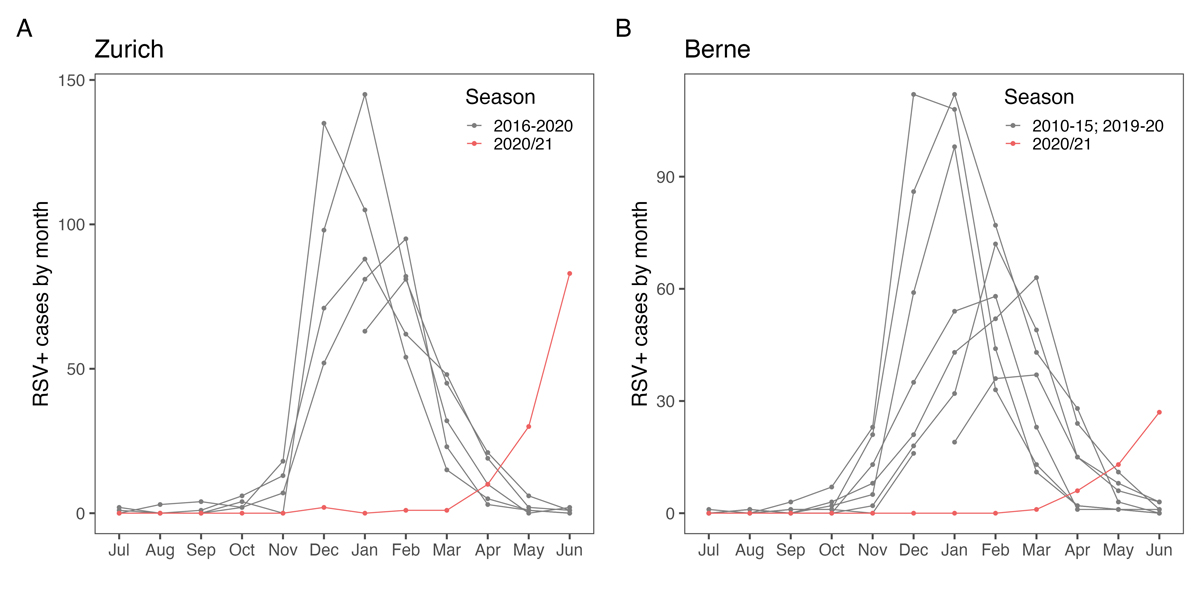
Figure 1 Comparison of RSV case numbers before and during COVID-19 pandemic in (A) Zurich and (B) Berne.
DOI: https://doi.org/10.4414/SMW.2021.w30057
Respiratory syncytial virus (RSV) is a major cause of respiratory tract infection in children under 5 years of age worldwide, with a substantial clinical burden [1, 2]. Before the onset of the COVID-19 pandemic, typical seasonality was reported for regions with temperate climates, with RSV infections peaking during the autumn and winter seasons [3]. However, since the onset of the COVID-19 pandemic, reports from Australia and the US, and more recently from other European countries and Israel, have provided evidence of altered RSV epidemiology, with low activity during the typical season and an interseasonal outbreak [4–10]. Although RSV is not yet listed as a notifiable disease in the European Union [11, 12], the European Centre for Disease Prevention and Control (ECDC) publishes data from several European countries [13] and urges systematic real-time reporting to provide data for relevant decision making on preventive measures [12]. In Switzerland, no national real-time surveillance system for RSV has been established to date. Here, we describe RSV EpiCH, a multicentre database managed from paediatric hospitals across Switzerland that has continuously collected and disseminated data on RSV infections in children since January 2021.
