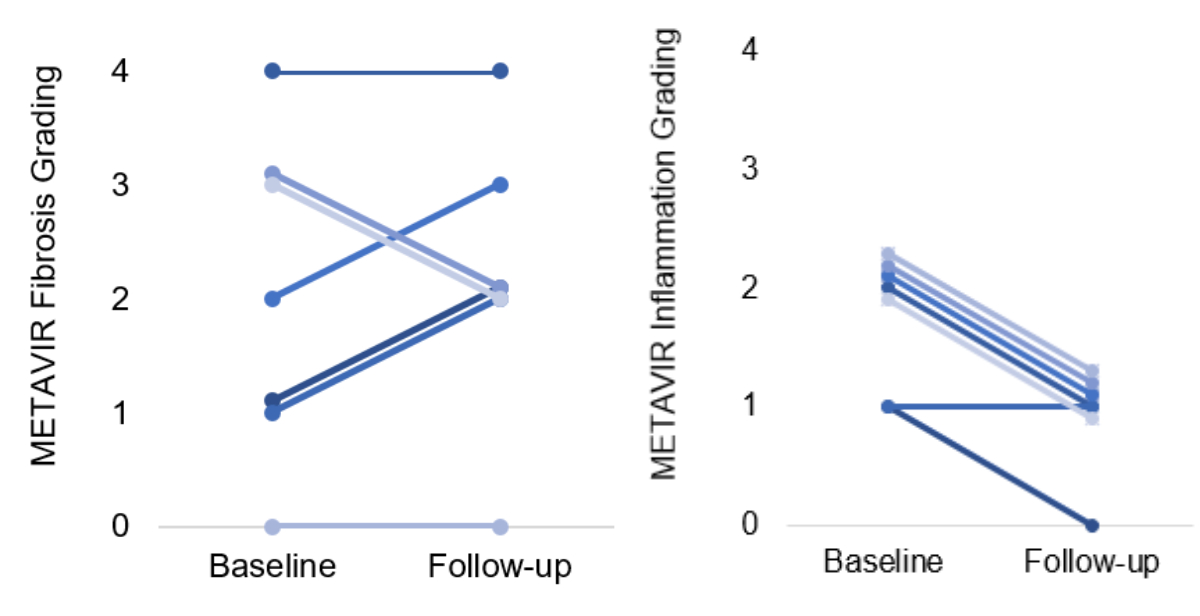
Figure 3METAVIR grading of fibrosis and inflammation on liver
histology before bulevirtide treatment and during follow-up. Only patients with a
biopsy before and
during treatment are represented on this graph (n = 7).
Liver stiffness measurement
Baseline liver stiffness measurements (LSM) were available in
13 of 14 patients (93%), with a median value of 15.3 kPa (11.8–18.94). The
median time between baseline LSM and treatment initiation was 0.67 years (0.28–1.23).
During follow-up, LSM was repeated in 8 patients (57%). In this subgroup,
median LSM decreased from 14.05 kPa (11.3–17.0) at baseline to 9.15 kPa (5.5–13.7)
at follow up (p = 0.161). The median interval between treatment initiation and
the most recent LSM was 1.93 years (1.36–2.48).
Liver-related events and survival
Decompensation, hepatocellular carcinoma and liver transplantation
One patient (7%) developed ascites at month 36, which persisted
until the end of follow-up. No additional events of hepatic decompensation were
reported. Another patient (7%) developed a portal vein thrombosis after 48
months of treatment, in the absence of hepatocellular carcinoma. Two patients (14%)
were diagnosed with hepatocellular carcinoma during follow-up: one at month 6 and
one at month 12. Both were treated with local therapy. One of these patients
had a known history of hepatocellular carcinoma prior to bulevirtide initiation
and developed recurrent lesions during treatment. This patient ultimately underwent
liver transplantation 13 months after starting bulevirtide.
Adverse events and treatment compliance
Bile acid levels increased from a median of 10.8 μmol/l at baseline (9.9–13.2, n =
7) to
55.8 μmol/l (23.2–96.2) at last
follow-up. Two patients (14%) reported mild pruritus at month 6, which resolved
spontaneously. No further adverse events were reported during treatment. One patient
(7.14%) discontinued therapy due to non-compliance. This patient had a history
of substance use and psychosocial instability, which likely contributed to
treatment discontinuation.
Discussion
This study presents long-term real-world data on the
efficacy and safety of bulevirtide 2 mg/day in patients with HDV infection and
compensated liver cirrhosis treated within a compassionate use programme in Switzerland,
with a follow-up of up to 60 months.
The baseline characteristics of our cohort were comparable to those reported in the
registration study [8] and the SAVE-D study [9], with similar levels of disease severity,
prior interferon exposure, and biochemical and virological parameters. Our cohort
had a higher proportion of non-Caucasian patients and men, and three patients received
concomitant pegylated interferon-α therapy, which was not used in the SAVE-D study
[9].
