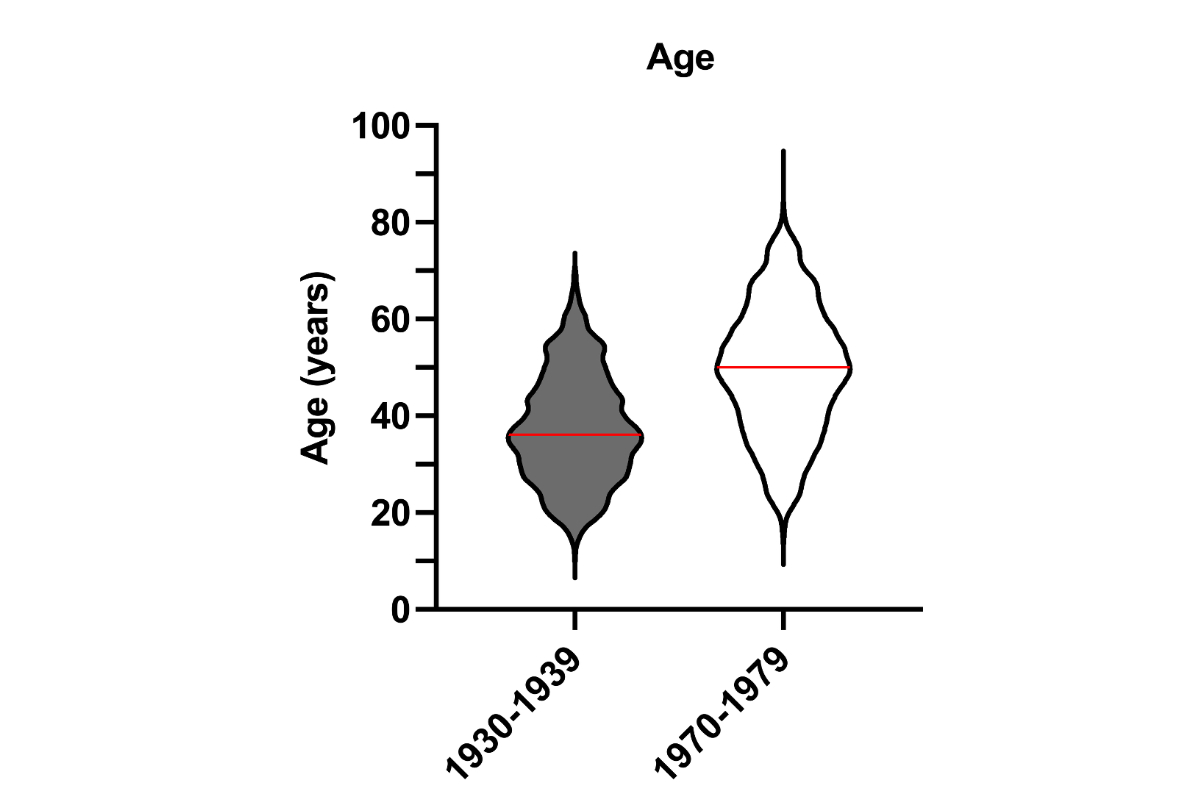
Figure 1Age of patients in 1930–1939 vs 1970–1979. The red line indicates the mean. 1930s cohort: n = 1826; mean age: 37.3 years (range: 11–69). 1970s cohort: n = 1454, mean age: 49.3 years (range: 15–74).
DOI: https://doi.org/https://doi.org/10.57187/4390
Endocrine surgery as a treatment of thyroid disorders is a relatively young subspecialty of surgery. By the end of the 19th century when thyroid surgery was initiated, postoperative mortality rates were almost as high as 40% [1]. With advances in anaesthesia, infection prevention, novel haemostatic techniques and surgical methods, outcomes of thyroid surgery considerably improved [1–4]. Most improvements were related to the work of the Nobel prize winner Theodor Kocher, who pioneered thyroid surgery and achieved a decrease in the mortality rate of thyroid surgery to 0.5% [5]. His main findings were published in an 1883 report entitled “Kropfextirpation und ihre Folgen” (Removal of the goitre and its consequences) [6].
While most cases were due to iodine-deficiency goitre, in 1922 Switzerland was the first country to introduce iodised salt, which has been available nationwide since 1952 [7]. The amount of iodine added to the salt has been gradually increased from 3.75 mg/kg back then to 20–40 mg/kg today. After the introduction, the incidence of goitre decreased rapidly and goitre due to iodine deficiency was eliminated [7].
The two most common complications specific to thyroidectomy, in the past and today, arise from the close anatomical proximity of the thyroid gland to the recurrent laryngeal nerves and the parathyroid glands.Today, reversible recurrent laryngeal nerve injury occurs in 5–11% of all cases and irreversible symptoms in 1–3.5% of thyroid surgeries, while transient hypoparathyroidism occurs in 20–30% of cases, but may persist and become chronic in 1–4% of thyroid surgeries [8–15]. The recurrent laryngeal nerve injury may give rise to vocal changes, and aspiration and respiratory symptoms [16–19], significantly impairing quality of life (QoL) [20–22]. Acute symptoms of hypoparathyroidism-induced hypocalcaemia are primarily due to neuromuscular irritability,including paraesthesia and signs of neuromuscular excitability such as Chvostek tetany [23, 24]. Even by using conventional treatment of hypoparathyroidism (i.e. active vitamin D analogues), complications may still occur resulting mainly in renal complications (kidney stones) and rarely in cataract, seizures, arrhythmia, ischaemic heart disease, greater susceptibility to infections and an overall increased all-cause mortality [25–28]. In addition, patients with surgery-induced hypoparathyroidism reported significantly impaired QoL compared to patients without hypoparathyroidism after surgery [29]. Therefore, post-thyroidectomy complications may lead to a significant burden of disease with a wide spectrum of symptoms, frequently necessitating multimodal and multidisciplinary management [30].
Multiple parameters are established as risk factors for post-thyroidectomy complications such as surgical volume of the institution, experience of the individual surgeon, underlying thyroid pathology, size of thyroid gland, patient-related risk factors such as previous neck surgery and procedure-related risk factors such as lymph node dissection [8, 11, 31–35].
