Beware the inhaled steroids or corticophobia?
DOI: https://doi.org/10.4414/smw.2021.20450
Dimos
Gidarisab, Stella
Staboulib, Andrew
Bushc
a University of Nicosia Medical School, Cyprus
b Aristotle University of Thessaloniki, Thessaloniki, Greece
c Imperial College, National Heart and Lung Institute, Royal Brompton and Harefield NHS Foundation Trust, London, United Kingdom
Summary
Inhaled corticosteroids have been the cornerstone of atopic, allergic asthma management for more than half a century. They have side effects (as does every medication), but they have transformed the control of asthma and saved many lives since their introduction. The current review revisits local and systemic side effects of inhaled corticosteroids and explores the prevalence and nature of parental fears (“corticophobia”) in different parts of the world. The empathetic physician treating asthma should bear in mind the impact of parental concerns on the management of this potentially life-threatening disease and try to dispel myths at every opportunity by emphasising the well-established benefits and excellent safety profile of inhaled corticosteroids.
Introduction
Inhaled corticosteroids (ICSs) have been the bedrock of asthma therapeutics for more than 50 years [1, 2], in particular for those with the treatable trait of eosinophilic airway inflammation [3]. Families, patients and clinicians recognise that these drugs have improved quality of life and saved so many lives, and many adopt a realistic approach regarding treatment benefits as well as their safety profile [4]. The Global Initiative for Asthma (GINA) guidelines have highlighted the risks of managing even step 1 asthma without a preventer, and no longer recommend salbutamol only treatment in adolescents and adults. This combined reliever is recommended at all stages in this age group [5]. Corticophobia is something that many practitioners have to deal with, since the risk-benefit ratio of ICSs troubles many families all over the world [6–9]; no consultation can be regarded as holistic, unless there is empathetic exploration of parental and patients’ concerns, with the aim of improving adherence [10].
ICSs reduce asthma morbidity and mortality, but – as with every medication – they are not perfect [11]. The ideal ICS would have maximal efficacy (high lung deposition and long duration of action) combined with low systemic bioavailability and rapid systemic clearance [12].
Local and systemic side effects of inhaled corticosteroids
Side effects from the use of ICSs are potentially local or systemic. Potential side effects upon the skin and mucous membranes of the respiratory tract are shown in table 1. These local side effects are frequently underreported by the families [13] and can be minimised by proper education regarding inhalation technique (use of a spacer and immediate mouth rinsing after inhalation) [10]. Occasionally, changing to a different inhalation device may be warranted, depending on the specific local side effect [14]. Devices have different balances between oropharyngeal and lower airway deposition, and may be more or less convenient to use. Thus a spacer will ensure better lower airway deposition than a pressurised metered dose inhaler alone, but is less convenient than dry powder or breath-actuated devices, which have intermediate effects on lower airway deposition. Nebulisers have no place in home management of asthma, because they have repeatedly been shown not to be more efficient than a pressurised metered dose inhaler and spacer. Systemic side effects of inhaled corticosteroids are outlined in table 2. It is worth emphasising that these systemic adverse effects occur only when these medications are systemically available [15].
Table 1 Potential local adverse effects of inhaled corticosteroids.
| Reflex cough |
| Candidiasis (oropharyngeal – oesophageal) |
| Dermatitis (perioral) |
| Hoarse voice |
| Feeling thirsty – dry mouth |
| Foul breath (halitosis) |
| Increased risk of tuberculosis, atypical mycobacterial infection and pneumonia, at least in adults |
Table 2 Potential systemic adverse effects of inhaled corticosteroids.
| Effects on linear growth (suppressed growth velocity –reduced final height) |
| Adrenal insufficiency/crisis due to hypothalamus-hypophysis-adrenal axis suppression |
Effects on bone health
– Osteoporosis
– Bone fractures |
| Effects on immunity |
Ophthalmological complications
– Cataract
– Glaucoma |
| Impairment of glycaemic control |
Suggested low, intermediate and high doses for different age groups for the different steroids are summarised in tables 3a and 3b
[5].
Table 3a Total inhaled corticosteroid dose (μg/day) in children 6–11 years old.
|
ICS
|
Low dose
|
Intermediate dose
|
High dose
|
| Beclomethasone (pMDI, HFA) |
100–200 |
>200–400 |
>400 |
| Budesonide (DPI) |
100–200 |
>200–400 |
>400 |
| Budesonide (nebules) |
250–500 |
>500–1000 |
>1000 |
| Ciclesonide (pMDI, HFA) |
80 |
>80–160 |
>160 |
| Fluticasone propionate (pMDI, HFA or DPI) |
50–100 |
>100–200 |
>200 |
| Mometasone furoate (pMDI, HFA) |
100 |
200 |
Table 3b Total inhaled corticosteroid dose (μg/day) in patients older than 12 years.
