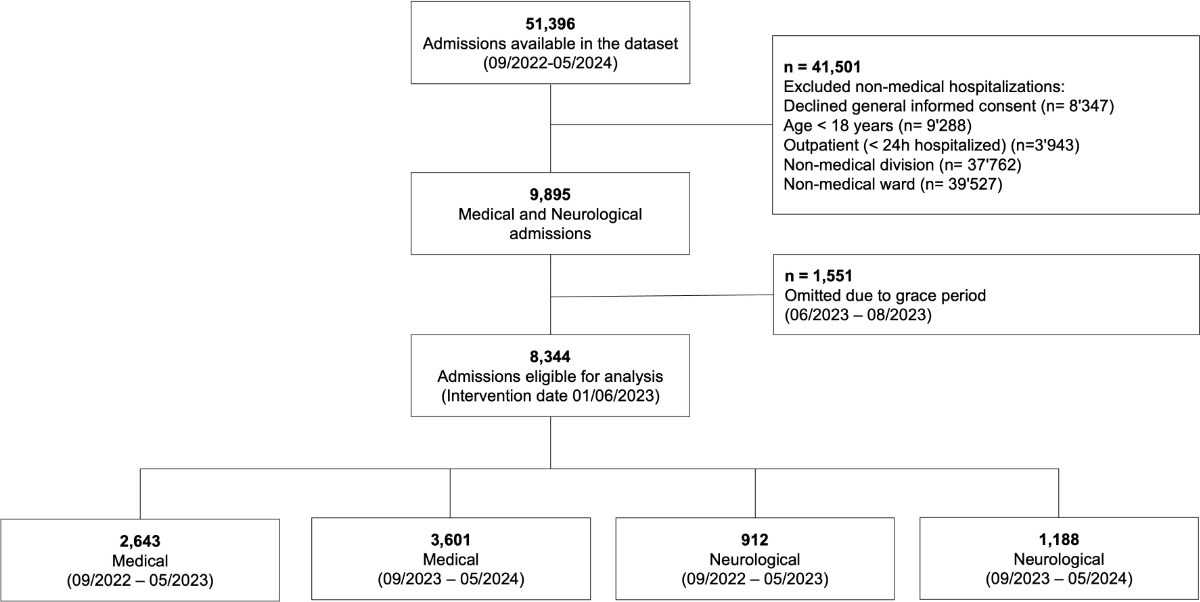
Figure 1Study flowchart.
DOI: https://doi.org/https://doi.org/10.57187/4539
In recent decades, healthcare costs have surged in all developed countries, exacerbated by an ageing population and an increasing shortage of medical personnel [1–4]. Several studies have shown an association between high hospital bed occupancy and increased in-hospital mortality [5]. Also, studies indicate that low staff retention with both nurses and physicians [6], and low nursing skill levels are associated with inpatient mortality [7]. On the other hand, medical overuse is increasingly being recognised as harmful [8]. In addition to medication overuse, overuse of routine diagnostic measures may also have negative effects on hospital resources and costs Yet, there is still insufficient evidence on which and how many interventions are meaningful for maintaining consistent quality in medical inpatient care [9].
While the United States (US) launched the “Choosing Wisely” campaign [10], the Swiss Society of Medical Physicians also initiated a similar campaign called smarter medicine in 2014 [11]. The aims of smarter medicine are to ensure that medical interventions are only applied when they provide benefit to patients, promote discussions and research on unnecessary treatments, and offer support in implementation. We used these recommendations to develop internal guidelines in our hospital, known as copAIN, which aim to reduce unnecessary procedures such as vital sign measurements, blood sugar tests and laboratory orders while ensuring high-quality patient care. The word is a neologism combining “cop” which refers to the co-operation between nursing staff and physicians, and “AIN”, which stands for “Allgemeine Innere Medizin” (“General Internal Medicine” in German).
The objective of this study was to quantify the impact of implementing copAIN on resource utilisation and patient care quality by applying an interrupted time-series analysis following its implementation on 1 June 2023.
The Cantonal Hospital Aarau is a 500-bed tertiary care, academic hospital. Faced with staff shortages, the interdisciplinary copAIN project was launched on 7 February 2023 to improve quality while maintaining an efficient use of resources. Interdisciplinary working groups identified measures for workload reduction focusing on standardising admission protocols, improving communication, and streamlining diagnostic and therapeutic procedures. Following internal workshops and consultative processes, measures were approved within the General Internal Medicine department in April 2023. These measures were distributed as both printed pocket guides and electronic resources. They included pathways for communication between nursing staff and physicians, adjustment of default settings for patient admissions (e.g. frequency of vital sign monitoring), giving agency to nurses to adjust medication timing and algorithms for scenarios such as fall-related head injury risk assessment or diagnostic pathways for pulmonary embolism. All these pathways were supported by existing literature [12–17].
