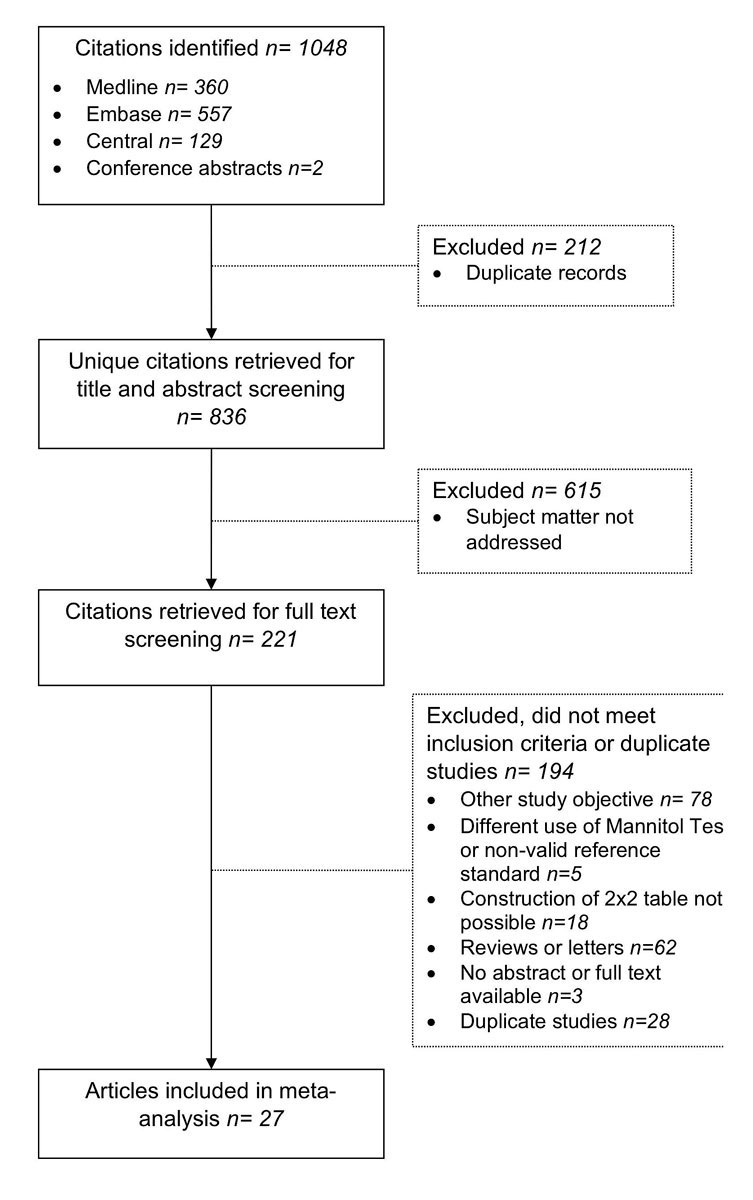
Figure 1 Identification of studies.
DOI: https://doi.org/10.4414/smw.2019.20100
Global Initiative for Asthma
bronchial provocation tests
eucapnic voluntary hyperpnoea
Quality Assessment of Diagnostic Accuracy Studies
American Thoracic Society
European Respiratory Society
hierarchical summary receiver operating characteristic
confidence interval
forced expiratory volume in 1 second
true positive
false positive
false negative
true negative
receiver operating characteristic
chronic obstructive pulmonary disease
Asthma is a chronic inflammatory airway disease with an estimated 300 million affected individuals worldwide [1]. The chronic inflammation of the airways is associated with airway hyperresponsiveness with recurrent episodes of wheezing, breathlessness, chest tightness, coughing and provocation by typical triggers [1].
A correct diagnosis of asthma is essential if appropriate drug therapy is to be given. The diagnosis of asthma should not be based on respiratory symptoms alone as the symptoms may be unspecific [1, 2]. According to the Global Initiative for Asthma (GINA) guidelines, the diagnosis of asthma is made by the presence of variable respiratory symptoms and a confirmed variable expiratory airflow limitation with objective pulmonary function tests [1].
Bronchial provocation tests (BPTs) are particularly useful for the detection of airway hyperresponsiveness and diagnosing asthma on occasions where the lung function testing shows normal results. Two different methods for bronchial provocation tests exist, the ´direct´ and the “indirect” method. “Direct” bronchial provocation tests cause airway narrowing by acting “directly“ on their respective receptors on bronchial smooth muscle, causing contraction [1, 3, 4]. The “direct” tests are very sensitive for identifying airway hyperresponsiveness. A limitation of direct tests is that they act directly on the smooth muscle, and can show airway hyperresponsiveness even without any active airway inflammation. This direct effect may cause false positive test results and thus reduce specificity [1, 3, 5].