The literature about systematically investigated postoperative complications of thyroidectomies is scarce. Similarly, data on potential risk factors for complications are limited and there is considerable variation in the reported data. Our comprehensive retrospective analysis of the medical records of 3230 thyroidectomies performed in Riehen (Switzerland) in the last century aims to analyse the specific complications of thyroid surgery. By analysing two decades, namely 1930–1939 (shortly after salt iodination was introduced in Switzerland) and 1970–1979 (salt iodination is well established), we aimed to investigate a potential risk factor for complications, i.e. the influence of salt iodisation on the size of the thyroid gland over time. These historical data were compared with actual Swiss DRG data and registry-based data from a European endocrine surgery registry (EUROCRINE) including five large endocrine centres of Switzerland.
This is an observational study of two retrospective cohorts. All patients who underwent a thyroidectomy in Riehen in the decades 1930–1939 and 1970–1979 and who had a standardised file/report available were included in the study. These reports were found in a loft in the community hall of Riehen and were made available to GZ. Due to the extremely standardised way of reporting the details of thyroid surgery over time, a data form and a secuTrial database at the Clinical Trials Unit of University Hospital Basel were set up for the detailed collection of anonymised data, namely on demographics, clinical and surgical characteristics, histological findings and postoperative complications. Data were entered by AA and MM and validated by GZ and EC.
Demographics included age, sex and origin of patients. Surgical characteristics comprised the weight of resected thyroid glands, the need for recurrent thyroid surgery, hemithyroidectomy, total thyroidectomy and intrathoracic goitre, defined anatomically as thyroid tissue below the thoracic inlet. Postoperative in-hospital complications such as mortality, Chvostek tetany as a surrogate for hypocalcaemia/hypoparathyroidism (no calcium concentration measurements were available), clinically diagnosed recurrent laryngeal nerve injury (dysphonia), partially confirmed by laryngoscopy and histological findings were recorded.
The relationship between the weight of the resected thyroid glands and occurrence of complications (recurrent laryngeal nerve injuries, Chvostek tetanies) was assessed as well as the risk for surgical complications in case of recurrent thyroid surgery. The data of both decades were compared.
In addition, we carried out a further comparison with recent data of surgical complications in Switzerland by analysing thyroidectomies between 2011 and 2015 using the corresponding DRGs (diagnosis-related groups) in a nationwide inpatient database. The study population consisted of adult (³18 years) inpatients who underwent thyroidectomy as the main procedure. High-volume institutions (blinded to the authors) were defined as institutions with >200 thyroidectomies per year. Postoperative complications documented in this database were those occurring within 30 days postoperatively. Hypocalcaemia was defined according to the following codes of the 10th version of the International Statistical Classification of Diseases and Related Health Problems (ICD-10): E58 Alimentary calcium deficiency; E83.58 Other disorders of calcium metabolism; E83.59 Disorders of calcium metabolism, unspecified. The recurrent laryngeal nerve injury was defined accordingly to the ICD-10 Codes: J38.00–J38.03 Paralysis of vocal folds and larynx, unspecified, partial on one side, complete on one side, partial on both sides; G52.2 Diseases of vagus nerve [X cranial nerve]. In addition, complication rates of the five largest Swiss endocrine centres (≥200 operations/year) in 2024 from the EUROCRINE registry are included. EUROCRINE (European Registry for Endocrine Surgery) documents all endocrine surgical procedures and their complications. The largest Swiss centres document their interventions in this registry.
The primary endpoint was defined as postoperative complications such as in-hospital mortality, Chvostek tetany as a surrogate for hypoparathyroidism and clinically diagnosed recurrent laryngeal nerve injury. Secondary endpoints comprised an analysis of complications first stratified by thyroid weight and second related to recurrent thyroid surgery. Thyroid weight was documented postoperatively and classified into predefined categories.
Descriptive statistics were used to summarise the patient demographics, clinical and surgical characteristics, histological findings and postoperative complications. Categorical variables were presented as counts and percentages, while continuous variables were reported as means with standard deviation and range.
Comparative analyses were performed to identify differences between the two decades in the baseline characteristics and the defined endpoints. The data were compared using appropriate statistical tests, such as the chi-squared test for categorical variables and the Mann–Whitney U test for non-normally distributed continuous variables. The incidence of surgical complications was analysed in roughly equal-sized categories of resected thyroid weight. These groups were compared with the ANOVA test. For the correlation analysis, the Spearman rank correlation was used, investigating the correlation between recurrent thyroid surgery and complications such as recurrent laryngeal nerve injuries and Chvostek tetanies. Data analysis was performed using statistical software Prism GraphPad. A p-value <0.05 was considered statistically significant. Regarding the DRG-based data, multivariate regression models were used to determine complications and mortality in relation to hospital volume. Analysed complications included recurrent laryngeal nerve injuries and hypocalcaemia.
The cantonal ethics committee (Ethikkommission Nordwest- und Zentralschweiz, EKNZ 2020 - 01982) approved this study.
The baseline characteristics of all individuals undergoing thyroidectomy are presented in table 1, stratified by time period. In the two decades analysed, 3280 patients received a thyroidectomy, 1826 patients in 1930–1939 and 1454 patients in 1970–1979. Over the entire period from 1930 to 1980, thyroid surgery at the hospital in Riehen involved only four surgeons.
Table 1Baseline characteristics.
| Characteristic | 1930–1939 (n = 1826) | 1970–1979 (n = 1454) | p | |
| Demographics | Female sex, n (%) | 1561 (85.5%) | 1209 (83.1%) | <0.01 |
| Age in years, mean ± SD, (range) | 37.3 ± 11.1, (11–69) | 49.3 ± 13.2, (15–74) | <0.01 | |
| Origin: Switzerland, n (%) | 1719 (94.1%) | 983 (67.6%) | <0.01 | |
| Origin: Canton of Basel-Stadt, n (%) | 1127 (61.7%) | 587 (40.4%) | <0.01 | |
| Clinical and surgical characteristics | Recurrent thyroid surgery, n (%) | 174 (9.5%) | 133 (9.1%) | 0.04 |
| Total thyroidectomy, n (%) | 1549 (84.8%) | 1001 (68.8%) | <0.01 | |
| Hemithyroidectomy, n (%) | 257 (14.1%) | 436 (30%) | <0.01 | |
| Extent of operation not documented, n (%) | 20 (1.1%) | 17 (1.2%) | ||
| Intrathoracic goitre, n (%) | 910 (49.8%) | 280 (19.3%) | <0.01 | |
| Thyroid weight in g, mean ± SD, (range) | 141.2 ± 99.9, (4.0–808.0) | 107.0 ± 89.3, (1.0–956.0) | <0.01 | |
| Histology | Carcinoma, n (%) | 16 (0.9%) | 23 (1.6%) | <0.01 |
| Adenoma, n (%) | 82 (4.5%) | 402 (27.6%) | 0.04 | |
| Other thyroid hyperplasia, n (%) | 1728 (94.6%) | 1029 (70.8%) | <0.01 | |
Continuous data are shown as mean ± standard deviation, (range); categorical data as count (percentage). SD: standard deviation.