On 5 June 2023, the copAIN project was officially implemented. One key element was establishing and maintaining a culture of continuous feedback, allowing for regular adjustments and improvements. This approach facilitated ongoing enhancements in communication pathways, daily routines, ward rounds and the expansion of nursing competencies, all aimed at improving patient care.
Single-centre prospective cohort study in adult inpatients (≥18 years) hospitalised in the Medical University Department, Cantonal Hospital Aarau, Switzerland between 1 September 2022 and 31 May 2024. Individual-level data was extracted from the electronic health records (EHR) for all hospitalisations. The data included patient demographics, healthcare utilisation, medical diagnoses, diagnostic tests, clinical procedures and in-hospital outcomes. Medical diagnoses were coded using the International Classification of Disease version 10, German Modification (ICD-10 GM).
Institutional review board approval was provided by the Institutional Review Board of Northwestern Switzerland (EKNZ Project-ID: Req-2021-01397). As the study data consisted of data obtained during standard clinical care, the need for individualised and project-specific informed consent was waived. This study was conducted according to the principles stated in the Declaration of Helsinki and followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines [33].
To be eligible, patients had to be general internal medicine inpatients aged ≥18 years with a length of stay (LOS) of at least 24 hours. Therefore, all other non-medical cases, including those from the intensive care unit and intermediate-care units (e.g. stroke unit), were not analysed. Cases with missing or invalid data in key variables were excluded through filtering and intersection steps. No imputation was performed. Only complete cases meeting all inclusion criteria were analysed.
Neurological inpatients, including those discharged from the stroke unit, served as a control group, as they were cared for in other wards not part of the intervention. _Hlk205885419Patients who refused to grant general informed consent were excluded. In Switzerland, the general informed consent is a broad consent sought at hospital admission that permits the use of patient health data for research and quality improvement while protecting individual privacy [18, 19].
The following elements were part of the copAIN project. The standard order set for vital signs was electronically modified by the admission team to a default of once-daily measurements replacing the previous routine of three times daily. The default did not vary depending on the degree of illness. However, physicians retained the ability to override this default if more-frequent monitoring was clinically indicated. Additionally, blood sugar (BS) management was adjusted to a monitoring frequency based on comorbidities (diabetes mellitus), therapy status and risk of hyperglycemia (BMI >30 kg/m², fasting BS >7 mmol/l, random BS >11 mmol/l or >10mg prednisone-equivalent). The intensity of all monitoring was re-evaluated every 3 days; both an increase and decrease in monitoring, depending on the clinical situation, could be instigated by the nursing staff instead of relying on physician approval. The scope and limitations of the nursing staff’s authorisation to adjust medication intake were defined in the copAIN pamphlet (page 6: “Nurses are able to start, stop, or change the frequency of surveillance measures on their own.”). This means that nurses have documentation competence and may independently start or stop documenting relevant parameters in accordance with clinical guidelines (e.g. a normal respiratory rate maintained for 2 days).
As laboratory orders are time-consuming, costly and burdensome for patients, we optimised their timing and frequency. We defined a laboratory order as a single blood draw that includes multiple tests. Specific schedules were defined: initial laboratory orders on admission (usually performed in the emergency room), follow-up laboratory orders on days 3–4 and weekly laboratory orders for longer stays exceeding 7 days [20].
Given that the project required time for implementation, a 3-month grace period was defined. This ensured that the groups before and after the intervention were also seasonally matched leading to 9 months before (Sept 2022 to May 2023) and 9 months after (Sept 2023 to May 2024) the intervention. While longer observation periods of 12 or 18 months could potentially provide additional insights, data beyond May 2024 was not available at the time of analysis, which limited the feasible duration of the post-intervention period. Therefore, the study period was chosen based on both methodological considerations and data availability. We did not perform a formal sample size calculation. Instead, we included all available and eligible data during the defined study period to maximise the robustness and representativeness of the analysis.
The primary outcome was an absolute reduction in vital sign measurements and laboratory orders adjusted for LOS (i.e. the average number of measurements per patient per day of hospitalisation, calculated as the total number of measurements recorded during the hospital stay divided by the patient’s LOS in days). The primary outcome was analysed at the individual hospitalisation level, with measurements aggregated per patient per LOS. To identify adverse events, we defined all-cause in-hospital mortality as a safety outcome.