In contrast to this, “indirect“ challenge tests such as the mannitol test cause airway narrowing by releasing a wide variety of mediators of bronchoconstriction from inflammatory cells within the walls of the airways [3]. Because of this mechanism indirect tests are more specific for identifying asthma that is currently active [1, 3, 5, 6]. The mannitol test has the advantage of a standardised protocol, ease of administration, shortness of procedure and good safety profile due to a progressive dose-response challenge. The test can be stopped before severe falls of FEV1 occur, making it an attractive alternative to the “direct” test methods where different protocols exist [3, 6, 7]. In 2007, mannitol was included as a bronchoprovocation test in the GINA guidelines [1].
Several studies have investigated the accuracy of the mannitol test to identify asthma. However, the results from these studies differ substantially [8–13]. This systematic review aims to clarify this point. Our objective was to investigate the sensitivity and specificity of the mannitol test to diagnose asthma compared to accepted reference standards (GINA) in children and adults with and without asthma symptoms. We included cross-sectional, cohort and case-controlled studies. Characteristics (reference standard, different settings, populations) were recorded. Methodological quality of the studies was assessed with QUADAS-2 [14].
The methods of data extraction and inclusion criteria were specified in advance and documented in a protocol, which is available upon request.
We performed a systematic search of three electronic databases to identify studies evaluating the accuracy of the mannitol BPT for the diagnosis of asthma. A research librarian experienced in literature searches for systematic reviews developed a search strategy in collaboration with the investigators (see appendix 1). We systematically searched Medline (through Ovid or PubMed), Embase (through Ovid) and Central databases from January 1997, as the mannitol BPT was originally described by Anderson et al. in 1997 [15], to February 2019. Participants of any age were considered. No publication status restrictions were imposed. We checked the reference lists of the identified studies as well as the reference lists of identified narrative reviews, published on diagnostic tests for asthma after 1997, and visited the Aridol website (accessed February 2019) to identify other relevant studies. A hand-search for conference proceedings of the American Thoracic Society (ATS), European Respiratory Society (ERS), and Chest and World Allergy Congress was performed to search for possible additional studies.
To be eligible for inclusion in the systematic review, studies had to fulfil the following criteria:
We excluded animal studies and “dose-finding studies”, as well as studies in which a two-by-two table could not be established even after contacting the relevant investigators. We took care to exclude duplicate studies.
In a first step, two reviewers independently screened titles and abstracts. Any articles that were deemed to be potentially relevant by one of the reviewers were marked. Studies that were judged to be ineligible by both reviewers based on the title and abstract were not assessed further. In a second step, the full texts of all the potentially eligible articles were retrieved so that they could be screened, again independently and in duplicate by two reviewers. Study eligibility was evaluated using pre-piloted forms with the above mentioned inclusion and exclusion criteria. Any disagreement was resolved by consensus. If consensus was not achieved, a third reviewer had the decisive vote.
The methodological quality and risk of bias of the selected studies was assessed independently and in duplicate by two reviewers with the Quality Assessment of Diagnostic Accuracy Studies tool (QUADAS-2; see appendix 2), which assessed common sources of bias in diagnostic studies.
From all the eligible studies after full text screening, two reviewers extracted predefined data independently and in duplicate using an extraction form (see appendix 3). Further data were extracted post hoc, including FEV1, atopy status and stopping of medication prior to testing. In cases of missing information from the articles, we contacted the authors of the respective studies to provide further details. If it was not possible to construct two-by-two tables, the studies were excluded from the final analysis. Controversies were resolved by discussion. If consensus was not achieved, a third reviewer had the decisive vote.
For each included study, we constructed two-by-two tables by comparing the results of the mannitol BPT with the respective reference standard. Sensitivity and specificity were the main measures of accuracy of the mannitol test, which we calculated using the data of the two-by-two tables. The true and false negative and positive rates were recorded. Sensitivity and specificity were plotted in receiver operating characteristic space. To explore the different populations, we grouped studies according to study design, study setting and age group in a forest plot.
The literature search provided a total of 836 citations after removing duplicates. After those in which the subject of this review was not addressed were excluded, 221 remained potentially relevant and were retrieved for full text screening (fig. 1). Finally, 27 studies [8–10, 12, 13, 15–36] met the inclusion criteria for our systematic review, all of them published in English between 1997 and 2018.

Figure 1 Identification of studies.
Eighteen of the included studies were cross-sectional studies [8–10, 12, 16–20, 26–30, 32, 34–36], nine studies used a case-controlled design [13, 15, 16, 25–29, 35], including asthmatics and a healthy control group.