Alerted by reports of exceptional RSV activity during the summer 2020/21 in New South Wales, Australia [5], and first cases of RSV infections in Swiss centres, we contacted 20 of 29 paediatric acute care hospitals in Switzerland in April 2021. Members of the "Paediatric infectious disease group Switzerland" at each site were informed about RSV EpiCH and were invited to provide their local data to this central database on a weekly basis. No ethical consent was required as only aggregated and anonymised data were processed. The 20 participating centres include all 7 tertiary care hospitals (including all 5 paediatric university hospitals), 10 of 13 secondary care regional hospitals, and 3 of 9 primary care local hospitals (table 1). Centres were given individual access to a REDCap database and provided weekly aggregated numbers of children with a positive RSV detection, divided into three age groups (infants, 1–2 years old, and older than 2 years), with no missing data for these values. For most hospitals, we also captured the number of total tests performed per week (not split into age groups). The complete data processing workflow from downloading the data from the REDCap database using the REDCap application programming interface to generating tables and graphics for public dissemination was coded in R [14].
For each hospital, we recorded which patient populations were generally tested (inpatient versus outpatient versus both), the testing strategy (untargeted, i.e., all children with fever or respiratory symptoms, versus targeted testing, i.e., only infants with bronchiolitis), and the type of test used (table 1). We retrospectively collected data from January 2021.
Table 1Description of centres and testing modalities.
| Abbr. | Institution | Type | Testing strategy | Targeted / untargeted | Tests used |
| AG | Cantonal Hospital Aarau | Regional hospital | April–September: in- and outpatient. October–March: inpatient only | April–September: targeted. October–March: untargeted | PCR, Antigen |
| AG | Cantonal Hospital of Baden | Regional hospital | In- and outpatient | Targeted | PCR, Antigen |
| BS | University Children’s Hospital Basel | Tertiary care hospital | In- and outpatient | Untargeted | PCR |
| BE | University Hospital Bern | Tertiary care hospital | Inpatient only | Untargeted | Antigen |
| BE | Spitalzentrum Biel | Regional hospital | Inpatient only | Untargeted | PCR |
| FR | Fribourg Hospital HFR | Regional hospital | Inpatient only | Targeted | PCR |
| GE | Geneva University Hospital | Tertiary care hospital | Inpatient only | Targeted | Antigen |
| GR | Cantonal Hospital of Graubünden | Regional hospital | Inpatient only | Untargeted | PCR |
| KSM | Cantonal Hospital Münsterlingen | Regional hospital | Inpatient only | Untargeted | PCR |
| KSW | Cantonal hospital of Winterthur | Regional hospital | Inpatient only | Targeted | PCR |
| LU | Children’s Hospital Lucerne, Cantonal Hospital of Lucerne | Tertiary care hospital | Inpatient only | Untargeted | PCR |
| Targeted | Antigen | ||||
| NE | Réseau hospitalier neuchâtelois | Regional hospital | Inpatient only | Targeted | PCR |
| SG | Cantonal Hospital of St. Gallen | Tertiary care hospital | Inpatient only | Targeted | PCR |
| TI | Istituto Pediatrico della Svizzera Italiana (Locarno, Bellinzona, Lugano, Mendrisio), Ente Ospedaliero Cantonale | One Regional hospital, two local hospitals | Inpatient only, occasionally outpatient | Targeted | PCR, Antigen |
| VD | Lausanne University Hospital | Tertiary care hospital | Inpatient only | Targeted | PCR |
| ZH | University Children’s Hospital Zurich | Tertiary care hospital | Inpatient only | Untargeted | PCR |
| ZH | Triemli Hospital Zurich | Regional hospital | In- and outpatient | Targeted | Antigen |
PCR = polymerase chain reaction; Antigen = RSV antigen test
Prior to the COVID-19 pandemic, most children with RSV infections in Swiss hospitals were diagnosed between November and April, with peak infections occurring between December and February [15] (fig. 1).

Figure 1 Comparison of RSV case numbers before and during COVID-19 pandemic in (A) Zurich and (B) Berne.
In contrast, during the 2020/2021 winter season, virtually no RSV infections were observed in hospitals participating in RSV EpiCH (n = 12, January and February 2021). However, RSV case numbers increased steadily from April 2021 onward, coinciding with a decline in SARS-CoV-2 case numbers and an associated relaxation of non-pharmacological interventions in Switzerland (fig. 2).