Descriptive statistics were used to describe the population. Data transformation and preparation was done in R and the Elixhauser index was calculated using the comorbidity package based on the available ICD-10 GM codes [21]. To calculate the average number of daily measurements per patient, the total number of measurements per hospitalisation was divided by the number of hospitalisation days for each vital and laboratory order.
_Hlk206080498_Hlk206396640We used a linear mixed-effects regression model (Stata v17, StataCorp, USA) to evaluate the effect of the intervention over time, structured as an interrupted time series (ITS) with repeated patient-level measurements. The model included fixed effects for time (pre-, grace and post-intervention), their interactions with the intervention indicator and covariates (age, sex, insurance class, Elixhauser Comorbidity Index), selected based on prior evidence of association with resource use and to adjust for potential confounding. Random effects included both a patient-level random intercept and random slopes for each time period, accounting for intra-individual correlation. Standard errors were estimated using restricted maximum likelihood (REML) with an unstructured covariance matrix [22].
We generated marginal plots using the marginscontplot2 command. While this command allows inclusion of 95% confidence intervals, graphical limitations prevented a clear visual plot in figure 2. For precise estimates, we refer to table 2 [23]. All significant tests were two-sided at an alpha level of 5%.
We performed a sensitivity analysis by increasing the time threshold for measurement inclusion from 1 to 2 hours, thereby excluding a greater proportion of acute and intensified monitoring typically associated with interventions and emergency cases (see tables S1–S4 and figure S1 in the appendix).
A total of 51,396 cases were registered between 1 September 2022 and 31 May 2024. Of the 8344 cases eligible for analysis, the medical group had 2643 cases before the intervention (1 September 2022 to 31 May 2023) and 3601 cases after the intervention (1 September 2023 to 31 May 2024). The neurology group had 912 cases before and 1188 cases after the intervention, and served as a control group (see figure 1).

Figure 1Study flowchart.
Baseline characteristics were similar in pre- and post-intervention groups with minor differences in age, sex distribution and comorbidity burden. Supplementary private insurance rates as a proxy of socioeconomic status were comparable.
In-hospital mortality and Elixhauser comorbidity index scores were consistently higher in the medical group than in the neurology group.
Median LOS decreased in both groups over the study period and emergency admissions remained stable. Overall, there were no major differences in baseline characteristics between the two groups, except for those mentioned above (table 1).
Table 1Patient characteristics (intervention start date: 1 June 2023).
| Characteristics | Medicine (copAIN project) | Neurology (control group) | |||
| Before intervention (n = 2643) | After intervention (n = 3601) | Before intervention (n = 912) | After intervention (n = 1188) | ||
| Age in years, mean (SD) | 68.5 (15.4) | 69.1 (15.5) | 65.5 (18.1) | 67.3 (17.7) | |
| Female, n (%) | 994 (37.6%) | 1480 (41.1%) | 423 (46.4%) | 591 (49.7%) | |
| Supplementary hospital insurance, n (%) | 471 (17.8%) | 650 (18.1%) | 175 (19.2%) | 247 (20.8%) | |
| Length of stay in days, median (IQR) | 5.30 (2.90–9.50) | 3.90 (2.20–7.00) | 4.10 (2.90–8.00) | 3.60 (2.30–5.80) | |
| Emergency admission, n (%) | 1927 (72.9%) | 2520 (70.0%) | 731 (80.2%) | 972 (81.8%) | |
| Preadmission, n (%) | Home | 2250 (85.1%) | 3022 (83.9%) | 698 (76.5%) | 895 (75.3%) |
| Skilled Nursing Facility (SNF) | 141 (5.3%) | 223 (6.2%) | 46 (5.0%) | 71 (6.0%) | |
| Hospital | 183 (6.9%) | 237 (6.6%) | 125 (13.7%) | 185 (15.6%) | |
| Other | 69 (2.6%) | 119 (3.3%) | 43 (4.7%) | 37 (3.1%) | |
| Department, n (%) | Medicine | 593 (22.4%) | 979 (27.2%) | 0 | 0 |
| Oncology | 559 (21.2%) | 649 (18.0%) | 0 | 0 | |
| Cardiology | 821 (31.1%) | 1,025 (28.5%) | 0 | 0 | |
| Nephrology | 106 (4.0%) | 100 (2.8%) | 0 | 0 | |
| Pulmonology | 203 (7.7%) | 373 (10.4%) | 0 | 0 | |
| Gastroenterology | 315 (11.9%) | 393 (10.9%) | 0 | 0 | |
| Neurology | 0 | 0 | 912 (100%) | 1,188 (100%) | |
| Other (Dermatology, Angiology) | 46 (1.7%) | 82 (2.3%) | 0 | 0 | |
| Discharge, n (%) | Home | 2011 (76.1%) | 2814 (78.1%) | 599 (65.7%) | 889 (74.8%) |
| Health care institution | 439 (16.6%) | 561 (15.6%) | 262 (28.7%) | 245 (20.6%) | |
| Hospital | 78 (3.0%) | 81 (2.2%) | 30 (3.3%) | 40 (3.4%) | |
| Deceased | 103 (3.9%) | 128 (3.6%) | 12 (1.3%) | 8 (0.7%) | |
| Elixhauser index, median (IQR) | 14.0 (5.00–26.0) | 13.0 (3.00–25.0) | 7.00 (0–16.0) | 6.00 (0–13.0) | |
IQR: interquartile range.