The included studies involved a total of 4589 participants. The age range was from 6 to 85 years, and the percentage of males ranged from 25 to 100%. Most studies included only adults [8, 12, 15, 16, 21, 23–28, 30–33], eight studies included adults and children [9, 10, 13, 17, 18, 21, 23, 33], three studies included only children [19, 24, 26]. Twelve studies were conducted in a clinical setting [10, 13, 15, 17, 21–24, 28, 29, 32, 36], with participants attending any kind of clinical institution. The other studies used a nonclinical setting [8, 9, 12, 16, 18–20, 25–27, 30, 31, 33–35]; most of these included elite athletes as their study population, and two studies specifically investigated military conscripts and fire fighters [8, 12].
In most of the studies, having symptoms consistent with asthma was an inclusion criterion. However, in some studies, all of them in a nonclinical setting, having asthma symptoms was not required. Most of the case-controlled studies included a population that already had a diagnosis of asthma and a healthy control group.
In eight studies, it was mentioned that smokers were excluded [10, 15, 18, 26–29, 34]. Nine studies showed the numbers of current smokers included in the study [8, 9, 12, 16, 25, 31, 32, 35, 36], and in ten studies, no information was given on the smoking status of the participants [13, 17, 19–24, 30, 33].
In all the included studies, the mannitol challenge test was conducted according to the protocol by Anderson et al. or by the Aridol package leaflet.
The following reference standards were used. Twelve studies used “clinical diagnosis” as a reference standard [8–10, 12, 13, 17, 21, 23, 24, 32, 35, 36], eight studies used “test result” [15, 17, 20, 23, 24, 26, 31, 33], and seven studies used “physician diagnosed asthma” [16, 22, 29, 30, 32, 34, 35]. In eight studies, anti-asthmatic therapy was appropriately stopped prior to the tests [10, 20, 23, 25, 27, 33–35]. The individual characteristics of the included studies and their population are summarised in tables 1 and 2 , including information about atopy status and FEV1.
Table 1 Study and population characteristics of all studies included in the systematic review – I.
| Study author, year of publication | Country | Study design | Population setting | No. of participants analysed | Male, n (%) | Mean age, years | Age range, years | Health inclusion criteria | Mannitol test: protocol, definition of positive test | Reference standard | Sensitivity, % (95% CI) | Specificity, % (95% CI) | TP | FP | FN | TN |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Anderson, 1997 [ 15 ] | Australia | Case-control | Clinical: local community | 50 | Asthmatics: 11 (26%), non-asthmatics: not reported | 24 | 18–39 | Asthma diagnosis with current response to hypertonic saline and a healthy control group | Protocol by Anderson, FEV1 fall >20% (PD15 calculated) | Test result: 4.5% NaCl* | 99.0 (88.0–100.0) | 99.0 (52.0–100.0) | 43 | 0 | 0 | 7 |
| Anderson, 2009 [ 10 ] | USA | Cross-sectional | Clinical: university hospital | 375 | 182 (49%) | 24.3 | 6–50 | Asthma symptoms | Package leaflet, FEV1 fall >15% or dFEV1>10% between consecutive doses | Clinical diagnosis | 55.8 (49.3–62.2) | 74.8 (66.6–81.9) | 134 | 34 | 106 | 101 |
| Brannan, 2005 [ 13 ] | Australia | Case-control | Clinical: general population and from pulmonary function clinics | 592 | 272 (46%) | 34.7 | 6–83 | With or without Asthma symptoms | Protocol by Anderson, FEV1 fall >15% | Clinical diagnosis | 59.8 (55.2–64.1) | 95.2 (89.2–98.4) | 291 | 5 | 196 | 100 |
| Clearie, 2010 [ 22 ] | Scotland, UK | Cross-sectional | Nonclinical: elite swimmers | 59 | Not reported | 15.2 | Not reported | Athletes with and without physician diagnosed asthma | Package leaflet, FEV1 fall >15% or dFEV1>10% between consecutive doses | Physician diagnosed | 30.0 (6.7–65.2) | 89.8 (77.8–96.6) | 3 | 5 | 7 | 44 |