Figure 2 Weekly number of RSV infections in children in Swiss hospitals from 1 January to 4 July 2021, stratified by patient age. The black horizontal bars on top of the plot indicate the main non-pharmaceutical interventions in Switzerland, with increasing bar height indicating stronger restrictions:
General recommendations
1. Public hygiene and social distancing >1.5 m (since 2 March 2020). Mandatory face masks in public transport for persons older than 10–12 years old (depending on region, since 6 July 2020) and in publicly accessible indoor venues (since 19 October 2020).
Restrictions on private life; maximum number of people allowed to meet privately indoors
2. 30 persons (since 31 May 2021)
3. 15 persons (19 Apr to 30 May 2021)
4. 10 persons (29 Oct 2020 to 17 Jan 2021; 22 Mar to 18 Apr 2021)
5. 5 persons (18 Jan - 21 Mar 2021)
Restrictions on public life
6. Closure of restaurants and bars (22 Dec 2020 to 30 May 2021)
7. Cultural venues closed (22 Dec 2020 to 18 Apr 2021)
8. Non-essential shops closed (18 Jan to 28 Feb 2021)
The large number of RSV infections detected in children in May (337 cases) and June 2021 (425 cases) are well outside the normal range for spring and summer months in Switzerland (fig. 1). Interestingly, we observed regional differences, with more cases detected in larger cities and Western Switzerland affected earlier than the rest of Switzerland (fig. 3).
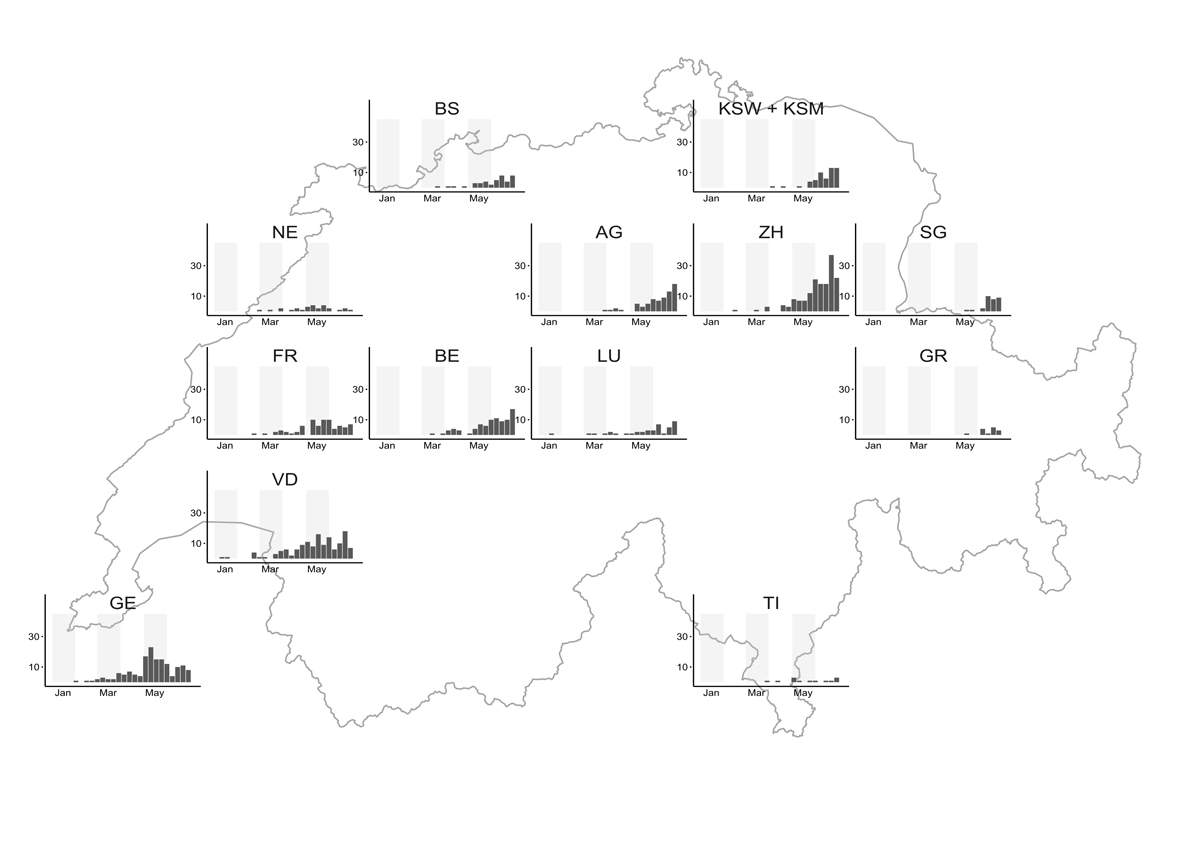
Figure 3 Regional distribution of the weekly number of RSV infections in children in Switzerland from 1 January to 4 July 2021 (calendar weeks 126).