|
ICS
|
Low dose
|
Intermediate dose
|
High dose
|
| Beclomethasone (pMDI, HFA) |
200–500 |
>500–1000 |
>1000 |
| Budesonide (DPI) |
200–400 |
>400–800 |
>800 |
| Ciclesonide (pMDI, HFA) |
80–160 |
160 – 320 |
>320 |
| Fluticasone propionate (pMDI, HFA or DPI) |
100–250 |
>250–500 |
>500 |
| Mometasone furoate (pMDI, HFA) |
200 |
400 |
| Mometasone furoate (DPI) |
200–400 |
>400 |
| DPI = dry powder inhaler; HFA = hydrofluoroalkane propellant; ICS = inhaled corticosteroid; pMDI = pressurised metered-dose inhaler |
Linear growth
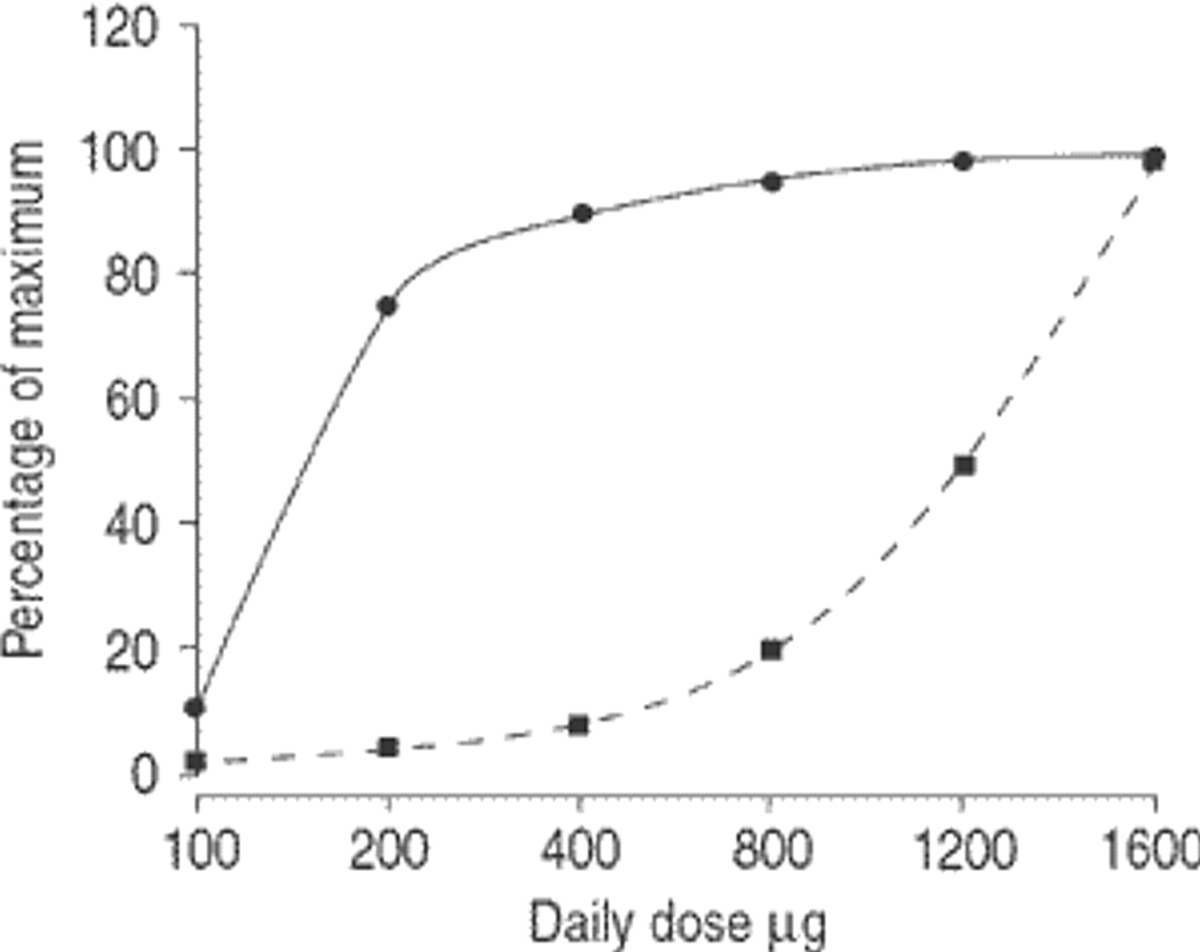
Despite the dictum that “short stature never killed anybody” [16] and the well-known fact that ICSs control a potentially life-threatening disease, parents’ main concerns regarding these medications understandably centre around linear growth suppression [17]. Systemically available corticosteroids can affect linear growth through several mechanisms: growth hormone suppression, down-regulation of growth hormone receptors, inhibition of insulin-like growth factor-1 and suppression of collagen synthesis at the epiphyseal growth plate [18, 19]. A Cochrane review in 2014 concluded that “regular use of ICS at low or medium daily doses is associated with a mean reduction of 0.48 cm/y in linear growth velocity and a 0.61-cm change from baseline in height during a one-year treatment period in children with mild to moderate persistent asthma” [20]. In the same review it was pointed that the growth effects of ICSs are molecule dependent, with newer drugs such as ciclesonide and mometasone – albeit with fewer patients in relevant papers – appearing to be safer [20]. The common-sense approach to use the lowest effective dose has been substantiated by another Cochrane review published the same year, which reported that low doses produced a smaller reduction of growth velocity compared with medium doses in prepubescent school-aged children with mild to moderate persistent asthma [21]. It must be emphasised that before escalating ICS doses it is essential to check that all the basics of asthma management have been addressed [22], given that the dose-response curves of pulmonary anti-inflammatory effects and undesired systemic adverse effects differ substantially [23] (fig. 1). Indeed, most children have no additional benefit from increasing ICS dose beyond 200 μg/day [24]. More recently, Axelson et al concluded that the effects of ICSs may also be device dependent [25]. However, it should be noted that the randomised controlled trials which inform these systematic meta-analyses do not mirror what happens in real life. Specifically, in these studies fixed doses of ICSs were used, whereas in clinical practice doses are reduced when asthma is controlled. Additionally, most of these studies enrolled prepubertal children with mild persistent asthma who may exhibit more steroid-induced growth suppression than children with more severe asthma [15]. The best longitudinal study is the Children’s Asthma Management Program (CAMP) study, which showed that mean adult height of children who received budesonide 400 μg/day for 4.5 years was only 1.2 cm lower compared with the placebo group [26]. However, it should not be forgotten that uncontrolled asthma per se, as well as atopy, also may impair linear growth [17, 27–29]; additionally, some of the asthmatic patients prescribed an ICS have an extra steroid burden (topical steroids for allergic rhinitis and atopic dermatitis) [30] and/or may be on other medications that may affect growth, such as central stimulants (methylphenidate or dextroamphetamine) for the treatment of attention deficit hyperactivity disorder or antiepileptic drugs [31–33]. Measuring the height and showing the growth chart to the family is a sine qua non of every asthma consultation [17, 22].