Briefly, when compared with the 1970s cohort, the 1930s cohort was statistically younger (figure 1), with slightly more female patients and originated more from Switzerland and the region of Basel. There were significantly more recurrent thyroid surgeries, total thyroidectomies and intrathoracic goitre operations in the 1930s than in the 1970s. The mean weight of the operated thyroid gland was higher in the 1930s (figure 2) whereas hemithyroidectomy was performed significantly more frequently in the 1970s. On histology, there were significantly fewer carcinomas and adenomas but more thyroid hyperplasias in the 1930s.

Figure 1Age of patients in 1930–1939 vs 1970–1979. The red line indicates the mean. 1930s cohort: n = 1826; mean age: 37.3 years (range: 11–69). 1970s cohort: n = 1454, mean age: 49.3 years (range: 15–74).
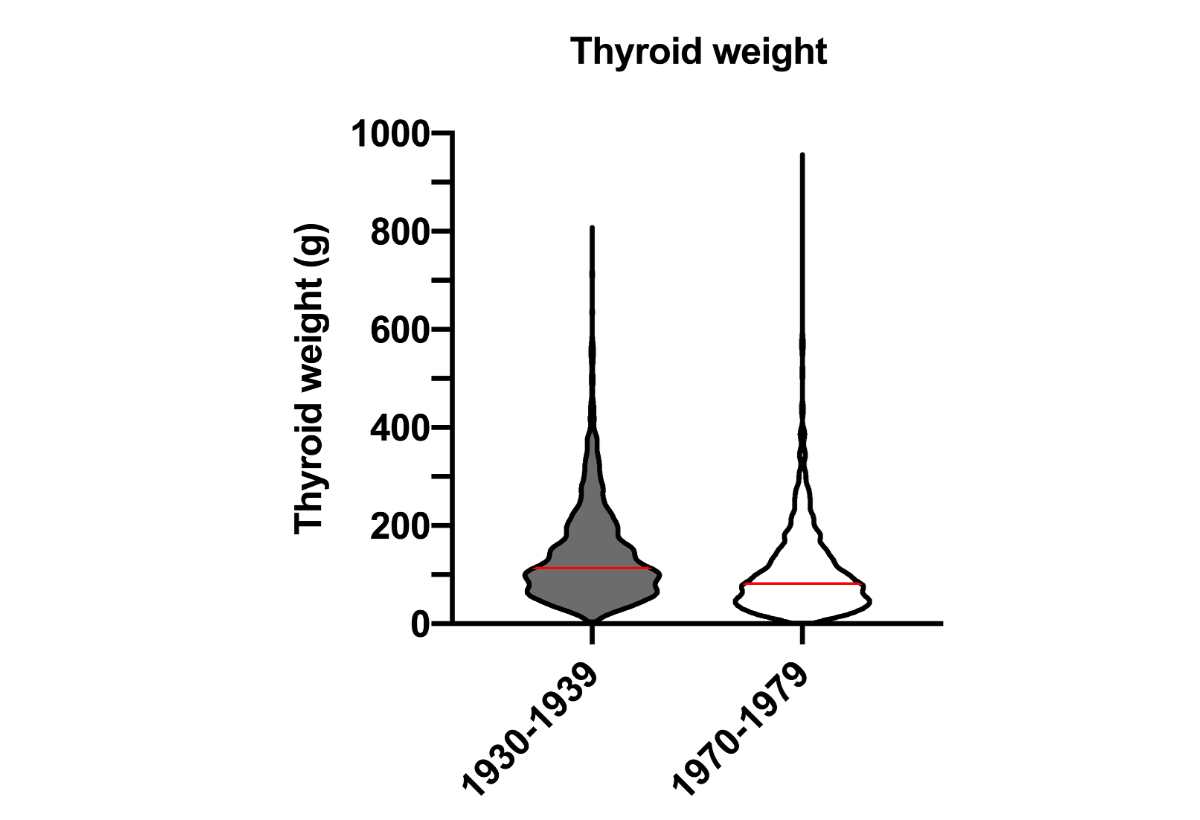
Figure 2Weight of resected thyroid gland in grams in 1930–1939 vs 1970–1979. The red line indicates the mean. 1930s cohort: n = 1826; mean weight: 141.2 g (range: 4–808). 1970s cohort: n = 1454, mean weight: 107 g (range: 1–956).
The mortality was 0.7% (12/1826 patients) in 1930–1939, compared to 0.2% (3/1454 patients) in 1970–1979. The rate of surgical complications such as recurrent laryngeal nerve injuries and Chvostek tetanies was significantly higher in the 1930s than in the 1970s (table 2).
Table 2Postoperative thyroid surgery complications in 1930–1939 vs 1970–1979.
| Postoperative complication | 1930–1939 (n = 1826) | 1970–1979 (n = 1454) | p | 95% CI |
| Mortality rate during hospitalisation, n (%) | 12 (0.7%) | 3 (0.2%) | 0.06 | 0.01–0.9 |
| Recurrent laryngeal nerve injuries, n (%) | 293 (16.0%) | 61 (4.2%) | <0.01 | 9.9–13.8 |
| Bilateral recurrent laryngeal nerve injuries, n (%) | 20 (1.1%) | 2 (0.1%) | <0.01 | 0.5–1.5 |
| Chvostek tetanies, n (%) | 112 (6.1%) | 15 (1.0%) | <0.01 | 3.9–6.3 |
Data are shown as count (percentage). Statistical test is chi-squared test. CI: confidence interval.