In the medical group, there was an overall reduction in the frequency of vital sign measurements per day per length of stay (LOS) across all parameters (blood pressure, heart rate, oxygen saturation and temperature). In contrast, the neurology group exhibited stable or slightly increasing measurement rates (table 2).
Table 2Results (intervention start date: 1 June 2023).
| Measurements (per day per length of stay), mean (SD) | Medicine (copAIN project) | Neurology (control group) | Model adjusted difference* | |||||
| Before intervention (n = 2643) | After intervention (n = 3601) | Difference (95% CI) | Before intervention (n = 912) | After intervention (n = 1188) | Difference(95% CI) | Model | p-value | |
| Blood pressure | 2.82 (1.01) | 2.54 (1.09) | 0.28 (0.23 to 0.33) | 2.81 (1.05) | 2.95 (1.04) | –0.14 (–0.23 to –0.05) | –0.12 (–0.15 to –0.09) | <0.01 |
| Heart rate | 2.86 (1.01) | 2.60 (1.09) | 0.26 (0.21 to 0.31) | 2.81 (1.02) | 2.95 (1.04) | –0.14 (–0.23 to –0.05) | –0.12 (–0.15 to –0.09) | <0.01 |
| Oxygen saturation | 2.86 (1.00) | 2.58 (1.09) | 0.28 (0.23 to 0.33) | 2.63 (0.96) | 2.79 (1.03) | –0.16 (–0.25 to –0.07) | –0.11 (–0.14 to –0.08) | <0.01 |
| Temperature | 2.65 (1.02) | 2.38 (1.13) | 0.27 (0.22 to 0.32) | 1.61 (0.68) | 1.53 (0.66) | 0.08 (0.02 to 0.14) | –0.10 (–0.13 to –0.07) | <0.01 |
| Blood glucose | 2.24 (1.43) | 2.39 (1.48) | –0.15 (–0.22 to –0.08) | 1.50 (1.11) | 1.68 (1.12) | –0.18 (–0.28 to –0.08) | –0.05 (–0.12 to 0.01) | 0.18 |
| Laboratory orders | 0.77 (0.59) | 0.68 (0.60) | 0.09 (0.06 to 0.12) | 0.46 (0.49) | 0.37 (0.47) | 0.09 (0.05 to 0.13) | –0.01 (–0.02 to 0.01) | 0.29 |
| Mortality, n (%) | 103 (3.9%) | 128 (3.6%) | –0.3% (–0.6% to 1.3%) | 12 (1.3%) | 8 (0.7%) | –0.6% (–0.2% to 1.5%) | 0.03 (–0.13% to 0.18%) | 0.74 |
*Adjusted with multivariable mixed-effects model with random slope for age, sex, insurance and Elixhauser index; for details, see table S1 in the appendix.
There was no absolute reduction in the number of blood glucose measurements per day per length of stay observed (table 2). Even when analysing non-diabetic and diabetic patients separately, no decrease in blood glucose measurements emerged.
There was a slight absolute reduction in the number of laboratory orders taken per day per length of stay in both groups (table 2).
The mixed-effects model showed a significant downward trend (change in slope) in vital sign measurements compared with no significant change in the control group. Complete model results for all parameters, including beta coefficients and confidence intervals, are presented in table 2 and illustrated in figure 2.