Adrenal insufficiency / crisis due to hypothalamus-hypophysis-adrenal axis suppression
Exogenous administration of high-dose corticosteroids may produce adrenal suppression through negative feedback at the level of hypothalamus and hypophysis [34]. This may go unnoticed until the impact of a new physiological stress or reduction in asthma treatment leads to adrenal insufficiency [19]. Suppression of the hypothalamus-hypophysis-adrenal axis secondary to ICS use was considered extremely rare, until Todd et al. published in 2002 a survey of UK paediatricians and endocrinologists that identified 33 asthmatic patients (28 children and 5 adults) with adrenal crises [35]. This iatrogenic adrenal failure typically initially produces a nonspecific clinical picture of fatigue, malaise, vomiting, poor weight gain, arthralgias, myalgias, dizziness and psychiatric symptoms. Adrenal crisis presents with hypotension and hypoglycaemia which –if left untreated – will lead to seizures and coma. Longer duration (>18 months) and higher doses (>400 μg/day of beclomethasone or equivalent) are obvious risk factors [36], but idiosyncratic responses to low-moderate doses cannot be ruled out, meaning that a high index of suspicion in children presenting with nonspecific symptoms is mandatory [37]. The clinician should also inquire about co-administration of other medications that potentiate the systemic effects of steroids (for example, some antifungals, antiretrovirals, antidepressants and ciclosporin) or other prescriptions of exogenous steroids that increase the steroid burden [38]. Symptomatic and high-risk patients should be investigated for adrenal suppression with the help of an experienced paediatric endocrinologist. There is a lack of consensus regarding the optimal test [39], but dynamic testing of the axis with low-dose (1 μg) adrenocorticotropic hormone (ACTH) stimulation is considered superior to early morning cortisol screening, since the latter may be affected by disturbances of the sleep-wake cycle [37]. The management of adrenal suppression should ideally be guided by a paediatric endocrinologist and the asthma specialist should ensure that patients and families are educated about recognising this potentially lethal adverse event and have a rescue plan in the event of it occurring [38].
Effects on bone health
It is well known that systemic administration of glucocorticosteroids modifies osteoblastic and osteoclastic activity leading to osteoporosis and pathological fractures [40]. Loke et al. in their meta-analysis (2015) did not demonstrate any significant association between ICSs and fractures in children (pooled odds ratio [OR] 1.02, 95% confidence interval [CI] 0.94–1.10, two studies), or adults (pooled OR 1.09, 95% CI 0.45–2.62, four studies). In the same work no bone mineral density reduction was reported with ICS prophylaxis as compared with controls [41]. In a more recent population-based nested case-control study, Gray et al. showed no significant association between first fracture after asthma diagnosis and current use (OR 1.07, 95% CI 0.97–1.17), recent use (OR 0.96, 95% CI 0.86–1.07), or past use (OR 1.00, 95% CI 0.91–1.11) of ICSs compared with controls [42]. However, in the same population, use of systemic corticosteroids led to a greater odds ratio for fracture (OR 1.17, 95% CI 1.04–1.33) [42]. Clinicians should exercise greater caution when there are additional risk factors for reduced bone mineral density, such as poor diet, other chronic diseases or a family history of osteoporosis. Nutritional interventions that ensure adequate intake of vitamin D and calcium are essential for every child’s bone health [43, 44]. Additionally, given that vitamin D deficiency may influence the inflammatory response of the respiratory tract, monitoring vitamin D levels in asthmatic children is advised [45, 46]. Weight-bearing exercise should be encouraged to increase bone mineral density. The Pediatric Endocrine Society Drugs and Therapeutics Committee recently concluded that routine dual-energy X-ray absorptiometry screening is not justified in children taking ICS without other major risk factors [18].
Effects on immunity
Systemic administration of corticosteroids leads to immunosuppression and therefore there is an understandable concern that ICSs may also impair innate airway defence mechanisms [17]. In the adult population, recent work points towards an increase in the incidence of pneumonia in patients with asthma prescribed ICSs [47]. Similar concerns exist regarding tuberculosis in adults with obstructive lung diseases who are treated with inhaled steroids [48]. Finally, nontuberculous mycobacterial pulmonary disease has been associated with preceding ICS use, with evidence for a dose-response relationship [49]. Data in children are too sparse to draw any firm conclusions [50].
Ophthalmological complications (cataract and glaucoma)
Because systemic steroids lead to an increased risk of cataract formation and glaucoma, the association between inhaled steroid therapy and ocular side effects has been investigated [51–53]. The CAMP study was reassuring regarding cataract formation; out of 311 children who were randomly assigned to receive budesonide, only one child was classified as having a barely measurable (<0.5 mm) posterior subcapsular cataract [54]. Similarly, no increase of glaucoma has been found in children prescribed ICSs [55].
Impairment of glycaemic control
Glycaemic effects of ICSs in children have been investigated in only one small study. A Turkish group reported that levels of glycated haemoglobin (HbA1c) in asthmatic children were significantly higher than in controls, but no significant correlation between cumulative steroid dose and HbA1c levels was demonstrated [56]. It is clear that more data are needed. Clinical experience is that glycaemic complications of ICSs are incredibly rare if indeed they occur.