The DRG-based data of surgery and complication rates of the five largest Swiss endocrine surgery centres (2024) documented in EUROCRINE are summarised in table 3. There are similar rates of recurrent laryngeal nerve paralysis in the two databases, but EUROCRINE reports a smaller rate of persistent hypocalcaemia compared to the DRG-based data (1.3% vs 3.7%). Compared to the Riehen cohorts, there is mainly a significantly lower mortality rate.
Table 3DRG-based data of thyroid surgery complications in Switzerland 2011–2015: complications in high-volume institutions (>200 thyroidectomies per year).
| DRG | Number of Swiss centres, n | 5 |
| Number of patients, n | 5797 | |
| Recurrent laryngeal nerve paralysis, % | 2.1% | |
| Hypocalcaemia, % | 3.7% | |
| EUROCRINE | Number of Swiss centres (2024), n | 5 |
| Number of patients (2024), n | 1131 | |
| Recurrent laryngeal nerve paralysis, % (persistent) | 2.4% (2.9) | |
| Hypocalcaemia (persistent), % (persistent) | 1.3% (0.8) |
DRG = diagnosis-related groups
There was a significant positive correlation between thyroid weight and complications (recurrent laryngeal nerve injuries, Chvostek tetanies) in the 1970s (tables 4 and 5, figures 3 and 4). In the 1930s, there was only a significant positive association between thyroid weight and Chvostek tetanies, but not between thyroid weight and recurrent laryngeal nerve injuries. Recurrent thyroid surgeries were only significantly associated with recurrent laryngeal nerve injuries in the 1970s (tables 4 and 5). There was no significant correlation between recurrent thyroid surgeries and Chvostek tetanies when data were analysed separately for the 1930s and the 1970s.
Table 4Correlation analysis between recurrent thyroid surgery and complications (chi-squared test).
| n (%) | p | |
| Recurrent thyroid surgery and recurrent laryngeal nerve injuries in 1930s | 62 (0.2%) | 0.27 |
| Recurrent thyroid surgery and recurrent laryngeal nerve injuries in 1970s | 26 (0.4%) | 0.02 |
| Recurrent thyroid surgery and Chvostek tetanies in 1930s | 12 (0.1%) | 0.19 |
| Recurrent thyroid surgery and Chvostek tetanies in 1970s | 1 (0.1%) | 0.46 |
| Table 5. Incidence of complications according to resected thyroid weight categories (ANOVA). | ||||||
| ≤50 g | 50–100 g | 100–200 g | ≥200 g | Mean ± SD, (range) | p | |
| Recurrent laryngeal nerve injuries in 1930s, n (%) | 32 (14.8%) | 80 (14.3%) | 117 (17.3%) | 63 (17%) | 73 ± 35.43, (32–117) | 0.48 |
| Recurrent laryngeal nerve injuries in 1970s, n (%) | 5 (1.2%) | 23 (4.9%) | 21 (5.2%) | 12 (6.9%) | 15.2 ± 8.34, (5–23) | <0.01 |
| Chvostek tetanies in 1930s, n (%) | 9 (4.2%) | 84 (15%) | 80 (11.8%) | 42 (11.3%) | 53.7 ± 35.3, (9–84) | <0.01 |
| Chvostek tetanies in 1970s, n (%) | 2 (0.5%) | 8 (1.7%) | 14 (3.5%) | 6 (3.5%) | 7.5 ± 5, (2–14) | 0.01 |
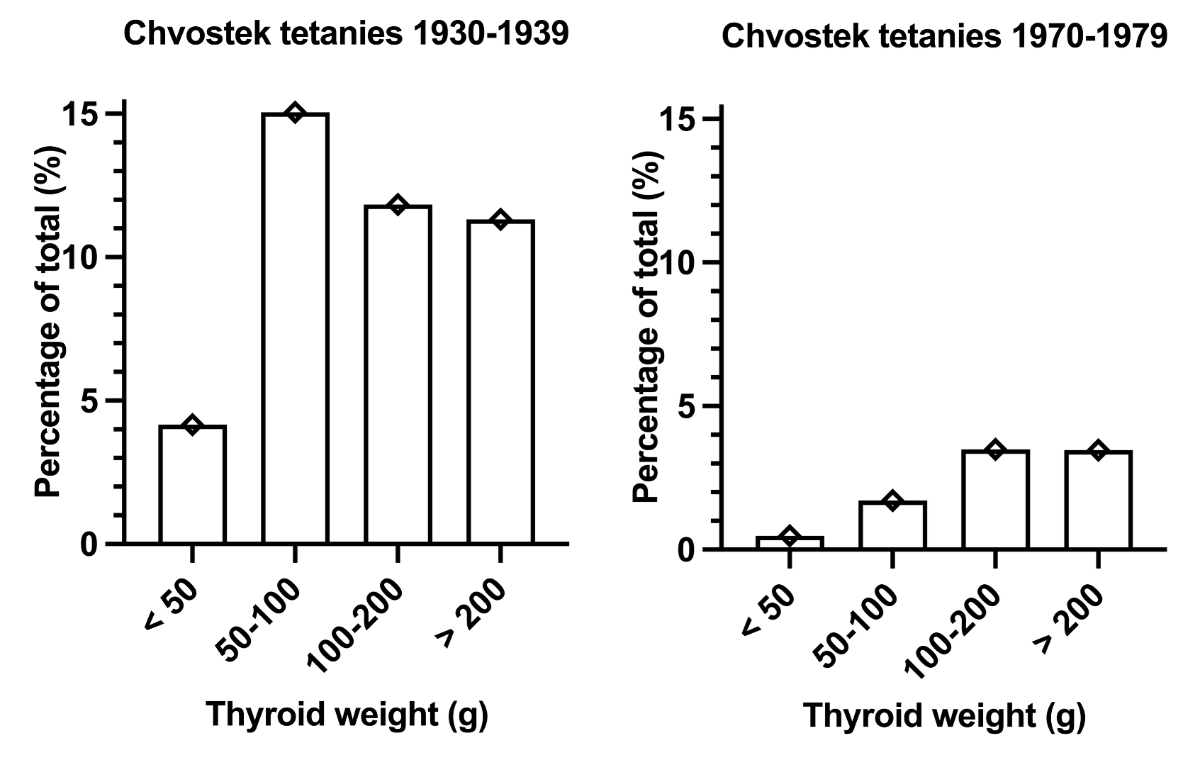
Figure 3Incidence of Chvostek tetanies according to resected thyroid weight in 1930–1939 vs 1970–1979.