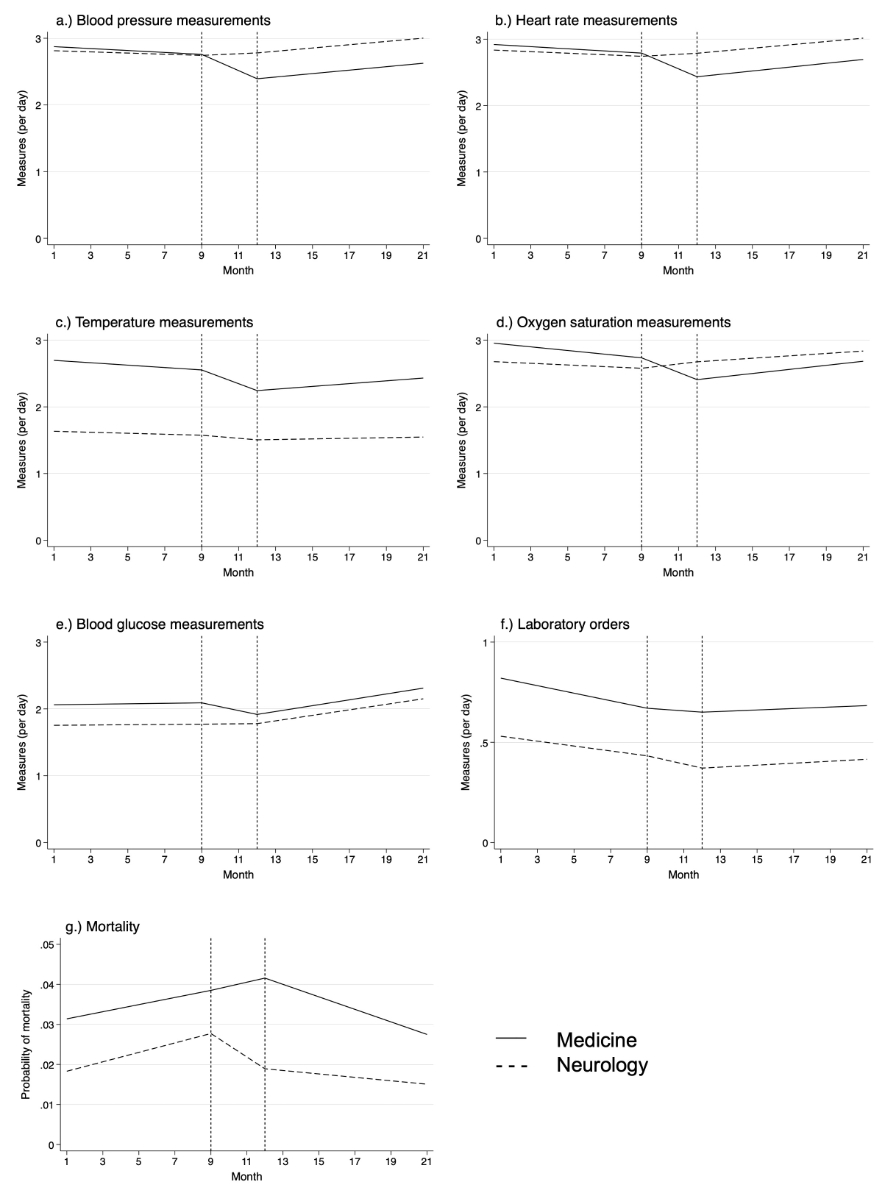
Figure 2Multivariable random slope mixed-effects model.
The results were as expected and consistent with the primary analysis, confirming the robustness of our findings (see tables S1–S4 and figure S1 in the appendix).
There was no significant change in in-hospital mortality over time before and after the intervention in both groups (table 2).
In this retrospective single-centre cohort study in 8344 admissions, we analysed the implementation of the copAIN project. Using cases from the neurology clinic as a control group, our analysis showed that the copAIN project led to an absolute reduction in vital sign measurements and laboratory orders but had no effect on blood glucose measurements. There was a weaning effect after the implementation period. No adverse effects were detected.
Similar process optimisation projects have been reported. The patient-centred management tool developed at University Hospital Basel is strengthening interprofessional and interdisciplinary collaboration. Its aim is to ensure high-quality healthcare while also achieving positive financial effects. Currently, the authors plan on extending the study over a longer period to assess the impact and data on financial effects is not yet available [24].
Also, a recent study by Rawal et al. exemplified that targeted measures can help save resources by reducing the number of laboratory orders without compromising quality [25]. They demonstrated an increased resident perception of both mindful ordering and team discussion resulting in a decrease of 20% in laboratory orders per week in the first year. The reduction in laboratory ordering was sustained throughout 127 weeks. There was an approximately 10% decrease in patients undergoing venipuncture each day per month. The study shows the impact of focusing interventions on resident-identified barriers to mindful ordering to create a sustained decrease in laboratory ordering. On the other hand, a reduction in the number of blood draws can lower resource use and costs which in turn can be offset by an increase in the number of tests performed per draw. However, we did not address this aspect in this study.