Corticophobia
The topic of corticophobia in families of asthmatic children has been explored in 13 original studies in the English-language literature from 1996 to 2018 [7, 8, 57–66]. Only two research groups used the Beliefs about Medicine Questionnaire (BMQ) [63–66]; BMQ is considered a psychometrically sound instrument that explores commonly held beliefs about medicines [67]. The other papers used unvalidated questionnaires that each group devised for the purposes of their study. Corticophobia appears to trouble 5.23–67.3% of families of asthmatic children. This wide range may be attributed to many factors, including but not limited to the following: using different and often unvalidated tools, sociocultural differences, populations that have received different educational approaches, and changing eras. It is worth emphasising that steroid phobia in different parts of the world may have different qualities [68]. Lim et al. (1996) interviewed parents from Singapore and reported general concerns regarding use of inhalers, including fears of addiction and drug overdosage [57]. Chan et al. compared parents in Malaysia with and without concerns regarding use of inhalers and found no significant differences regarding worries about steroid use [58]. This study leads to the important conclusion that the fear may be of devices, not ICS, and therefore it is important to determine what exactly the family fears. If it is the device, talking about the safety of ICSs will not allay those fears. Yoos et al., in a study that explored barriers to asthma management, found that ethnic minority families in the USA were more likely to have negative attitudes towards prophylactic ICSs [59] Conversely, Conn et al. reported that “non-minority parents were more likely to have necessity scores that exceed concern scores (79% vs 68%; p = 0.03) compared with ethnic minority parents” [64]. In a small study from Israel, Gazala et al. highlight that parental fears focus on addiction, irritability and steroid effects on weight and height [60]. Zedan et al. – in the only study from Africa – reported that in the majority of parents the fear of nonspecific side effects and, interestingly – in a minority of patients – concerns regarding immunity and bone density [8]. Zhao et al – in the largest study published on this topic to date – reported that 24% of the Chinese families they studied are worried about potential adverse effects of steroids on their child’s intelligence [61]. Another facet of parental corticophobia is the pragmatic concern about the high cost of these medications in some healthcare systems [69]. Anecdotally, some families may confuse ICSs with anabolic steroids and need to have fears that, for example, their child may grow a beard, allayed. Clearly corticophobia is a complex phenomenon with many different underlying factors that vary with ethnicity and geographical location.
A recent review article highlighted the paucity of data on the perspective of adolescents and called for papers that compare the views of parents and their asthmatic children on their concerns regarding inhaled steroids [70]. Physicians have been shown to overestimate parental and patients’ concerns [71]. Therefore we need studies that will explore the views of all stakeholders (patients, families and health professionals) using validated tools. Rich and novel insights into adolescent and parental corticophobia can be derived by using data from online asthma forums that minimise geographical and other restrictions in participating in research studies [72].
Corticophobia and its consequences on asthma
Clearly, a substantial proportion of parents have significant concerns regarding steroid therapy [70]; whether corticophobia has a significant effect on adherence is not completely clear [73]. A Malaysian study showed that children of families who had greater fears regarding medication use were more likely to miss 25% of the prescribed steroid doses compared to families with no such concerns [58]. Gazala et al. found that concerned Israeli mothers reduced the recommended dosage far more frequently than mothers with no such concerns [60]. Similarly, one fourth of the families in a recent study from Turkey stopped or reduced ICSs owing to corticophobia [9]. Conn et al. in a study of 150 families using a self-reported medication adherence scale and the necessities and concerns subscales of the BMQ reported that “parental concerns about medications independently predicted medication adherence (α= −0.317, p = 0.03, overall model r2 = 0.168)” [63]. However, note that more than 80% of non-adherence could not be accounted for by corticophobia, at least in this study. Two years later, the same group, using the same methodology in a sample of 622 families, concluded that the mean adherence score increased as the necessity – concern differential score increased [64]. This score is the difference in scores between the necessity subscale and the concern subscale of the BMQ, with a positive score indicating greater necessity or need for the medications, and a negative score indicating greater concern about the medications [67]. Carroll et al., in an international study(the Room to Breathe Survey) found that those parents who self-reported “concerns about steroid medication” were more likely to have a child with very poor control (three or more GINA-uncontrolled items) (OR 1.65, 95% CI 1.23–2.23; p = 0.0006) [74]. Koster et al. showed that “children whose parents had high concerns about medication also showed a two times higher risk for uncontrolled asthma at age 8” despite no differences in self-reported adherence between parents with strong and parents with minor concerns regarding prophylactic medication [66]. These findings are likely to reflect the well-known shortcomings of subjective measurements of medication adherence [73] and also confirm that non-adherence is multifactorial [75]. The findings of Ip et al. from Hong Kong [62] echo Koster’s results and at the same time highlight that concerned parents resort to alternative medicine more frequently. We agree with the views of Hui et al. that poor asthma control may reflect the impact of parental steroid phobia more accurately than reported medication adherence, especially when the latter is not supported by objective measurements [70].
Engelkes et al in a systematic review concluded that “the majority of the papers of good quality indicated that higher levels of adherence were associated with a reduced risk of severe asthma exacerbations” [76]. Given that adherence is a multifaceted and difficult to quantify phenomenon, this causal relationship is not always easy to prove [77, 78]. It is worth noting that some studies have even reported the inverse association, probably because of the bias that treatment need is higher in patients with uncontrolled asthma, who are by definition at risk of asthma attacks [76]. It should never be forgotten that poor adherence may be totally rational if in fact unnecessary or excessive treatment has been prescribed! Chongmelaxme et al. in a recent meta-analysis concluded that “the highest reduction in the odds of exacerbation was associated with patients achieving 80% or more adherence, and the odds were also reduced among those with greater than equal to 50% adherence”; they also demonstrated a substantial increase in asthma attacks with discontinuation of therapy [79].
Summary and conclusions
Through this paper we have used the term “corticophobia”. According to the Cambridge Dictionary, phobia is defined as “an extreme fear or dislike of a particular thing or situation, especially one that is not reasonable” [80]. Given that ICSs – as any other medication – do have side effects, we urge the medical community to reconsider whether this term is useful. We believe that using the words “fears” or “concerns” is more appropriate, since they do not imply that parental worries are irrational.
ICSs are now and for the foreseeable future the cornerstone of the management of asthma, and have dramatically reduced morbidity and mortality. They are not perfect, but they are far safer that systemic corticosteroids or any other alternative. Until a cure for asthma is found, we need to continue educating families, being clear about potential great benefits and likely minimal side effects of this prophylactic therapeutic approach [5, 70]. Dispelling the myths regarding corticosteroids is of the utmost importance [81].