Biochemical response rates, defined by ALT normalisation, were comparable to published
data (66.7% in our cohort vs 60–64% in previous studies) [8, 9]. The decline in response
rate at month 60 (50%) is difficult to interpret due to the low number of patients
(n = 2) at that time point.
Virological response rates in our study (66.7–75% up to month 48) were also in line
with previously reported data (76–79%) [8, 9]. Combined response rates were similar
to those in the SAVE-D trial [9].
One patient discontinued treatment due to non-adherence. This individual had a history
of intravenous drug use, benzodiazepine and opioid dependency, and psychosocial instability,
highlighting the need for personalised adherence strategies in vulnerable populations.
Importantly, all patients were included based on a clinical diagnosis of compensated
cirrhosis, established using non-invasive tests such as liver stiffness measurement
(LSM), typical imaging and laboratory findings. Histological confirmation was not
required, and in some cases liver biopsies revealed fibrosis stages inconsistent with
cirrhosis. This likely reflects sampling variability and the recognised limitations
of liver biopsy in staging HDV-related liver diseases, particularly in heterogeneous
or patchy fibrosis. All patients in our cohort were already receiving nucleos(t)ide
analogue therapy for HBV prior to initiation of bulevirtide. Therefore, it is also
conceivable that some patients experienced partial regression of fibrosis under long-term
HBV suppression, contributing to the discrepancy between clinical staging and histological
findings. Interestingly, in three patients, the follow-up biopsy showed a higher fibrosis
stage compared to baseline histology, as illustrated in figure 3. This observation
appears paradoxical in the context of concurrent virological and biochemical improvement.
Several factors may contribute to this finding. First, liver biopsy is subject to
sampling variability, especially in diseases such as HDV where fibrosis is heterogeneously
distributed [11, 12]. It is possible that fibrosis was underestimated in the initial
biopsy and better captured in the follow-up specimen. Second, semi-quantitative scoring
systems such as METAVIR are susceptible to interobserver variability, particularly
in distinguishing between advanced stages [13]. These considerations underline the
interpretive limitations of liver histology in HDV and reinforce the value of combining
biopsy with longitudinal non-invasive assessments for reliable staging and monitoring.
Our findings underscore the relevance of integrating non-invasive tests into clinical
decision-making for patient selection and monitoring in HDV, as also recommended by
EASL guidelines [1]. Follow-up histology in seven patients demonstrated reduced inflammatory
activity in most cases, consistent with biochemical and virological improvement.
In parallel, LSM decreased from a median of 14.05 to 9.15 kPa during follow-up in
a subgroup of eight patients. Although this reduction did not reach statistical significance
(p = 0.161), it mirrors the improvements seen in laboratory and histological parameters,
and suggests a trend towards fibrosis regression with ongoing therapy.
Notably, two patients (14%) developed de novo hepatocellular carcinoma, and one underwent
liver transplantation. These rates are higher than in the SAVE-D study (hepatocellular
carcinoma 2.5%) [9], likely reflecting the advanced disease stages of patients included
in the compassionate use programme, where enrolment was limited to high-risk individuals.
One additional patient experienced hepatic decompensation. These findings highlight
the aggressive nature of HDV and the importance of early antiviral intervention.
Safety and tolerability of bulevirtide were excellent. Only two patients reported
mild, transient pruritus, and no serious adverse events were observed. Despite the
requirement for daily subcutaneous administration, adherence was high in all but one
patient. Bile acid levels increased during therapy, as expected with NTCP inhibition,
but without clinical consequences.
Limitations of this study include the small cohort size, its retrospective design
and heterogeneous HDV RNA assays across participating centres. These factors limit
direct comparison of absolute viral load changes. Nonetheless, the consistent trends
in virological and biochemical response support the overall validity of our findings.
The main strength of this study is its real-world nature and the inclusion of long-term
follow-up data from multiple Swiss centres, providing valuable insight into national
treatment experience.
Conclusions
This real-world Swiss cohort confirms the antiviral efficacy
and favourable safety profile of bulevirtide in patients with compensated HDV-related
cirrhosis, including those with features of portal hypertension. Bulevirtide
was well tolerated and associated with sustained virological and biochemical
responses. Future studies with larger sample sizes and longer follow-up are
needed to evaluate the long-term impact of bulevirtide on liver-related outcomes
and disease progression.
Data sharing statement
The data supporting the findings of this study is not
publicly available due to ethical restrictions. However, aggregated or summarised
data may be available from the corresponding author upon reasonable request.
Acknowledgments
Author contributions: MK: initiation, data acquisition, analysis, writing the
draft, finalising the manuscript; NS: conceptualisation, writing the draft,
revision and approval of the final manuscript; all other authors: data acquisition,
revision and approval of the final manuscript.