| Holzer, 2003 [ 23 ] | Australia | Cross-sectional | Nonclinical: elite athletes | 50 | 15 (30%) | 21 | 16–42 | Asthma symptoms or doctors diagnosis of asthma | protocol by Anderson, FEV1 fall >15% | Test result: eucapnic voluntary hyperpnoea challenge test* | 76.0 (55.0–91.0) | 92.0 (74.0–99.0) | 19 | 2 | 6 | 23 |
| Koskela, 2003 [ 25 ] | Finland | Case-control | Clinical: patients from outpatient clinic and healthy volunteers | 47 | 26 (55%) | Asthmatics: 49, non-asthmatics: 41 | 19–85 | Patients with recently diagnosed asthma and healthy control group | Protocol by Anderson, FEV1 fall >15% | Clinical diagnosis | 51.4 (34.4–68.1) | 100 (69.2–100) | 19 | 0 | 18 | 10 |
| Lund, 2009 [ 16 ] | Denmark | Case-control | Nonclinical: elite athletes and general population | 111 | 64 (58%) | Asthmatics: 24–27.8, non-asthmatics 20.4–25.1 | 18–35 | Elite athletes with and without asthma and non-athletes with and without asthma | Protocol by Anderson, FEV1 fall >15% | Physician diagnosed | 53.7 (37.4–69.3) | 95.7 (88–99.1) | 22 | 3 | 19 | 67 |
| Miedinger, 2007 [ 12 ] | Switzerland | Cross-sectional | Nonclinical: full time fire fighters | 94 | 94 (100%) | 41 | 23–64 | Fire fighters with and without physician diagnosed asthma | Protocol by Anderson, FEV1 fall >15% | Clinical diagnosis | 92.9 (66.1–99.8) | 97.5 (91.3–99.7) | 13 | 2 | 1 | 78 |
| Miedinger, 2010 [ 8 ] | Switzerland | Cross-sectional | Nonclinical: military conscripts | 235 | 235 (100%) | Not reported | 18–20 | Conscripts with and without physician diagnosed asthma | Package leaflet, FEV1 fall >15% or dFEV1>10% between consecutive doses | Clinical diagnosis | 40.5 (25.6–56.7) | 92.7 (88.1–96.0) | 17 | 14 | 25 | 179 |
| Sverrild, 2009 [ 9 ] | Denmark | Cross-sectional | Nonclinical: general population | 238 | 96 (40%) | 18.9 | 14–24 | None specified | Protocol by Anderson, FEV1 fall >15% | Clinical diagnosis | 58.8 (44.2–72.4) | 98.4 (95.4–99.7) | 30 | 3 | 21 | 184 |
| Aronsson, 2011 [ 28 ] | Sweden | Case-control | Clinical: outpatient department at a university hospital and control group | 49 | 23 (47%) | 35 | 21–65 | Asthma diagnosis and healthy control group | Package leaflet, FEV1 fall >15% or dFEV1>10% between consecutive doses | Clinical diagnosis | 38.2 (22.2–56.4) | 100 (78.2–100) | 13 | 0 | 21 | 15 |
| Subbarao, 2000 [ 26 ] | Canada | Case-control | Clinical: general population | 34 | 21 (60%) | 10 | 6–13 | Asthma diagnosis and healthy control group | Protocol by Anderson, FEV1 fall >20% (PD15 calculated) | Test result: Methacholine test* | 87.5 (67.6–97.3) | 100 (69.2–100) | 21 | 0 | 3 | 10 |
| Barben, 2011 [ 19 ] | Switzerland | Cross-sectional | Clinical: outpatient clinic | 99 | 63 (64%) | 12 | 6–17 | Asthma symptoms | Package leaflet, FEV1 fall >15% or dFEV1>10% between consecutive doses | Clinical diagnosis | 43.5 (31–56.7) | 94.6 (81.8–99.3) | 27 | 2 | 35 | 35 |
| Stenfors, 2010 [ 30 ] | Sweden | Cross-sectional | Nonclinical: cross-country skiing or biathlon athletes | 46 | 24 (52%) | 21 | 19–31 | None specified | Protocol by Anderson, FEV1 fall >15% | Physician diagnosed | 8.3 (1.0–27.0) | 95.5 (77.2–99.9) | 2 | 1 | 22 | 21 |
| McClean, 2011 [ 29 ] | Australia | Case-control | Nonclinical: workers at a research institute, hospital, university and volunteers | 67 | 31 (46%) | Asthmatics: 39.4, non-asthmatics: 34 | 18–66 | Asthma diagnosis and healthy control group | Protocol by Anderson, FEV1 fall >15% | Physician diagnosed | 61.5 (47.0–74.7) | 86.7 (59.5–98.3) | 32 | 2 | 20 | 13 |
| Romberg, 2012 [ 17 ] | Sweden | Cross-sectional | Nonclinical: elite swimmers | 97 | 55 (54%) | 16 | 13–17 | None specified | Protocol by Anderson, FEV1 fall >15% | Test result: exercise test* | 50.0 (18.7–81.3) | 78.2 (68.0–86.3) | 5 | 19 | 5 | 68 |