References
1
van Aalderen
WM
,
Sprikkelman
AB
. Inhaled corticosteroids in childhood asthma: the story continues. Eur J Pediatr. 2011;170(6):709–18. doi:.https://doi.org/10.1007/s00431-010-1319-z
2
Crompton
G
. A brief history of inhaled asthma therapy over the last fifty years. Prim Care Respir J. 2006;15(6):326–31. doi:.https://doi.org/10.1016/j.pcrj.2006.09.002
3
Pavord
ID
,
Beasley
R
,
Agusti
A
,
Anderson
GP
,
Bel
E
,
Brusselle
G
, et al.
After asthma: redefining airways diseases. Lancet. 2018;391(10118):350–400. doi:.https://doi.org/10.1016/S0140-6736(17)30879-6
4
Heffler
E
,
Madeira
LNG
,
Ferrando
M
,
Puggioni
F
,
Racca
F
,
Malvezzi
L
, et al.
Inhaled Corticosteroids Safety and Adverse Effects in Patients with Asthma. J Allergy Clin Immunol Pract. 2018;6(3):776–81. doi:.https://doi.org/10.1016/j.jaip.2018.01.025
5Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. 2020. Available from: https://ginasthma.org/gina-reports/
6
Rubin
BK
,
Zhao
W
,
Winders
TA
. What do patients want from their asthma care doctors?
Paediatr Respir Rev. 2018;27:86–9. doi:.https://doi.org/10.1016/j.prrv.2017.11.002
7
Abu-Shaheen
AK
,
Nofal
A
,
Heena
H
. Parental Perceptions and Practices toward Childhood Asthma. BioMed Res Int. 2016;2016:6364194. doi:.https://doi.org/10.1155/2016/6364194
8
Zedan
MM
,
Ezz El Regal
M
,
A Osman
E
,
E Fouda
A
. Steroid Phobia among Parents of Asthmatic Children: Myths and Truth. Iran J Allergy Asthma Immunol. 2010;9(3):163–8.
9
Özçeker
D
,
Uçkun
U
,
İslamova
D
,
Tamay
Z
,
Güler
N
. Corticosteroid phobia among parents of asthmatic children. Turk J Pediatr. 2018;60(2):142–6. doi:.https://doi.org/10.24953/turkjped.2018.02.004
10
Hossny
E
,
Rosario
N
,
Lee
BW
,
Singh
M
,
El-Ghoneimy
D
,
Soh
JY
, et al.
The use of inhaled corticosteroids in pediatric asthma: update. World Allergy Organ J. 2016;9:26. doi:.https://doi.org/10.1186/s40413-016-0117-0
11
Bazzy-Asaad
A
. Safety of inhaled corticosteroids in children with asthma. Curr Opin Pediatr. 2001;13(6):523–7. doi:.https://doi.org/10.1097/00008480-200112000-00005
12
Gulliver
T
,
Morton
R
,
Eid
N
. Inhaled corticosteroids in children with asthma: pharmacologic determinants of safety and efficacy and other clinical considerations. Paediatr Drugs. 2007;9(3):185–94. doi:.https://doi.org/10.2165/00148581-200709030-00007
13
Carlsen
KH
. Inhaled steroids and local side-effects. Allergy. 2001;56(10):925–7.
14
Dubus
JC
,
Marguet
C
,
Deschildre
A
,
Mely
L
,
Le Roux
P
,
Brouard
J
, et al.; Réseau de Recherche Clinique en Pneumonologie Pédiatrique. Local side-effects of inhaled corticosteroids in asthmatic children: influence of drug, dose, age, and device. Allergy. 2001;56(10):944–8. doi:.https://doi.org/10.1034/j.1398-9995.2001.00100.x
15
Camargos
P
,
Zhang
L
,
Lasmar
L
,
Brand
P
. Question 2: What are the effects of inhaled corticosteroids on growth in children?
Paediatr Respir Rev. 2018;28:33–40. doi:.https://doi.org/10.1016/j.prrv.2018.03.008
16
Brook
CG
. Short stature never killed anybody. J Pediatr. 1998;133(5):591–2. doi:.https://doi.org/10.1016/S0022-3476(98)70095-2
17
Bush
A
. Inhaled corticosteroid and children’s growth. Arch Dis Child. 2014;99(3):191–2. doi:.https://doi.org/10.1136/archdischild-2012-303105
18
Kapadia
CR
,
Nebesio
TD
,
Myers
SE
,
Willi
S
,
Miller
BS
,
Allen
DB
, et al.; Drugs and Therapeutics Committee of the Pediatric Endocrine Society. Endocrine Effects of Inhaled Corticosteroids in Children. JAMA Pediatr. 2016;170(2):163–70. doi:.https://doi.org/10.1001/jamapediatrics.2015.3526
19
Wolfgram
PM
,
Allen
DB
. Effects of Inhaled Corticosteroids on Growth, Bone Metabolism, and Adrenal Function. Adv Pediatr. 2017;64(1):331–45. doi:.https://doi.org/10.1016/j.yapd.2017.03.006
20
Zhang
L
,
Prietsch
SO
,
Ducharme
FM
. Inhaled corticosteroids in children with persistent asthma: effects on growth. Cochrane Database Syst Rev. 2014;7(4):CD009471. doi:.https://doi.org/10.1002/ebch.1988
21
Pruteanu
AI
,
Chauhan
BF
,
Zhang
L
,
Prietsch
SO
,
Ducharme
FM
. Inhaled corticosteroids in children with persistent asthma: dose-response effects on growth. Cochrane Database Syst Rev. 2014;9(4):CD009878. doi:.https://doi.org/10.1002/ebch.1989
22
Bush
A
,
Fleming
L
. 2012 and never been KISSed: we need to improve the care of children with asthma. Prim Care Respir J. 2012;21(3):242–4. doi:.https://doi.org/10.4104/pcrj.2012.00062
23
Brand
PL
. Inhaled corticosteroids reduce growth. Or do they?