Implementation of change management projects is supported by clear communication with staff through training sessions, personalised emails, adjustments in the electronic health record (EHR), and printed materials. However, maintaining the desired outcome is challenging. The literature suggests that up to 70% of change management projects fail to meet their goals [26]. Despite changes in our standard admission orders in our EHR (KISIM®), frequent blood glucose measurements continued even in patients without diabetes. This lack of sustained effect is usually attributed to frequent staff turnover and the separation of the emergency and medical departments limiting the visibility and consistent application of copAIN measures. Another factor might be the fear of hypoglycaemic events by nurses that may lead to a more cautious approach, such as increased blood glucose monitoring.
Fewer routine monitoring tasks per patient could allow staff to focus on other activities, though quantifying efficiency gains is challenging. While relevant literature is limited, recent studies have explored cost savings with AI-assisted or continuous vital sign monitoring systems, which contrast with our project’s goals. These studies showed that such systems reduce serious adverse events, leading to lower healthcare costs through shorter hospital stays and avoided readmissions. For example, a Scandinavian study estimated potential savings of €2040 per patient due to reduced LOS and €43 from avoided readmissions [27]. A US study found potential annual savings of over $6.8 million in a community hospital by improving patient outcomes with continuous monitoring, driven by shorter stays, fewer intensive care unit (ICU) transfers and reduced rapid response team activations [28].
Various frameworks for change management exist such as Six Sigma, Lean, Sustainability Model, and the Model of Improvement [29, 30]. After initiating a project, it is key to implement Plan-Do-Study-Act cycles to gain knowledge by testing changes and reflecting on their effect. To achieve sustainable change, an intervention must become the new routine. Silver et al. showed examples of applying the Sustainability Model to help identify issues that affect long-term success of quality improvement projects [31, 32]. In the copAIN project, elements from various quality improvement frameworks were integrated including an initial interdisciplinary and interprofessional group phase to outline the intervention. This phase involved brainstorming and creating small groups to focus on specific improvement tasks such as communication between nurse and physicians, admission pathway, monitoring and documentation, empowerment (see copAIN handout available for download as a separate file at https://doi.org/10.57187/4539).
Following the initiation of copAIN, weekly internal meetings and regular training sessions were held to support the project and promote sustainable change, aiming for copAIN to become part of the routine. Despite these efforts, there was a weaning effect over time. To ensure sustainability, an interdisciplinary committee of physicians and IT specialists has now been established following our analysis to develop an interactive dashboard that provides staff with monthly, individualised feedback.
External validity of our study is constrained by its single-centre cohort design. Another limitation is the increase in admissions over the study period while the severity of illness and consequently LOS decreased, potentially explaining the reduced rate of monitoring. However, the neurology group had an increased frequency of monitoring, despite similar changes. The Division of General Internal and Emergency Medicine at the Cantonal Hospital Aarau encompasses different subdisciplines (e.g. haematology, oncology, cardiology, pneumology, nephrology, gastroenterology). Certain oncology patients such as high-dose chemotherapy in leukaemia were not subjected to the copAIN protocol but were still included in our analysis. Although this is not ideal, it makes our results more conservative.
Another limitation stems from the pre-post intervention study design, which inherently introduces potential biases. Notably, the intervention’s effect was only temporary, with an initial decline in measurements and laboratory orders followed by a steady increase shortly after the intervention. This pattern contrasts with the steady decline observed during the pre-intervention period. Additionally, adjusted analyses showed no significant difference between the pre- and post-intervention periods, which may reflect limitations of the study design or challenges in achieving lasting behaviour change. The relatively short follow-up period of just nine months with diminishing effect over time further limits the ability to assess long-term sustainability.
Regarding safety assessment, we accounted for in-hospital mortality but did not examine functional outcomes. While analysing functional outcomes (indicating quality of care) would be a valuable addition and could be the basis of a separate study, it is beyond the scope of this paper.
The project implemented resource-saving measures empowering nursing staff and reducing workload without compromising healthcare performance. However, the intervention showed a diminishing effect over time, with no sustained impact observed in adjusted analyses. To ensure sustained impact, continuous efforts in change management are essential for achieving sustainable and continuous improvement.
Restrictions apply to the availability of data generated or analysed during this study to preserve patient confidentiality or because it was used under licence. The corresponding author will on request detail the restrictions and any conditions under which access to the data may be provided. The code that produces the results in this manuscript can be accessed at https://github.com/SteckAn/copAIN_Project.
Author contributions: Data access: Steck, Struja, Sigrist and Kutz had full access to all the data in the study and assume responsibility for the integrity of the data and the accuracy of the data analysis. Steck, Struja, Kutz. Sigrist, Hefti, Steck, Struja, Kutz. Steck, Struja, Kutz. All authors. Steck, Hefti, Struja, Kutz.