| Andregnette-Roscigno, 2012 [ 24 ] | Spain | Cross-sectional | Nonclinical | 23 | 14 (61%) | 12.9 | 7–17 | Asthma symptoms | Protocol by Anderson, FEV1fall >15% | Test result: methacholine test* | 55.6 (30.8–78.5) | 100 (47.8–100) | 10 | 0 | 8 | 5 |
| Ulrik, 2012 [ 18 ] | Denmark | Cross-sectional | Nonclinical: elite canoe and kayak athletes | 29 | 24 (83%) | 25.1 | 17–43 | None specified | Protocol by Anderson, FEV1 fall >15% | Clinical diagnosis | 42.9 (9.9–81.6) | 100 (84.6–100) | 3 | 0 | 4 | 22 |
| Kim, 2014 [ 27 ] | Korea | Case-control | Clinical: university hospital and control group | 104 | 30 (29%) | 43.8 | 18–70 | Asthma diagnosis and healthy control group | Protocol by Anderson, FEV1 fall >15% | Clinical diagnosis | 48.0 (33.7–62.6) | 92.6 (82.1–98.0) | 24 | 4 | 26 | 50 |
| Toennesen, 2014 [ 20 ] | Denmark | Cross-sectional | Nonclinical: elite athletes | 57 | 42 (74%) | 27.5 | not reported | Elite athletes participating at the Olympic Games 2008 with or without asthma symptoms | Protocol by Anderson, FEV1 fall >15% | Test result: mannitol or methacholine test* | 50.0 (26.0–74.0) | 92.3 (79.0–98.4) | 9 | 3 | 9 | 36 |
| De Menezes, 2018 [ 31 ] | Brazil | Cross-sectional | Nonclinical: workers at a university | 811 | 326 (40%) | 32.4 | not reported | Contact with laboratory animals | Protocol by Anderson, FEV1 fall >15% | Test result: Mannitol test* | 99.9 (93.1–99.9) | 96.7 (95.1–97.8) | 66 | 24 | 0 | 721 |
| White, 2017 [ 32 ] | Australia | Cross-sectional | Clinical: wheezing population and general population | 920 | Not reported | 22 | 21–23 | Wheezing population: wheezing, general population: no wheezing | Protocol by Anderson, FEV1fall >15% | Physician diagnosis | 19.5 (13.6–26.6) | 97.1 (95.7–98.2) | 30 | 22 | 124 | 744 |
| Cancelliere, 2013 [ 33 ] | Spain | Cross-sectional | Clinical: university hospital | 28 | 7 (25%) | 32 | 15–54 | Asthma-like symptoms (shortness of breath, wheezing, cough) | Protocol by Anderson, FEV1 fall >15% | Test result: Methacholine test and/or Mannitol test* | 87.5 (51.6–97.9) | 99.7 (75.0–100.0) | 10 | 0 | 1 | 17 |
| Osthoff, 2013 [ 34 ] | Switzerland | Cross-sectional | Nonclinical: Elite athletes (Swiss paralympic team) | 44 | 30 (68%) | 34.4 | not reported | non specified (Swiss paralympic team) | Protocol by Anderson, FEV1 fall >10% | Physician diagnosed | 55.0 (23.4–83.3) | 94.0 (79.8–99.3) | 6 | 2 | 5 | 31 |
| Backer, 2015 [ 21 ] | Denmark | Cross-sectional | Clinical: university hospital | 190 | 82 (43%) | 32.1 | 15–not reported | Symptoms suggesting asthma | Protocol by Anderson, FEV1 fall >15% | Clinical diagnosis | 38.0 (34.0–44.0) | 82.0 (71.0–89.0) | 46 | 11 | 76 | 57 |
| Porpodis, 2016 [ 36 ] | Greece | Cross-sectional | Clinical: university hospital | 88 | 41 (47%) | 38.6 | not reported | Asthma-like symptoms (shortness of breath, wheezing, cough) | Protocol by Anderson, FEV1 fall >15% | Clinical diagnosis | 64.0 (51.5–75.5) | 95.0 (76.2–99.9) | 43 | 1 | 24 | 20 |
| Vakali 2016 [ 35 ] | Greece, UK | Case-control | Nonclinical: elite athletes | 200 | 100 (50%) | Asthmatics: 20.4, non-asthmatics: 22.1 | 20.7–22.5 | Athletes with and without physician diagnosed asthma | Protocol by Anderson, FEV1 fall >15% | Physician diagnosed | 21.8 (12.0–35.0) | 95.0 (89.4–97.6) | 12 | 8 | 43 | 137 |
CI = confidence interval; FEV1 = forced expiratory volume in 1 second; TP = true positive; FP = false positive; FN = false negative; TN = true negative * A positive test result was only accepted as a reference standard when the included subjects had respiratory symptoms
Table 2 Study and population characteristics of all included studies in the systematic review – II.