Eur Respir J. 2001;17(2):287–94. doi:.https://doi.org/10.1183/09031936.01.17202870
24
Lemanske
RF, Jr
,
Mauger
DT
,
Sorkness
CA
,
Jackson
DJ
,
Boehmer
SJ
,
Martinez
FD
, et al.; Childhood Asthma Research and Education (CARE) Network of the National Heart, Lung, and Blood Institute. Step-up therapy for children with uncontrolled asthma receiving inhaled corticosteroids. N Engl J Med. 2010;362(11):975–85. doi:.https://doi.org/10.1056/NEJMoa1001278
25
Axelsson
I
,
Naumburg
E
,
Prietsch
SO
,
Zhang
L
. Inhaled corticosteroids in children with persistent asthma: effects of different drugs and delivery devices on growth. Cochrane Database Syst Rev. 2019;6:CD010126. doi:.https://doi.org/10.1002/14651858.CD010126.pub2
26
Kelly
HW
,
Sternberg
AL
,
Lescher
R
,
Fuhlbrigge
AL
,
Williams
P
,
Zeiger
RS
, et al.; CAMP Research Group. Effect of inhaled glucocorticoids in childhood on adult height. N Engl J Med. 2012;367(10):904–12. doi:.https://doi.org/10.1056/NEJMoa1203229
27
Zhang
L
,
Lasmar
LB
,
Castro-Rodriguez
JA
. The impact of asthma and its treatment on growth: an evidence-based review. J Pediatr (Rio J). 2019;95(Suppl 1):10–22. doi:.https://doi.org/10.1016/j.jped.2018.10.005
28
Cohen
MB
,
Abram
LE
. Growth patterns of allergic children; a statistical study using the grid technique. J Allergy. 1948;19(3):165–71. doi:.https://doi.org/10.1016/0021-8707(48)90049-5
29
Russell
G
. Childhood asthma and growth--a review of the literature. Respir Med. 1994;88(Suppl A):31–6, discussion 36–7. doi:.https://doi.org/10.1016/S0954-6111(05)80038-1
30
Ekins-Daukes
S
,
Simpson
CR
,
Helms
PJ
,
Taylor
MW
,
McLay
JS
. Burden of corticosteroids in children with asthma in primary care: retrospective observational study. BMJ. 2002;324(7350):1374. doi:.https://doi.org/10.1136/bmj.324.7350.1374
31
Allen
DB
. Inhaled corticosteroids and growth: still an issue after all these years. J Pediatr. 2015;166(2):463–9. doi:.https://doi.org/10.1016/j.jpeds.2014.09.055
32
Powell
SG
,
Frydenberg
M
,
Thomsen
PH
. The effects of long-term medication on growth in children and adolescents with ADHD: an observational study of a large cohort of real-life patients. Child Adolesc Psychiatry Ment Health. 2015;9(1):50. doi:.https://doi.org/10.1186/s13034-015-0082-3
33
Lee
HS
,
Wang
SY
,
Salter
DM
,
Wang
CC
,
Chen
SJ
,
Fan
HC
. The impact of the use of antiepileptic drugs on the growth of children. BMC Pediatr. 2013;13(1):211. doi:.https://doi.org/10.1186/1471-2431-13-211
34
Bayman
E
,
Drake
AJ
. Adrenal suppression with glucocorticoid therapy: still a problem after all these years?
Arch Dis Child. 2017;102(4):338–9. doi:.https://doi.org/10.1136/archdischild-2016-311601
35
Todd
GR
,
Acerini
CL
,
Ross-Russell
R
,
Zahra
S
,
Warner
JT
,
McCance
D
. Survey of adrenal crisis associated with inhaled corticosteroids in the United Kingdom. Arch Dis Child. 2002;87(6):457–61. doi:.https://doi.org/10.1136/adc.87.6.457
36
Anurhada
KWDA
, Prematilake GLDC, Batuwita BAUI, Kannangoda KASR, Hewagamage US, Wijeratne S, et al.
Effect of long term inhaled corticosteroid therapy on adrenal suppression, growth and bone health in children with asthma. BMC Pediatr. 2019;19(1):411. doi:.https://doi.org/10.1186/s12887-019-1760-8
37
Ahmet
A
,
Mokashi
A
,
Goldbloom
EB
,
Huot
C
,
Jurencak
R
,
Krishnamoorthy
P
, et al.
Adrenal suppression from glucocorticoids: preventing an iatrogenic cause of morbidity and mortality in children. BMJ Paediatr Open. 2019;3(1):e000569. doi:.https://doi.org/10.1136/bmjpo-2019-000569
38
Ahmet
A
,
Kim
H
,
Spier
S
. Adrenal suppression: A practical guide to the screening and management of this under-recognized complication of inhaled corticosteroid therapy. Allergy Asthma Clin Immunol. 2011;7(1):13. doi:.https://doi.org/10.1186/1710-1492-7-13
39
Zöllner
EW
,
Lombard
CJ
,
Galal
U
,
Hough
S
,
Irusen
EM
,
Weinberg
E
. Screening for hypothalamic-pituitary-adrenal axis suppression in asthmatic children remains problematic: a cross-sectional study. BMJ Open. 2013;3(8):e002935. doi:.https://doi.org/10.1136/bmjopen-2013-002935
40
Tse
SM
,
Kelly
HW
,
Litonjua
AA
,
Van Natta
ML
,
Weiss
ST
,
Tantisira
KG
; Childhood Asthma Management Program Research Group. Corticosteroid use and bone mineral accretion in children with asthma: effect modification by vitamin D. J Allergy Clin Immunol. 2012;130(1):53–60.e4. doi:.https://doi.org/10.1016/j.jaci.2012.04.005
41
Loke
YK
,
Gilbert
D
,
Thavarajah
M
,
Blanco
P
,
Wilson
AM
. Bone mineral density and fracture risk with long-term use of inhaled corticosteroids in patients with asthma: systematic review and meta-analysis. BMJ Open. 2015;5(11):e008554. doi:.https://doi.org/10.1136/bmjopen-2015-008554
42
Gray
N
,
Howard
A
,
Zhu
J
,
Feldman
LY
,
To
T
. Association Between Inhaled Corticosteroid Use and Bone Fracture in Children With Asthma. JAMA Pediatr. 2018;172(1):57–64. doi:.https://doi.org/10.1001/jamapediatrics.2017.3579
43
Fuhlbrigge
AL
,
Kelly
HW
. Inhaled corticosteroids in children: effects on bone mineral density and growth. Lancet Respir Med. 2014;2(6):487–96. doi:.https://doi.org/10.1016/S2213-2600(14)70024-4
44
Chalitsios
CV
,
Shaw
DE
,
McKeever
TM
. Risk of osteoporosis and fragility fractures in asthma due to oral and inhaled corticosteroids: two population-based nested case-control studies. Thorax. 2021;76(1):21–8. doi:.https://doi.org/10.1136/thoraxjnl-2020-215664
45
Hall
SC
,
Agrawal
DK
. Vitamin D and Bronchial Asthma: An Overview of Data From the Past 5 Years. Clin Ther. 2017;39(5):917–29. doi:.https://doi.org/10.1016/j.clinthera.2017.04.002
46
Liu
J
,
Dong
YQ
,
Yin
J
,
Yao
J
,
Shen
J
,
Sheng
GJ
, et al.