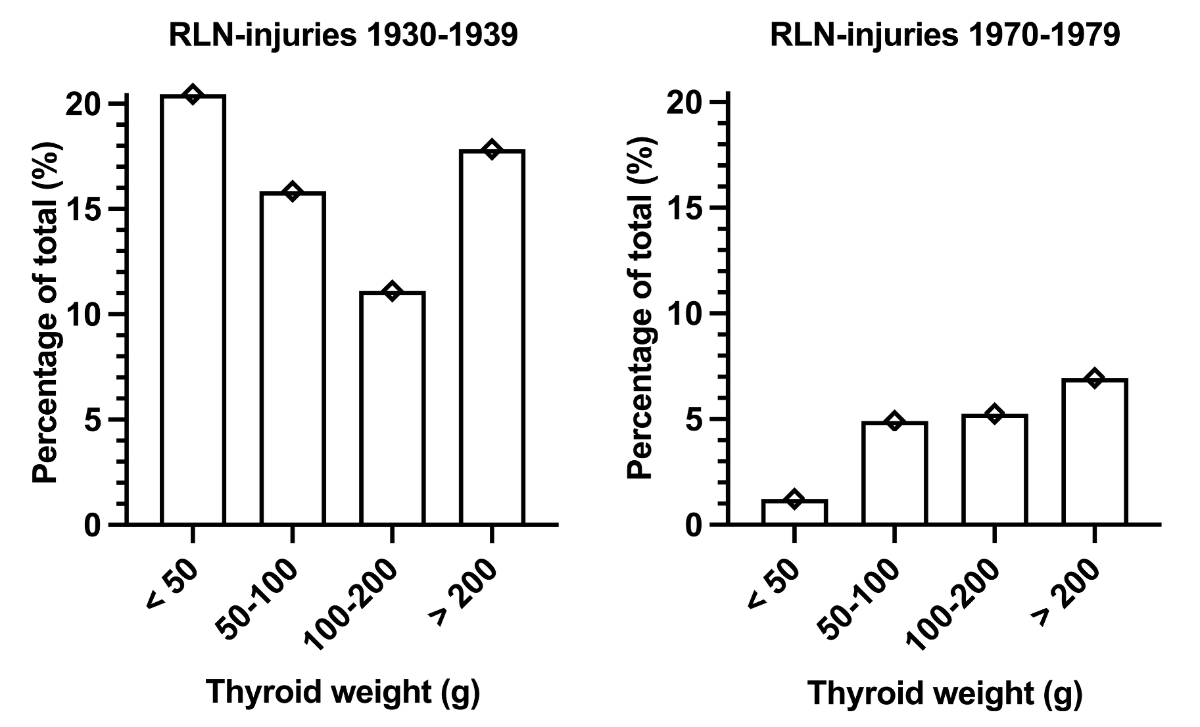
Figure 4Incidence of recurrent laryngeal nerve injuries (RLN-injuries) according to resected thyroid weight in 1930–1939 vs 1970–1979.
The significant decrease in the weight of the resected thyroid gland can be observed simultaneously with the decline in the prevalence of goitre in Switzerland due to salt iodisation starting in 1922 [7]. It is likely that the iodine supplementation led to a decrease in thyroid weight, including occurrence of intrathoracic goitre, which, in turn, may have resulted in fewer postoperative complications in the 1970s. Whether iodine supplementation explains the older age of the patients with goitre in the 1970s is conceivable but more hypothetical.
The carcinoma rate showed a significant increase over time but is considerably lower than in current autopsy data [36]. In this regard, consideration should be given to the younger age of the patients and the presence of symptoms in our data. In contrast, current autopsy data involve significantly older patients and carcinomas can often be macroscopically invisible or asymptomatic.
Currently, we would consider the hospital in Riehen (Switzerland) a high-volume hospital for thyroid surgery. It is established that in high-volume hospitals, fewer postoperative complications occur [8, 37, 38]. Consistent with this, the occurrence of postoperative symptomatic Chvostek tetany significantly decreased over time and showed a similar rate in the 1970s as described today in high-volume hospitals in Switzerland (table 3) and internationally [8–13, 15]. However, we must consider the fact that the clinical assessment of hypocalcaemia (Chvostek tetany) is significantly less sensitive than serum calcium concentration measurements as is performed nowadays as the gold standard [39, 40]. It is therefore likely that the reported hypocalcaemia-associated complication is underestimated in the Riehen cohorts, and the absolute complication rate was probably higher than in the current century.
The significant positive correlation between the resected thyroid weight and the number of Chvostek tetanies in our analysis indicates a conceivable association between thyroid size and postoperative hypocalcaemia. Consistently, more recent studies describe an increased risk of postoperative hypocalcaemia in patients with large goitres, where the parathyroid glands may lie beneath the thyroid capsule, or even be completely intrathyroidal and impossible to identify at surgery, leading to a higher rate of incidental parathyroidectomy [41, 42]. Furthermore, in patients with a large goitre or thyroid cancer requiring extensive nodal dissection, the lower parathyroid glands are often removed [41–43].
Recurrent thyroid surgery is an established risk factor for postoperative complications [8, 12, 32, 33, 38, 43, 44]. This can be confirmed in the current findings of the Riehen cohorts where an increased risk of Chvostek tetany can be documented in the setting of recurrent thyroid surgery. Consistently, in a more recent review of recurrent thyroid surgery, an increased rate of an incidental finding of parathyroid gland(s) was reported, suggesting an increased risk for postoperative hypocalcaemia [43]. The same review mentioned a higher risk of postoperative hypocalcaemia in the reoperation setting due to repeat cervical exploration in the presence of adhesions, which may result in an increased risk of devascularisation of the parathyroid glands [43].