| Study author, year of publication | Smoking status | Appropriate stop of antiasthmatic therapy prior to test* | Asthma related symptoms | Time between asthma diagnosis and mannitol test | FEV1 (L) asthma | FEV1(% pred) asthma | FEV1 (L) control | FEV1 (% pred) control | FEV1 (L) all | FEV1 (% pred) all | Atopy in asthmatics | Atopy in controls | Atopy in all | Publication status |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Anderson, 1997 [ 15 ] | All non-smokers | No | Asthmatics: yes, non-asthmatics: no symptoms | Not reported | Not reported | 82.9 (SD 12.9) | Normal values | Normal values | Not reported | Not reported | 100% | 43% | 92% | Peer-reviewed |
| Anderson, 2009 [ 10 ] | All non-smokers | Yes | Yes, current | Couple of weeks | Not reported | Not reported | Not reported | Not reported | 3.32 (SD 0.82) | 93.6 (SD 10) | Not reported | Not reported | 16% to 50% | Peer-reviewed |
| Brannan, 2005 [ 13 ] | Not reported | No | Asthmatics: yes, current, non-asthmatics: no symptoms | Simultaneously | Not reported | Not reported | Not reported | Not reported | 3.0 (SD 0.9) | 95.0 (SD 14.5) | Not reported | Not reported | Not reported | Peer-reviewed |
| Clearie, 2010 [ 22 ] | Not reported | No | Yes, 26 (43%) with exercise induced symptoms | 3 days | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | 14 | 24% | Peer-reviewed |
| Holzer, 2003 [ 23 ] | Not reported | Yes | Yes, current | 1 week | Not reported | Not reported | Not reported | Not reported | Within normal limits (>80% of predicted) | 103.6 (SD 10.8) | Not reported | Not reported | Not reported | Peer-reviewed |
| Koskela, 2003 [ 25 ] | Current smokers: 6; former smokers: 14; never smoked: 27 | Yes | Asthmatics: yes, current, non-asthmatics: no symptoms | 2 weeks | 2.9 (95% CI 2.6–3.1) | 81 (95% CI 76–86) | 3.9 (95% CI 3.1–4.7) | 99 (95% CI 95–104) | Not reported | Not reported | 38% | 10% | Not reported | Peer-reviewed |
| Lund, 2009 [ 16 ] | Current smokers: 7 | Elite athletes: no, non-athletes: yes | All athletes (54): yes | Simultaneously | 4.37 (SD 0.19) for elite athletes; 3.73 (SD 0.14) for non-elite athletes | 98.2 (SD 1.94) for elite athletes; 82.6 (SD 2.63) for non-elite athletes | 4.91 (SD 0.15) for elite athletes, 3.87 (SD 0.13) for non-elite athletes | Elite: 105.4 (SD 2.10); non-elite: 96.9 (SD 1.82) | Not reported | Not reported | Elite athletes: 21%; non-elite athletes: 100% | Elite athletes: 51%; non-elite athletes: 29% | Not reported | Peer-reviewed |
| Miedinger, 2007 [ 12 ] | Current smokers: 33 | Not reported | Asthmatics: yes, in the past 12 months, non-asthmatics: unclear | 1 week | Not reported | Range of 42–105 | Not reported | Not reported | Not reported | 103 (SD 12) | 86% | Not reported | 51% | Peer-reviewed |
| Miedinger, 2010 [ 8 ] | Current smokers: 77 | Not reported | Asthmatics: yes, in the past 12 months, non-asthmatics: unclear | 48 hours | Not reported | 95 (IQR 88;102) | Not reported | 98 (IQR 91;105) | Not reported | Not reported | 74% | 36% | 42% | Peer-reviewed |
| Sverrild, 2009 [ 9 ] | Current smokers: 52 | No | Asthmatics: yes, in the past 12 months, non-asthmatics: unclear | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | 96.92 (SD 10.60) | Not reported | Not reported | 42% | Peer-reviewed |
| Aronsson, 2011 [ 28 ] | All non-smokers | No | Asthmatics: current exercise induced symptoms 27 (79%), non-asthmatics: no symptoms | A couple of weeks | 3.7 (SD 1.0) | 95.5 (SD 14.2) | 3.6 (SD 0.8) | 98.6 (SD 6.8) | Not reported | Not reported | 82% | 0% | Not reported | Peer-reviewed |
| Subbarao, 2000 [ 26 ] | All non-smokers | No | Asthmatics: yes, current, non-asthmatics: no symptoms | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | 87% | 0% | Not reported | Peer-reviewed |
| Barben, 2011 [ 19 ] | Not reported | No | Yes, current | A few days | Not reported | Not reported | Not reported | Not reported | Not reported | 97 (IQR 88;104) | Not reported | Not reported | 89.0% | Peer-reviewed |
| Stenfors, 2010 [ 30 ] | Not reported | No | In 17% classical Symptoms of exercise induced asthma | Not reported | 4.7 (SD 1.1) | 97.7 (SD 14.0) | 4.5 (SD 0.5) | 101.4 (SD 7.5) | Not reported | Not reported | 55% | 50% | Not reported | Peer-reviewed |
| McClean, 2011 [ 29 ] | All non-smokers | No | Asthmatics: were well controlled | Not reported | Not reported | 87 (SD 13.0) | Not reported | 104 (SD 14.4) | Not reported | Not reported | 83% | 53% | Not reported | Peer-reviewed |
| Romberg, 2012 [ 17 ] | Not reported | No | Exercise induced symptoms 75 (77.3%), current asthma symptoms 60 (62.0%), current asthma symptoms with exercise induced symptoms 54 (55.7%): past 12 months | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | 112 (IQR 104;118) | Not reported | Not reported | 54% | Peer-reviewed |