Meta-analysis of vitamin D and lung function in patients with asthma. Respir Res. 2019;20(1):161. doi:.https://doi.org/10.1186/s12931-019-1072-4
47
Kim
MH
,
Rhee
CK
,
Shim
JS
,
Park
SY
,
Yoo
KH
,
Kim
BY
, et al.
Inhaled Corticosteroids in Asthma and the Risk of Pneumonia. Allergy Asthma Immunol Res. 2019;11(6):795–805. doi:.https://doi.org/10.4168/aair.2019.11.6.795
48
Castellana
G
,
Castellana
M
,
Castellana
C
,
Castellana
G
,
Resta
E
,
Carone
M
, et al.
Inhaled Corticosteroids And Risk Of Tuberculosis In Patients With Obstructive Lung Diseases: A Systematic Review And Meta-Analysis Of Non-randomized Studies. Int J Chron Obstruct Pulmon Dis. 2019;14:2219–27. doi:.https://doi.org/10.2147/COPD.S209273
49
Liu
VX
,
Winthrop
KL
,
Lu
Y
,
Sharifi
H
,
Nasiri
HU
,
Ruoss
SJ
. Association between Inhaled Corticosteroid Use and Pulmonary Nontuberculous Mycobacterial Infection. Ann Am Thorac Soc. 2018;15(10):1169–76. doi:.https://doi.org/10.1513/AnnalsATS.201804-245OC
50
Levy
J
,
Zalkinder
I
,
Kuperman
O
,
Skibin
A
,
Apte
R
,
Bearman
JE
, et al.
Effect of prolonged use of inhaled steroids on the cellular immunity of children with asthma. J Allergy Clin Immunol. 1995;95(4):806–12. doi:.https://doi.org/10.1016/S0091-6749(95)70122-2
51
Behbehani
AH
,
Owayed
AF
,
Hijazi
ZM
,
Eslah
EA
,
Al-Jazzaf
AM
. Cataract and ocular hypertension in children on inhaled corticosteroid therapy. J Pediatr Ophthalmol Strabismus. 2005;42(1):23–7. doi:.https://doi.org/10.3928/01913913-20050101-03
52
Nootheti
S
,
Bielory
L
. Risk of cataracts and glaucoma with inhaled steroid use in children. Compr Ophthalmol Update. 2006;7(1):31–9.
53
Simons
FE
,
Persaud
MP
,
Gillespie
CA
,
Cheang
M
,
Shuckett
EP
. Absence of posterior subcapsular cataracts in young patients treated with inhaled glucocorticoids. Lancet. 1993;342(8874):776–8. doi:.https://doi.org/10.1016/0140-6736(93)91541-S
54
Szefler
S
,
Weiss
S
,
Tonascia
J
,
Adkinson
NF
,
Bender
B
,
Cherniack
R
, et al., Childhood Asthma Management Program Research Group. Long-term effects of budesonide or nedocromil in children with asthma. N Engl J Med. 2000;343(15):1054–63. doi:.https://doi.org/10.1056/NEJM200010123431501
55
Chang
LS
,
Lee
HC
,
Tsai
YC
,
Shen
LS
,
Li
CL
,
Liu
SF
, et al.
Decreased incidence of glaucoma in children with asthma using inhaled corticosteroid: a cohort study. Oncotarget. 2017;8(62):105463–71. doi:.https://doi.org/10.18632/oncotarget.22252
56
Yucel
O
,
Eker
Y
,
Nuhoglu
C
,
Ceran
O
. Hemoglobin a1c levels in children with asthma using low dose inhaled corticosteroids. Indian Pediatr. 2009;46(4):300–3.
57
Lim
SH
,
Goh
DY
,
Tan
AY
,
Lee
BW
. Parents’ perceptions towards their child’s use of inhaled medications for asthma therapy. J Paediatr Child Health. 1996;32(4):306–9. doi:.https://doi.org/10.1111/j.1440-1754.1996.tb02559.x
58
Chan
PW
,
DeBruyne
JA
. Parental concern towards the use of inhaled therapy in children with chronic asthma. Pediatr Int. 2000;42(5):547–51. doi:.https://doi.org/10.1046/j.1442-200x.2000.01278.x
59
Yoos
HL
,
Kitzman
H
,
McMullen
A
. Barriers to anti-inflammatory medication use in childhood asthma. Ambul Pediatr. 2003;3(4):181–90. doi:.https://doi.org/10.1367/1539-4409(2003)003<0181:BTAMUI>2.0.CO;2
60
Gazala
E
,
Sadka
R
,
Bilenko
N
. Parents’ Fears and Concerns Toward Inhaled Corticosteroid Treatment for Their Asthmatic Children. Pediatr Asthma Allergy Immunol. 2005;18(2):82–7. doi:.https://doi.org/10.1089/pai.2005.18.82
61
Zhao
J
,
Shen
K
,
Xiang
L
,
Zhang
G
,
Xie
M
,
Bai
J
, et al.