Prof. Dr Nasser Semmo
Hepatology, University Clinic
for Visceral Surgery and Medicine
Inselspital Bern
Murtenstrasse 35
CH-3010
Bern
nasser.semmo[at]insel.ch
References
1. Brunetto MR, Ricco G, Negro F, Wedemeyer H, Yurdaydin C, Asselah T, et al.; European
Association for the Study of the Liver. EASL Clinical Practice Guidelines on hepatitis
delta virus. J Hepatol. 2023 Aug;79(2):433–60. doi: https://doi.org/10.1016/j.jhep.2023.05.001
2. Stockdale AJ, Kreuels B, Henrion MY, Giorgi E, Kyomuhangi I, de Martel C, et al. The
global prevalence of hepatitis D virus infection: systematic review and meta-analysis.
J Hepatol. 2020 Sep;73(3):523–32. doi: https://doi.org/10.1016/j.jhep.2020.04.008
3. Miao Z, Zhang S, Ou X, Li S, Ma Z, Wang W, et al. Estimating the Global Prevalence,
Disease Progression, and Clinical Outcome of Hepatitis Delta Virus Infection. J Infect
Dis. 2020 Apr;221(10):1677–87. doi: https://doi.org/10.1093/infdis/jiz633
4. Wedemeyer H, Yurdaydin C, Hardtke S, Caruntu FA, Curescu MG, Yalcin K, et al.; HIDIT-II
study team. Peginterferon alfa-2a plus tenofovir disoproxil fumarate for hepatitis
D (HIDIT-II): a randomised, placebo controlled, phase 2 trial. Lancet Infect Dis.
2019 Mar;19(3):275–86. doi: https://doi.org/10.1016/S1473-3099(18)30663-7
5. Wedemeyer H, Schöneweis K, Bogomolov P, Blank A, Voronkova N, Stepanova T, et al. Safety
and efficacy of bulevirtide in combination with tenofovir disoproxil fumarate in patients
with hepatitis B virus and hepatitis D virus coinfection (MYR202): a multicentre,
randomised, parallel-group, open-label, phase 2 trial. Lancet Infect Dis. 2023 Jan;23(1):117–29.
doi: https://doi.org/10.1016/S1473-3099(22)00318-8
6. Bogomolov P, Alexandrov A, Voronkova N, Macievich M, Kokina K, Petrachenkova M, et
al. Treatment of chronic hepatitis D with the entry inhibitor myrcludex B: first results
of a phase Ib/IIa study. J Hepatol. 2016 Sep;65(3):490–8. doi: https://doi.org/10.1016/j.jhep.2016.04.016
7. Asselah T, Chulanov V, Lampertico P, Wedemeyer H, Streinu-Cercel A, Pântea V, et al. Bulevirtide
Combined with Pegylated Interferon for Chronic Hepatitis D. N Engl J Med. 2024 Jul;391(2):133–43.
doi: https://doi.org/10.1056/NEJMoa2314134
8. Wedemeyer H, Aleman S, Brunetto M, Blank A, Andreone P, Bogomolov P, et al. Bulevirtide
monotherapy in patients with chronic HDV: efficacy and safety results through week
96 from a phase III randomized trial. J Hepatol. 2024 Oct;81(4):621–9. doi: https://doi.org/10.1016/j.jhep.2024.05.001
9. Degasperi E, Anolli MP, Jachs M, Reiberger T, De Ledinghen V, Metivier S, et al. Real-world
effectiveness and safety of bulevirtide monotherapy for up to 96 weeks in patients
with HDV-related cirrhosis. J Hepatol. 2025 Jun;82(6):1012–22. doi: https://doi.org/10.1016/j.jhep.2024.12.044
10. Archer AJ, Belfield KJ, Orr JG, Gordon FH, Abeysekera KW. EASL clinical practice guidelines:
non-invasive liver tests for evaluation of liver disease severity and prognosis. Frontline
Gastroenterol. 2022 Feb;13(5):436–9. doi: https://doi.org/10.1136/flgastro-2021-102064
11. Siddique I, El-Naga HA, Madda JP, Memon A, Hasan F. Sampling variability on percutaneous
liver biopsy in patients with chronic hepatitis C virus infection. Scand J Gastroenterol.
2003 Apr;38(4):427–32. doi: https://doi.org/10.1080/00365520310000825
12. Takyar V, Surana P, Kleiner DE, Wilkins K, Hoofnagle JH, Liang TJ, et al. Noninvasive
markers for staging fibrosis in chronic delta hepatitis. Aliment Pharmacol Ther. 2017 Jan;45(1):127–38.
doi: https://doi.org/10.1111/apt.13834
13. Regev A, Berho M, Jeffers LJ, Milikowski C, Molina EG, Pyrsopoulos NT, et al. Sampling
error and intraobserver variation in liver biopsy in patients with chronic HCV infection.
Am J Gastroenterol. 2002 Oct;97(10):2614–8. doi: https://doi.org/10.1111/j.1572-0241.2002.06038.x