AG = Cantonal Hospital Aarau and Cantonal Hospital of Baden; BS = University Children’s Hospital Basel; BE = University Hospital Bern and Spitalzentrum Biel; FR = Fribourg Hospital HFR; GE = Geneva University Hospital; GR = Cantonal Hospital of Graubünden; KSW + KSM = Cantonal Hospital of Winterthur and Cantonal Hospital Münsterlingen; LU = Lucerne Children’s Hospital, Cantonal Hospital of Lucerne; NE = Réseau hospitalier neuchâtelois; SG = Cantonal Hospital of St Gallen; TI = Istituto Pediatrico della Svizzera Italiana (Locarno, Bellinzona, Lugano, Mendrisio), Ente Ospedaliero Cantonale; VD = Lausanne University Hospital; ZH = University Children’s hospital Zurich and Triemli Hospital Zurich.
Overall, 54% of cases were observed in infants, 24% in children aged 1 to 2 years, and 23% in children older than 2 years (fig. 2).
An interseasonal increase in RSV infections has already been reported for the autumn of 2020 in New South Wales, Australia [5], and spring 2021 in New York, USA [4], coinciding with a local decline in COVID-19 case numbers and a relaxation of non-pharmacological interventions . Recently published data describe RSV activity delayed by about 2–3 months in France and Iceland, whereas in the Netherlands and the United Kingdom the onset of the RSV epidemic was not visible until after calendar week 20, about 6 months late [6, 10]. It is noteworthy that in France and Iceland only short or no school closures were enforced. In contrast, longer school closures were imposed in the Netherlands and the UK. As in France and Iceland, schools and day-care centres in Switzerland were not closed in the autumn of 2020 and winter of 2020/2021, and children younger than 10–12 years (depending on the region) did not wear masks at school. This contrasts with adolescents and adults, who had significant restrictions and thus marked changes in their contacts, indicating an important role of adolescents and adults as RSV reservoirs and for the spread of RSV in a community (fig. 2). The widespread implementation of non-pharmacological interventions can be considered the main reason for the low RSV transmission in the European winter [4, 5, 7].
In a study published in December 2020, Baker et. al predicted altered RSV epidemiology after long periods of non-pharmacological interventions, based on epidemiological models using data from previous RSV seasons in Texas and Florida [16]. In their analysis, an accumulation of RSV-susceptible individuals after a "missed" RSV season may lead to an unusually high number of RSV infections in the subsequent season (2021/2022). However, the impact of the interseasonal increase in RSV infections now observed in some regions on future epidemiology remains unclear.