| Andregnette-Roscigno, 2012 [ 24 ] | Not reported | No | Yes | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | 70% | Peer-reviewed |
| Ulrik, 2012 [ 18 ] | All non-smokers | No | Asthmatics with previous diagnosis of asthma: yes, asthmatics with no previous diagnosis of asthma: no, non-asthmatics: not reported | Simultaneously | 4.5 (SD 0.6) | 103.3 (SD 13.3) | 4.8 (SD 0.9) | 109.1 (SD 14.8) | Not reported | Not reported | 57% | 18% | Not reported | Peer-reviewed |
| Kim, 2014 [ 27 ] | All non-smokers | Yes | Asthmatics: yes, in the past 6 months, non-asthmatic: no | Not reported | Not reported | 91.2 (SD 12.2) | Not reported | 95.0 (SD 19.4) | Not reported | Not reported | Allergic rhinitis 66%, atopic dermatitis 18%, allergic conjunctivitis 22% | allergic rhinitis 15%, atopic dermatitis 0%, allergic conjunctivitis 2% | allergic rhinitis 39%, atopic dermatitis 9%, allergic conjunctivitis 12% | Peer-reviewed |
| Toennesen, 2014 [ 20 ] | Not reported | Yes | Asthmatics: yes (unclear if current or in the past), non-asthmatics: no | Not reported | Not reported | 117 (SD 15) | Not reported | 117.3 (SD 11.8) | Not reported | 117.2 (12.7) | 17% | 18% | 18.00% | Peer-reviewed |
| De Menezes, 2018 [ 31 ] | Current smokers: 69 (8.5%) | No | Yes, in the past 12 months | Not reported | 3.35 (SD 0.64) | 91.8 (SD 11.5) | 3.54 (SD 0.75) | 97.4 (SD 11.3) | Not reported | Not reported | Not reported | Not reported | 47% | Peer-reviewed |
| White, 2017 [ 32 ] | Current smokers: 158 | No | General population: unclear, wheezing population: 148 (100%) in the past 12 months | Not reported | Not reported | General population: 95 (SD 11.60), wheezing population: 95 (SD 11.58) | Not reported | General population: 98 (SD 10.76); wheezing populationd: 98 (SD 10.23) | Not reported | Not reported | General population: 75%, wheezing population 75% | General population: 55%; wheezing population: 62% | Not reported | Peer-reviewed |
| Cancelliere N, 2013 [ 33 ] | Not reported | Yes | Yes, current | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Not reported | Peer-reviewed |
| Osthoff, 2013 [ 34 ] | All non-smokers | Yes | Asthmatics: yes, current, non-asthmatics: unclear | Not reported | Not reported | Not reported | Not reported | Not reported | 3.63 | Not reported | 32% | Not reported | Not reported | Peer-reviewed |
| Backer, 2015 [ 21 ] | Only reported that individuals older than 40 years with > 10 pack-years were excluded | No | Yes, current | 2–3 weeks | 3.7 (SD 0.9) | 97 (SD 17) | 3.8 (0.9) | 97 (SD 17) | 3.8 (SD 0.9) | 95 (SD 17) | 63% | 44% | 56% | Peer-reviewed |
| Porpodis, 2016 [ 36 ] | Current smokers: 17, former smokers: 16, never smoked: 55 | No | Yes, in the last month | 14–20 days | Not reported | Not reported | Not reported | Not reported | 3.2 (SD 0.9) | 88.5 (SD 12.6) | Not reported | Not reported | 43% | Peer-reviewed |
| Vakali, 2016 [ 35 ] | Smokers: 8% | Yes (but refer to ATS Crapo criteria) | Yes | Not reported | 4.07 (95% CI 3.9–0.2) | Not reported | 4.29 (95%CI 4.1–4.5) | Not reported | 4.1 (95%CI 4.1–4.2) | Not reported | 62% | 44% | 49% | Peer-reviewed |
CI = confidence interval; FEV1 = forced expiratory volume in 1 second; IQR = interquartile range; SD = standard deviation * according to Anderson et al. [10]
Overall, the quality of the included studies was good with a low risk of bias from the procedure and interpretation of the mannitol test and the patient flow. Only nine studies mentioned that the results of the mannitol test were interpreted without knowing the results of the reference standard [8, 10, 12, 13, 17, 20, 21, 24, 34]. The domains “patient selection” and “reference standard” showed heterogeneous results concerning methodological quality (see appendix 4). The risk of bias in the patient selection was high in all the studies that used a case-controlled design [13, 15, 16, 25–29, 35]. Applicability was judged to be limited for studies that included only a special population such as elite athletes, young male military conscripts or fire fighters [8, 12, 16, 18, 19, 26, 30, 31, 33–35], and studies, that excluded smokers [10, 15, 18, 26–29, 34]. Concerning the reference standard, the risk of bias was usually rated low in all studies that used a “clinical diagnosis of asthma” as a reference standard, and remained unclear in studies using “physician diagnosed asthma” as a reference standard. In seven studies, we observed a high risk of bias as the mannitol test was part of the reference standard and blinding of the test results was not done [8, 12, 18–20, 31, 33].