The knowledge, attitudes and practices of parents of children with asthma in 29 cities of China: a multi-center study. BMC Pediatr. 2013;13(1):20. doi:.https://doi.org/10.1186/1471-2431-13-20
62
Ip
KI
,
Hon
KL
,
Tsang
KYC
,
Leung
TNH
. Steroid phobia, Chinese medicine and asthma control. Clin Respir J. 2018;12(4):1559–64. doi:.https://doi.org/10.1111/crj.12705
63
Conn
KM
,
Halterman
JS
,
Fisher
SG
,
Yoos
HL
,
Chin
NP
,
Szilagyi
PG
. Parental beliefs about medications and medication adherence among urban children with asthma. Ambul Pediatr. 2005;5(5):306–10. doi:.https://doi.org/10.1367/A05-004R1.1
64
Conn
KM
,
Halterman
JS
,
Lynch
K
,
Cabana
MD
. The impact of parents’ medication beliefs on asthma management. Pediatrics. 2007;120(3):e521–6. doi:.https://doi.org/10.1542/peds.2006-3023
65
Koster
ES
,
Raaijmakers
JA
,
Vijverberg
SJ
,
Maitland-van der Zee
AH
. Inhaled corticosteroid adherence in paediatric patients: the PACMAN cohort study. Pharmacoepidemiol Drug Saf. 2011;20(10):1064–72. doi:.https://doi.org/10.1002/pds.2228
66
Koster
ES
,
Wijga
AH
,
Koppelman
GH
,
Postma
DS
,
Brunekreef
B
,
De Jongste
JC
, et al.
Uncontrolled asthma at age 8: the importance of parental perception towards medication. Pediatr Allergy Immunol. 2011;22(5):462–8. doi:.https://doi.org/10.1111/j.1399-3038.2011.01150.x
67
Horne
R
,
Weinman
J
,
Hankins
M
. The beliefs about medicines questionnaire: The development and evaluation of a new method for assessing the cognitive representation of medication. Psychol Health. 1999;14(1):1–24. doi:.https://doi.org/10.1080/08870449908407311
68
Lenney
W
,
Bush
A
,
Fitzgerald
DA
,
Fletcher
M
,
Ostrem
A
,
Pedersen
S
, et al.
Improving the global diagnosis and management of asthma in children. Thorax. 2018;73(7):662–9. doi:.https://doi.org/10.1136/thoraxjnl-2018-211626
69Asthma UK. Paying to breathe: why unfair asthma prescription charges must be stopped. 2019.
70
Hui
RWH
. Inhaled corticosteroid-phobia and childhood asthma: Current understanding and management implications. Paediatr Respir Rev. 2020;33:62–6. doi:.https://doi.org/10.1016/j.prrv.2019.03.009
71
Gillissen
A
,
Lecheler
J
. Kortikophobie beim Asthma bronchiale [Corticophobia in asthma]. Med Klin (Munich). 2003;98(8):417–22. Article in German. doi:.https://doi.org/10.1007/s00063-003-1262-0
72
De Simoni
A
,
Horne
R
,
Fleming
L
,
Bush
A
,
Griffiths
C
. What do adolescents with asthma really think about adherence to inhalers? Insights from a qualitative analysis of a UK online forum. BMJ Open. 2017;7(6):e015245. doi:.https://doi.org/10.1136/bmjopen-2016-015245
73
Desager
K
,
Vermeulen
F
,
Bodart
E
. Adherence to asthma treatment in childhood and adolescence - a narrative literature review. Acta Clin Belg. 2018;73(5):348–55. doi:.https://doi.org/10.1080/17843286.2017.1409684
74
Carroll
WD
,
Wildhaber
J
,
Brand
PLP
. Parent misperception of control in childhood/adolescent asthma: the Room to Breathe survey. Eur Respir J. 2012;39(1):90–6. doi:.https://doi.org/10.1183/09031936.00048911
75
Klok
T
,
Kaptein
AA
,
Duiverman
EJ
,
Brand
PL
. Long-term adherence to inhaled corticosteroids in children with asthma: Observational study. Respir Med. 2015;109(9):1114–9. doi:.https://doi.org/10.1016/j.rmed.2015.07.016
76
Engelkes
M
,
Janssens
HM
,
de Jongste
JC
,
Sturkenboom
MCJM
,
Verhamme
KMC
. Medication adherence and the risk of severe asthma exacerbations: a systematic review. Eur Respir J. 2015;45(2):396–407. doi:.https://doi.org/10.1183/09031936.00075614
77
Vasbinder
EC
,
Belitser
SV
,
Souverein
PC
,
van Dijk
L
,
Vulto
AG
,
van den Bemt
PM
. Non-adherence to inhaled corticosteroids and the risk of asthma exacerbations in children. Patient Prefer Adherence. 2016;10:531–8. doi:.https://doi.org/10.2147/PPA.S92824
78
Fleming
L
. Asthma exacerbation prediction: recent insights. Curr Opin Allergy Clin Immunol. 2018;18(2):117–23. doi:.https://doi.org/10.1097/ACI.0000000000000428
79
Chongmelaxme
B
,
Chaiyakunapruk
N
,
Dilokthornsakul
P
. Association between adherence and severe asthma exacerbation: A systematic review and meta-analysis. J Am Pharm Assoc (2003). 2020;60(5):669–685.e2. doi:.https://doi.org/10.1016/j.japh.2020.02.010
80Cambridge Dictionary. Phobia [17/03/2020]. Available from: https://dictionary.cambridge.org/dictionary/english/phobia.
81
Rubin
BK
. Asthma myths, controversies, and dogma. Paediatr Respir Rev. 2015;16(2):83–7. doi:.https://doi.org/10.1016/j.prrv.2014.09.001