Although several vaccines and antiviral therapies are in active development, only palivizumab, a monoclonal antibody against the RSV fusion protein, is currently available for prophylaxis in high-risk patients (e.g., infants with bronchopulmonary dysplasia or congenital cyanotic heart disease) [17]. Palivizumab is usually administered at monthly intervals during the 5–6 months of the peak RSV season. In some regions the recent changes in RSV epidemiology have meant that the prophylaxis administered during the winter months of 2020/21 was of no clinical benefit, which raises questions of whether and when additional doses of palivizumab should be offered to high-risk patients [18]. Because the complete absence of the RSV epidemic could not be predicted with certainty for the winter of 2020/2021, high-risk patients in Switzerland continued to receive palivizumab prophylaxis for the usual 5–6 “RSV months” as reimbursed by insurance.
In preparation for a possible interseasonal RSV epidemic, RSV EpiCH study members approached the authorities in May 2021 and succeeded in having the usual 6-month limit for reimbursements lifted. By calendar week 27, several centres of the RSV EpiCH network had already recommended additional doses of palivizumab for at risk patients.
To raise awareness of RSV amongst clinicians and support primary care paediatricians in their decision to offer palivizumab to patients, we reported our data in a weekly newsletter of the Swiss Paediatric Society distributed to over 3000 healthcare professionals in Switzerland. RSV EpiCH data were also updated weekly on the society website [19].
We report a shift in the usual RSV epidemiology in infants and children in Switzerland in 2021, coinciding with a relaxation of non-pharmacological interventions due to the COVID-19 pandemic. A rapidly established clinician-led initiative to collect information on RSV cases in Swiss hospitals proved successful in demonstrating regional and national dynamics of RSV infection in Switzerland. Although regional differences may be partly explained by differences in awareness and testing strategies among participating hospitals, RSV EpiCH data have been instrumental for communication with authorities and insurers, may prove useful in raising awareness of RSV infection, and provide early information to guide national policy on RSV prevention and distribution of healthcare resources in paediatric hospitals. The long-term sustainability of such a clinician-led initiative requires additional support, and we are seeking discussions with the relevant authorities with the aim of introducing mandatory reporting of RSV detection by all laboratories in Switzerland.
No financial support and no other potential conflict of interest relevant to this article was reported.
1. Hall CB , Weinberg GA , Iwane MK , Blumkin AK , Edwards KM , Staat MA , et al. The burden of respiratory syncytial virus infection in young children. N Engl J Med. 2009 Feb;360(6):588–98. https://doi.org/10.1056/NEJMoa0804877
2. Shi T , McAllister DA , O’Brien KL , Simoes EA , Madhi SA , Gessner BD , et al.; RSV Global Epidemiology Network . Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in young children in 2015: a systematic review and modelling study. Lancet. 2017 Sep;390(10098):946–58. https://doi.org/10.1016/S0140-6736(17)30938-8
3. Chadha M , Hirve S , Bancej C , Barr I , Baumeister E , Caetano B , et al.; WHO RSV Surveillance Group . Human respiratory syncytial virus and influenza seasonality patterns-Early findings from the WHO global respiratory syncytial virus surveillance. Influenza Other Respir Viruses. 2020 Nov;14(6):638–46. https://doi.org/10.1111/irv.12726
4. Agha R , Avner JR . Delayed Seasonal RSV Surge Observed During the COVID-19 Pandemic. Pediatrics. 2021 Sep;148(3):e2021052089. https://doi.org/10.1542/peds.2021-052089