The study was supported by the Canton Hospital Aarau AG. The funder had no role in the design or conduct of the study; the collection, management, analysis or interpretation of the data; the preparation, review or approval of the manuscript; or the decision to submit the manuscript for publication.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflict of interest related to the content of this manuscript was disclosed.
1.Bundesamt für Statistik. Die Gesundheitskosten stiegen 2022 auf 91,5 Milliarden Franken. Available from: https://www.bfs.admin.ch/bfs/de/home/statistiken/gesundheit/kosten-finanzierung.assetdetail.31225971.html
2.Hehli S. Notstand in der Pflege: wie die Heime und Spitäler um die raren Fachkräfte kämpfen. NZZ 2022 29.12.2022; Available from: https://www.nzz.ch/schweiz/notstand-in-der-pflege-wie-die-heime-und-spitaeler-um-die-raren-fachkraefte-kaempfen-ld.1718978
3.Schweizerisches Gesundheitsobservatorium. Gesundheitspersonal in der Schweiz – Nationaler Versorgungsbericht 2021. Available from: https://www.obsan.admin.ch/sites/default/files/2021-10/Obsan_03_2021_BERICHT_0.pdf
4.Sommer P. Schwendener Schweizer Spitäler: So gesund waren die Finanzen 2021. October 8, 2022. Available from: https://www.pwc.ch/de/insights/gesundheitswesen/spitalstudie-2022.html
5.Sharma N, Moffa G, Schwendimann R, Endrich O, Ausserhofer D, Simon M. The effect of time-varying capacity utilization on 14-day in-hospital mortality: a retrospective longitudinal study in Swiss general hospitals. BMC Health Serv Res. 2022 Dec;22(1):1551. doi: https://doi.org/10.1186/s12913-022-08950-y
6.Mahase E. Higher staff turnover is linked to increased deaths in NHS hospitals, study finds. BMJ. 2024 Nov;387:q2578. doi: https://doi.org/10.1136/bmj.q2578
7.Zaranko B, Sanford NJ, Kelly E, Rafferty AM, Bird J, Mercuri L, et al. Nurse staffing and inpatient mortality in the English National Health Service: a retrospective longitudinal study. BMJ Qual Saf. 2023 May;32(5):254–63. doi: https://doi.org/10.1136/bmjqs-2022-015291
8.Brownlee S, Chalkidou K, Doust J, Elshaug AG, Glasziou P, Heath I, et al. Evidence for overuse of medical services around the world. Lancet. 2017 Jul;390(10090):156–68. doi: https://doi.org/10.1016/S0140-6736(16)32585-5
9.Wilson LM, Herzig SJ, Steinman MA, Schonberg MA, Cluett JL, Marcantonio ER, et al. Management of Inpatient Elevated Blood Pressures : A Systematic Review of Clinical Practice Guidelines. Ann Intern Med. 2024 Apr;177(4):497–506. doi: https://doi.org/10.7326/M23-3251
10.Bulger J, Nickel W, Messler J, Goldstein J, O’Callaghan J, Auron M, et al. Choosing wisely in adult hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013 Sep;8(9):486–92. doi: https://doi.org/10.1002/jhm.2063
11.smarter medicine – Choosing Wisely Switzerland. About Us. [accessed 03.06.2024]. Available from: https://www.smartermedicine.ch
12.Stiell IG, Lesiuk H, Wells GA, McKnight RD, Brison R, Clement C, et al.; Canadian CT Head and C-Spine Study Group. The Canadian CT Head Rule Study for patients with minor head injury: rationale, objectives, and methodology for phase I (derivation). Ann Emerg Med. 2001 Aug;38(2):160–9. doi: https://doi.org/10.1067/mem.2001.116796
13.Rajesh S, Wonderling D, Bernstein I, Balson C, Lecky F; Guideline Committee. Head injury: assessment and early management-summary of updated NICE guidance. BMJ. 2023 May;381:1130. doi: https://doi.org/10.1136/bmj.p1130
14.Valente JH, Anderson JD, Paolo WF, Sarmiento K, Tomaszewski CA, Haukoos JS, et al.; Members of the American College of Emergency Physicians Clinical Policies Committee (Oversight Committee). Clinical Policy: Critical Issues in the Management of Adult Patients Presenting to the Emergency Department With Mild Traumatic Brain Injury: Approved by ACEP Board of Directors, February 1, 2023 Clinical Policy Endorsed by the Emergency Nurses Association (April 5, 2023). Ann Emerg Med. 2023 May;81(5):e63–105. doi: https://doi.org/10.1016/j.annemergmed.2023.01.014