There was a significant decrease in the thyroidectomy-induced incidence of recurrent laryngeal nerve injuries over time; however, it remains a relevant complication with a similar incidence in the 1970s as in the current literature [14, 15, 45, 46]. According to a systematic review, the incidence varies depending on the follow-up time postoperatively, with an incidence of 5–11% in the immediate postoperative period and persistence at one year after the operation in 1–3.5% of thyroid surgeries [14]. However, we have to take into account that the current standard for assessing an recurrent laryngeal nerve injury includes a systematic laryngoscopy by an ENT specialist before and after the intervention. A systematic laryngoscopy detects about double as many cases with recurrent laryngeal nerve injury compared to a clinical diagnosis [14]. It is therefore likely that the complication rate of recurrent laryngeal nerve injury of the Riehen cohorts reported here underestimates the real number.
A multicentric study and a systematic review identified reoperation as an important risk factor for recurrent laryngeal nerve injuries [38, 44]. A higher risk for recurrent laryngeal nerve injuries in reoperations is consistent with our results, where a significant positive correlation between recurrent thyroid surgeries and recurrent laryngeal nerve injuries was seen. There were notably fewer recurrent thyroid surgeries in the first half of the 1930s, which possibly explains the positive trend but lack of statistical significance in the 1930s. In addition, the risk of recurrent laryngeal nerve injuries depends on the extent of surgery [38] and the weight of the operated thyroid gland. In particular, a substernal location, where the nerve is stretched and more exposed to injury during surgery [44], is associated with a higher risk of recurrent laryngeal nerve injuries. These data are consistent with the current findings, where a significant positive correlation between recurrent laryngeal nerve injuries and thyroid weight could be documented. Furthermore, it is conceivable that the surgical volume of a given institution and the corresponding surgical experience [11, 31, 38], as well as formal identification of the recurrent laryngeal nerve intraoperatively and functional perioperative monitoring, are important factors in reducing the risk of recurrent laryngeal nerve injury [38]. In this context, it is worth mentioning that only four surgeons have ever performed thyroid surgery in Riehen. It is conceivable that the high level of experience gained over time explains the low complication rate.
The observed mortality rate in both decades of the Riehen cohorts was low: 0.7% in the 1930s and 0.2% in the 1970s. Interestingly, a similar rate had already been reported by Theodor Kocher in the mid-1880s [5]. The DRG-based current data, however, suggest an even lower mortality rate of 0.05%, which is confirmed by the EUROCRINE registry (0%). It is conceivable that improved perioperative management explains these results. Interestingly, the DRG-based data and the EUROCRINE data confirm a low and very similar rate for recurrent laryngeal nerve palsy (2–3%); however, the rate of permanent hypoparathyroidism is significantly higher in the DRG-based database compared to EUROCRINE. Most likely this is related to the fact that in the DRG-based data, transient hypocalcaemia is also included since the observation period of the DRG-based database is only 30 days compared to EUROCRINE where long-term observation is registered. But overall, thyroidectomy appears to be a safe operation.
The main strength of this study is the large sample size of well-reported and standardised health records over decades, which can be considered a reliable source of historical evidence. In addition, a selection bias is probably not a confounder since all patients with standardised reports were included in the study. We cannot exclude that data of some patients were not documented in the respective files. However, the absolute number of reported cases per decade in this study is high and Riehen was a well-known centre for thyroid surgery (high-volume hospital). This suggests that the reported data are valid and potential missing patients (information bias) would probably not change the main findings.
Nevertheless, several limitations must be considered when interpreting our results.
First, the main population of our study originates from Basel, Switzerland, and the surrounding regions, so the study’s results may not be generalisable to other regions.
Second, surgical competence may be a confounder. Although there were only four surgeons over time, the quality of these surgeons was probably not identical and may have changed with time and experience.
Third, the surgical technique including perioperative recurrent laryngeal nerve monitoring and the assessment of hypocalcaemia (from clinical [Chvostek tetany] to standardised biochemical analysis of calcium levels) has changed. It is therefore likely that the rate of hypocalcaemia is still underestimated in the Riehen cohorts compared to the recent DRG and EUROCRINE data.
Lastly, the current health records indicate only complications during the hospitalisation. Long-term follow-up data of these cohorts are not available.
The large sample size of well-reported and standardised health records over time allows a coherent analysis over time. Given that Riehen and northwest Switzerland was an area endemic for goitre, many thyroid surgeries could be observed over time. Since the hospital in Riehen can be considered a high-volume institute for thyroid surgery, data can be compared with current data of Swiss high-volume hospitals.
The reduction in primary complications over time in the Riehen cohorts following thyroidectomy, namely recurrent laryngeal nerve injury and Chvostek tetany, is likely a result of multiple factors. The introduction of iodine supplementation, coupled with a decrease in goitre size, is probably one of the contributing factors. Whether surgical experience and technique as well as changes in perioperative management contribute to these findings remains speculative. The mortality rate after thyroidectomy in Riehen was low in the previous century and similar to that reported by Theodor Kocher [5]. In the current century, there is an extremely low mortality rate reported for this intervention, suggesting that thyroidectomy is a safe procedure. Due to the different methods used to assess the complication rate (mainly clinical in the Riehen cohort vs systematic laryngoscopy and calcium measurements in the current century), it is likely that the complication rates in the Riehen cohorts are underestimated.
The datasets analysed during this study are not publicly available due to patient confidentiality and the absence of informed consent for data sharing. Data are available from the corresponding author on reasonable request and subject to approval by the relevant ethics committee and applicable data protection regulations.
We thank Andrea Goldmann, Reto Kaderli, Frederic Triponez, Thomas Clerici, Stefanie Taha and Beatrice Kern for providing us with data from EUROCRINE.
Author contributions: AA conceived the study, performed data collection and data analysis, and wrote the paper. MM performed data collection. FG provided data. KK provided data. NP advised on data collection and data interpretation. EF provided data and proofread. KA provided data and proofread. SG conceived and mentored the entire study. CE conceived and mentored the entire study.
This research did not receive any specific grant from any funding agency in the public or commercial sector. The costs for the secuTrial database were covered by the Gemeinde Riehen.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflict of interest related to the content of this manuscript was disclosed.