5. Foley DA , Yeoh DK , Minney-Smith CA , Martin AC , Mace AO , Sikazwe CT , et al. The Interseasonal Resurgence of Respiratory Syncytial Virus in Australian Children Following the Reduction of Coronavirus Disease 2019-Related Public Health Measures. Clin Infect Dis. 2021 Feb;ciaa1906. https://doi.org/10.1093/cid/ciaa1906
6. van Summeren J , Meijer A , Aspelund G , Casalegno JS , Erna G , Hoang U , et al.; VRS study group in Lyon . Low levels of respiratory syncytial virus activity in Europe during the 2020/21 season: what can we expect in the coming summer and autumn/winter? Euro Surveill. 2021 Jul;26(29). https://doi.org/10.2807/1560-7917.ES.2021.26.29.2100639
7. Van Brusselen D , De Troeyer K , Ter Haar E , Vander Auwera A , Poschet K , Van Nuijs S , et al. Bronchiolitis in COVID-19 times: a nearly absent disease? Eur J Pediatr. 2021 Jun;180(6):1969–73. https://doi.org/10.1007/s00431-021-03968-6
8. Casalegno JS , Ploin D , Cantais A , Masson E , Bard E , Valette M , et al.; VRS study group in Lyon . Characteristics of the delayed respiratory syncytial virus epidemic, 2020/2021, Rhône Loire, France. Euro Surveill. 2021 Jul;26(29). https://doi.org/10.2807/1560-7917.ES.2021.26.29.2100630
9. Weinberger Opek M , Yeshayahu Y , Glatman-Freedman A , Kaufman Z , Sorek N , Brosh-Nissimov T . Delayed respiratory syncytial virus epidemic in children after relaxation of COVID-19 physical distancing measures, Ashdod, Israel, 2021. Euro Surveill. 2021 Jul;26(29). https://doi.org/10.2807/1560-7917.ES.2021.26.29.2100706
10. England PH . National flu and COVID-19 surveillance reports: 2021 to 2022 season. 2021 26th of August 2021 [cited 2021 31st of August]; Available from: https://www.gov.uk/government/statistics/national-flu-and-covid-19-surveillance-reports-2021-to-2022-season
11. Mollers M , Barnadas C , Broberg EK , Penttinen P , Teirlinck AC , Fischer TK ; European Influenza Surveillance Network . Current practices for respiratory syncytial virus surveillance across the EU/EEA Member States, 2017. Euro Surveill. 2019 Oct;24(40). https://doi.org/10.2807/1560-7917.ES.2019.24.40.1900157
12. Teirlinck AC , Broberg EK , Berg AS , Campbell H , Reeves RM , Carnahan A , et al. Recommendations for respiratory syncytial virus surveillance at national level. Eur Respir J. 2021 Apr;2003766. https://doi.org/10.1183/13993003.03766-2020
13. ECDC . Surveillance Atlas of Infectious Disease. 2021 [cited 2021 15th July]; Dataset provided by ECDC based on data provided by WHO and Ministries of Health from the affected countries]. Available from: https://atlas.ecdc.europa.eu/public/index.aspx
14. Team RC . R: A language and environment for statistical computing. 2021, R Foundation for Statistical Computing.
15. Duppenthaler A , Gorgievski-Hrisoho M , Frey U , Aebi C . Two-year periodicity of respiratory syncytial virus epidemics in Switzerland. Infection. 2003 Mar;31(2):75–80. https://doi.org/10.1007/s15010-002-3124-8
16. Baker RE , Park SW , Yang W , Vecchi GA , Metcalf CJ , Grenfell BT . The impact of COVID-19 nonpharmaceutical interventions on the future dynamics of endemic infections. Proc Natl Acad Sci USA. 2020 Dec;117(48):30547–53. https://doi.org/10.1073/pnas.2013182117
17. Agyeman P . Barazzone-Argiroffo C, Hammer J, Heininger U, Nadal D, Pfammatter J, et al. Konsensus Statement zur Prävention von Respiratory Syncytial Virus (RSV)-Infektionen mit dem humanisierten monoklonalen Antikörper Palivizumab (Synagis): Update 2016. Paediatrica. 2017;28:13–5.
18. Taylor RS . Respiratory syncytial virus and palivizumab prophylaxis in the COVID-19 era. CMAJ. 2021 Apr;193(15):E523. https://doi.org/10.1503/cmaj.78240
19. Agyeman P . Trück. J. Epidemiologie von RSV Infektionen. 2021 [cited 2021 July 15]; Pädiatrie Schweiz: Available from: https://www.paediatrieschweiz.ch/news/epidemiologie-von-rsv-infektionen-2/