15.Kline JA, Mitchell AM, Kabrhel C, Richman PB, Courtney DM. Clinical criteria to prevent unnecessary diagnostic testing in emergency department patients with suspected pulmonary embolism. J Thromb Haemost. 2004 Aug;2(8):1247–55. doi: https://doi.org/10.1111/j.1538-7836.2004.00790.x
16.Wells PS, Anderson DR, Rodger M, Stiell I, Dreyer JF, Barnes D, et al. Excluding pulmonary embolism at the bedside without diagnostic imaging: management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and d-dimer. Ann Intern Med. 2001 Jul;135(2):98–107. doi: https://doi.org/10.7326/0003-4819-135-2-200107170-00010
17.van der Hulle T, Cheung WY, Kooij S, Beenen LF, van Bemmel T, van Es J, et al.; YEARS study group. Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet. 2017 Jul;390(10091):289–97. doi: https://doi.org/10.1016/S0140-6736(17)30885-1
18.Swiss Academy of Medical Sciences. General consent. Available from: https://www.samw.ch/en/Ethics/Topics-A-to-Z/General-consent.html
19.Swissethics. General consent. Available from: https://swissethics.ch/en/documents/generalkonsent
20. copAIN. Massnahmen zur Ressourcen-schonenden Kooperation der Ärzteschaft und Pflege. Klinik für Allgemeine Innere und Notfallmedizin (AIN), Kantonsspital Aarau. 2023.
21.Gasparini P. comorbidity: an R package for computing comorbidity scores. J Open Source Softw. 2018;3(23):648. doi: https://doi.org/10.21105/joss.00648
22.Fife D. Flexplot: graphically-based data analysis. 2019. [accessed 03.06.2024]. Available from: https://doi.org/
23.Team R. RStudio: Integrated Development for R. 2020. [accessed 24.06.2024]. Available from: http://www.rstudio.com/
24.Ahlers MR, Ratnitsky A, Biesinger B. Erfolgsfaktoren für ein patientenzentriertes Spitalmanagement. Schweiz Arzteztg. 2024;105:40–2.
25.Rawal R, Adeyemo O, Kunnath P, Vartanyan A, Saad H, Lane A, et al. Empowering medicine residents to order labs mindfully to improve patient-centered care. J Hosp Med. 2023 May;18(5):398–404. doi: https://doi.org/10.1002/jhm.13081
26.Jones-Schenk J. 70% Failure Rate: An Imperative for Better Change Management. J Contin Educ Nurs. 2019 Apr;50(4):148–9. doi: https://doi.org/10.3928/00220124-20190319-03
27.Larsen AT, Sopina L, Aasvang EK, Meyhoff CS, Kristensen SR, Kjellberg J. Estimation of the maximum potential cost saving from reducing serious adverse events in hospitalized patients. Acta Anaesthesiol Scand. 2024 Nov;68(10):1471–80. doi: https://doi.org/10.1111/aas.14525
28.Beard JW, Sethi A, Jiao W, Hyatt HW, Yapici HO, Erslon M, et al. Cost savings through continuous vital sign monitoring in the medical-surgical unit. J Med Econ. 2023;26(1):760–8. doi: https://doi.org/10.1080/13696998.2023.2219156
29. Mikel Harry Ph.D., R.S., Six Sigma: The Breakthrough Management Strategy Revolutionizing the World's Top Corporations. 2006.
30. Langley, G.J., Nolan, K. M., Nolan, T. W., Norman, C. L., & Provost, L., The Improvement Guide: A Practical Approach to Enhancing Organizational Performance. 20.04.2009.
31.Silver SA, McQuillan R, Harel Z, Weizman AV, Thomas A, Nesrallah G, et al. How to Sustain Change and Support Continuous Quality Improvement. Clin J Am Soc Nephrol. 2016 May;11(5):916–24. doi: https://doi.org/10.2215/CJN.11501015
32.Silver SA, Harel Z, McQuillan R, Weizman AV, Thomas A, Chertow GM, et al. How to Begin a Quality Improvement Project. Clin J Am Soc Nephrol. 2016 May;11(5):893–900. doi: https://doi.org/10.2215/CJN.11491015
33.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008 Apr;61(4):344–9. doi: https://doi.org/10.1016/j.jclinepi.2007.11.008
The appendix is available in the pdf version of the article at https://doi.org/10.57187/4539.
The copAIN handout is available for download as a separate file at https://doi.org/10.57187/4539.