1. Dorairajan N, Pradeep PV. Vignette thyroid surgery: a glimpse into its history. Int Surg. 2013;98(1):70–5. doi: https://doi.org/10.9738/CC185.1
2. Dadan J, Nowacka A. A journey into the past—the history of thyroid surgery. Wiad Lek. 2008;61(1-3):88–92.
3. Harvey SC. The History of Hemostasis. Ann Med Hist. 1929 Mar;1(2):127–54.
4. Giddings, A.E., The history of thyroidectomy. J R Soc Med, 1998. 91 Suppl 33(Suppl 33): p. 3-6.
5. Becker WF. Presidential address: pioneers in thyroid surgery. Ann Surg. 1977 May;185(5):493–504. doi: https://doi.org/10.1097/00000658-197705000-00001
6. Theodor K. Kropfextirpation und ihre Folgen. Archiv für klinische. Chirurgie. 1883;29:254.
7. Bürgi H, Supersaxo Z, Selz B. Iodine deficiency diseases in Switzerland one hundred years after Theodor Kocher’s survey: a historical review with some new goitre prevalence data. Acta Endocrinol (Copenh). 1990 Dec;123(6):577–90. doi: https://doi.org/10.1530/acta.0.1230577
8. Thomusch O, Machens A, Sekulla C, Ukkat J, Lippert H, Gastinger I, et al. Multivariate analysis of risk factors for postoperative complications in benign goiter surgery: prospective multicenter study in Germany. World J Surg. 2000 Nov;24(11):1335–41. doi: https://doi.org/10.1007/s002680010221
9. Efremidou EI, Papageorgiou MS, Liratzopoulos N, Manolas KJ. The efficacy and safety of total thyroidectomy in the management of benign thyroid disease: a review of 932 cases. Can J Surg. 2009 Feb;52(1):39–44.
10. Bellantone R, Lombardi CP, Bossola M, Boscherini M, De Crea C, Alesina P, et al. Total thyroidectomy for management of benign thyroid disease: review of 526 cases. World J Surg. 2002 Dec;26(12):1468–71. doi: https://doi.org/10.1007/s00268-002-6426-1
11. Duclos A, Peix JL, Colin C, Kraimps JL, Menegaux F, Pattou F, et al.; CATHY Study Group. Influence of experience on performance of individual surgeons in thyroid surgery: prospective cross sectional multicentre study. BMJ. 2012 Jan;344 jan10 2:d8041. doi: https://doi.org/10.1136/bmj.d8041
12. Rosato L, Avenia N, Bernante P, De Palma M, Gulino G, Nasi PG, et al. Complications of thyroid surgery: analysis of a multicentric study on 14,934 patients operated on in Italy over 5 years. World J Surg. 2004 Mar;28(3):271–6. doi: https://doi.org/10.1007/s00268-003-6903-1
13. Zambudio AR, Rodríguez J, Riquelme J, Soria T, Canteras M, Parrilla P. Prospective study of postoperative complications after total thyroidectomy for multinodular goiters by surgeons with experience in endocrine surgery. Ann Surg. 2004 Jul;240(1):18–25. doi: https://doi.org/10.1097/01.sla.0000129357.58265.3c
14. Jeannon JP, Orabi AA, Bruch GA, Abdalsalam HA, Simo R. Diagnosis of recurrent laryngeal nerve palsy after thyroidectomy: a systematic review. Int J Clin Pract. 2009 Apr;63(4):624–9. doi: https://doi.org/10.1111/j.1742-1241.2008.01875.x
15. Lukinović J, Bilić M. Overview of Thyroid Surgery Complications. Acta Clin Croat. 2020 Jun;59 Suppl 1:81–6.
16. Borel F, Christou N, Marret O, Mathonnet M, Caillard C, Bannani S, et al. Long-term voice quality outcomes after total thyroidectomy: a prospective multicenter study. Surgery. 2018 Apr;163(4):796–800. doi: https://doi.org/10.1016/j.surg.2017.09.023
17. Lombardi CP, Raffaelli M, De Crea C, D’Alatri L, Maccora D, Marchese MR, et al. Long-term outcome of functional post-thyroidectomy voice and swallowing symptoms. Surgery. 2009 Dec;146(6):1174–81. doi: https://doi.org/10.1016/j.surg.2009.09.010
18. Hillenbrand A, Cammerer G, Dankesreiter L, Lemke J, Henne-Bruns D. Postoperative swallowing disorder after thyroid and parathyroid resection. Pragmat Obs Res. 2018 Oct;9:63–8. doi: https://doi.org/10.2147/POR.S172059
19. Lacoste L, Gineste D, Karayan J, Montaz N, Lehuede MS, Girault M, et al. Airway complications in thyroid surgery. Ann Otol Rhinol Laryngol. 1993 Jun;102(6):441–6. doi: https://doi.org/10.1177/000348949310200607
20. Wilson JA, Deary IJ, Millar A, Mackenzie K. The quality of life impact of dysphonia. Clin Otolaryngol Allied Sci. 2002 Jun;27(3):179–82. doi: https://doi.org/10.1046/j.1365-2273.2002.00559.x
21. Jones SM, Carding PN, Drinnan MJ. Exploring the relationship between severity of dysphonia and voice-related quality of life. Clin Otolaryngol. 2006 Oct;31(5):411–7. doi: https://doi.org/10.1111/j.1749-4486.2006.01291.x
22. Krekeler BN, Wendt E, Macdonald C, Orne J, Francis DO, Sippel R, et al. Patient-Reported Dysphagia After Thyroidectomy: A Qualitative Study. JAMA Otolaryngol Head Neck Surg. 2018 Apr;144(4):342–8. doi: https://doi.org/10.1001/jamaoto.2017.3378
23. Dawale K, Agrawal A. Parathyroid Hormone Secretion and Related Syndromes. Cureus. 2022 Oct;14(10):e30251.
24. Cusano NE, Bilezikian JP. Signs and Symptoms of Hypoparathyroidism. Endocrinol Metab Clin North Am. 2018 Dec;47(4):759–70. doi: https://doi.org/10.1016/j.ecl.2018.07.001
25. Yao L, Hui X, Li M, Li J, Ahmed MM, Lin C, et al. Complications, Symptoms, Presurgical Predictors in Patients With Chronic Hypoparathyroidism: A Systematic Review. J Bone Miner Res. 2022 Dec;37(12):2642–53. doi: https://doi.org/10.1002/jbmr.4673
26. Underbjerg L, Sikjaer T, Mosekilde L, Rejnmark L. Cardiovascular and renal complications to postsurgical hypoparathyroidism: a Danish nationwide controlled historic follow-up study. J Bone Miner Res. 2013 Nov;28(11):2277–85. doi: https://doi.org/10.1002/jbmr.1979
27. Almquist M, Ivarsson K, Nordenström E, Bergenfelz A. Mortality in patients with permanent hypoparathyroidism after total thyroidectomy. Br J Surg. 2018 Sep;105(10):1313–8. doi: https://doi.org/10.1002/bjs.10843
28. Bergenfelz A, Nordenström E, Almquist M. Morbidity in patients with permanent hypoparathyroidism after total thyroidectomy. Surgery. 2020 Jan;167(1):124–8. doi: https://doi.org/10.1016/j.surg.2019.06.056
29. Büttner M, Locati LD, Pinto M, Araújo C, Tomaszewska IM, Kiyota N, et al. Quality of Life in Patients With Hypoparathyroidism After Treatment for Thyroid Cancer. J Clin Endocrinol Metab. 2020 Dec;105(12):dgaa597. doi: https://doi.org/10.1210/clinem/dgaa597
30. Hadker N, Egan J, Sanders J, Lagast H, Clarke BL. Understanding the burden of illness associated with hypoparathyroidism reported among patients in the PARADOX study. Endocr Pract. 2014 Jul;20(7):671–9. doi: https://doi.org/10.4158/EP13328.OR
31. Stavrakis AI, Ituarte PH, Ko CY, Yeh MW. Surgeon volume as a predictor of outcomes in inpatient and outpatient endocrine surgery. Surgery. 2007 Dec;142(6):887–99. doi: https://doi.org/10.1016/j.surg.2007.09.003
32. Lefevre JH, Tresallet C, Leenhardt L, Jublanc C, Chigot JP, Menegaux F. Reoperative surgery for thyroid disease. Langenbecks Arch Surg. 2007 Nov;392(6):685–91. doi: https://doi.org/10.1007/s00423-007-0201-6
33. Erbil Y, Barbaros U, Işsever H, Borucu I, Salmaslioğlu A, Mete O, et al. Predictive factors for recurrent laryngeal nerve palsy and hypoparathyroidism after thyroid surgery. Clin Otolaryngol. 2007 Feb;32(1):32–7. doi: https://doi.org/10.1111/j.1365-2273.2007.01383.x
34. Simó R, Nixon IJ, Vander Poorten V, Quer M, Shaha AR, Sanabria A, et al. Surgical management of intrathoracic goitres. Eur Arch Oto-Rhino-Laryngol. 2019 Feb;276(2):305–14. doi: https://doi.org/10.1007/s00405-018-5213-z
35. Goldfarb M, Perry Z, A Hodin R, Parangi S. Medical and surgical risks in thyroid surgery: lessons from the NSQIP. Ann Surg Oncol. 2011 Dec;18(13):3551–8. doi: https://doi.org/10.1245/s10434-011-1938-2
36. Mohorea IS, Socea B, Şerban D, Ceausu Z, Tulin A, Melinte V, et al. Incidence of thyroid carcinomas in an extended retrospective study of 526 autopsies. Exp Ther Med. 2021 Jun;21(6):607. doi: https://doi.org/10.3892/etm.2021.10039
37. Liang TJ, Liu SI, Mok KT, Shi HY. Associations of Volume and Thyroidectomy Outcomes: A Nationwide Study with Systematic Review and Meta-Analysis. Otolaryngol Head Neck Surg. 2016 Jul;155(1):65–75. doi: https://doi.org/10.1177/0194599816634627
38. Dralle H, Sekulla C, Haerting J, Timmermann W, Neumann HJ, Kruse E, et al. Risk factors of paralysis and functional outcome after recurrent laryngeal nerve monitoring in thyroid surgery. Surgery. 2004 Dec;136(6):1310–22. doi: https://doi.org/10.1016/j.surg.2004.07.018
39. Sorell M, Rosen JF. Ionized calcium: serum levels during symptomatic hypocalcemia. J Pediatr. 1975 Jul;87(1):67–70. doi: https://doi.org/10.1016/S0022-3476(75)80070-9
40. Pepe J, Colangelo L, Biamonte F, Sonato C, Danese VC, Cecchetti V, et al. Diagnosis and management of hypocalcemia. Endocrine. 2020 Sep;69(3):485–95. doi: https://doi.org/10.1007/s12020-020-02324-2
41. Sakorafas GH, Stafyla V, Bramis C, Kotsifopoulos N, Kolettis T, Kassaras G. Incidental parathyroidectomy during thyroid surgery: an underappreciated complication of thyroidectomy. World J Surg. 2005 Dec;29(12):1539–43. doi: https://doi.org/10.1007/s00268-005-0032-y
42. Gourgiotis S, Moustafellos P, Dimopoulos N, Papaxoinis G, Baratsis S, Hadjiyannakis E. Inadvertent parathyroidectomy during thyroid surgery: the incidence of a complication of thyroidectomy. Langenbecks Arch Surg. 2006 Nov;391(6):557–60. doi: https://doi.org/10.1007/s00423-006-0079-8
43. Lin DT, Patel SG, Shaha AR, Singh B, Shah JP. Incidence of inadvertent parathyroid removal during thyroidectomy. Laryngoscope. 2002 Apr;112(4):608–11. doi: https://doi.org/10.1097/00005537-200204000-00003
44. Hartl DM, Travagli JP, Leboulleux S, Baudin E, Brasnu DF, Schlumberger M. Clinical review: current concepts in the management of unilateral recurrent laryngeal nerve paralysis after thyroid surgery. J Clin Endocrinol Metab. 2005 May;90(5):3084–8. doi: https://doi.org/10.1210/jc.2004-2533
45. Bergenfelz A, Jansson S, Kristoffersson A, Mårtensson H, Reihnér E, Wallin G, et al. Complications to thyroid surgery: results as reported in a database from a multicenter audit comprising 3,660 patients. Langenbecks Arch Surg. 2008 Sep;393(5):667–73. doi: https://doi.org/10.1007/s00423-008-0366-7
46. Sanapala A, Nagaraju M, Rao LN, Nalluri K. Management of bilateral recurrent laryngeal nerve paresis after thyroidectomy. Anesth Essays Res. 2015;9(2):251–3. doi: https://doi.org/10.4103/0259-1162.152419