Overall, sensitivity and specificity were very heterogeneous, with values ranging from 8% (95% CI 1–27%) to 100% (95% CI 93–100%) for sensitivity and 75% (95% CI 67–82%) to 100% (95% CI 85–100%) for specificity. [8–10, 12, 13, 15–36,] We graphically presented the high level of between-study heterogeneity in the ROC space plot in figure 2. To explore the different populations, we grouped studies according to the populations (clinical vs nonclinical and children vs adults and mixed; fig. 3). As case-controlled studies represent the highest risk for bias [37], we tabulated them in a separate forest plot in figure 3.
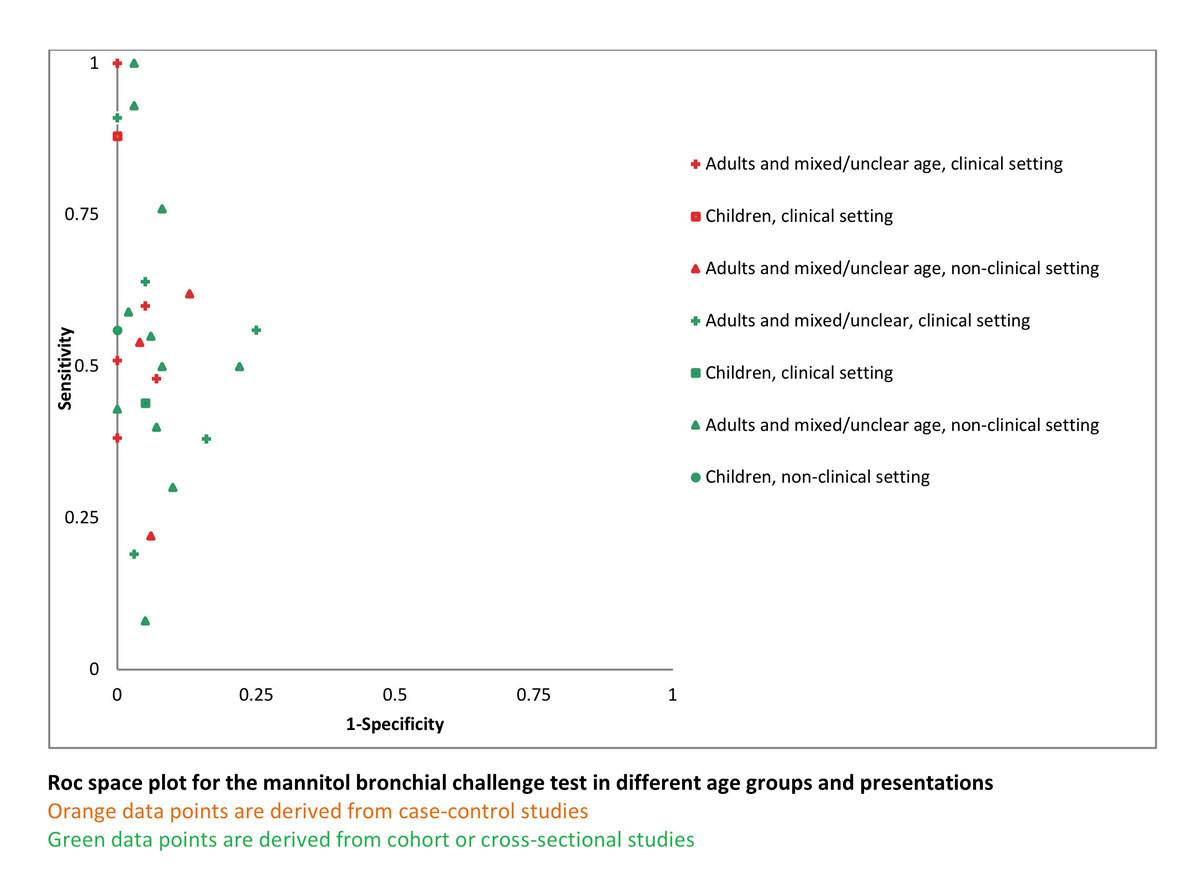
Figure 2 ROC space plot of all included studies (n = 27)
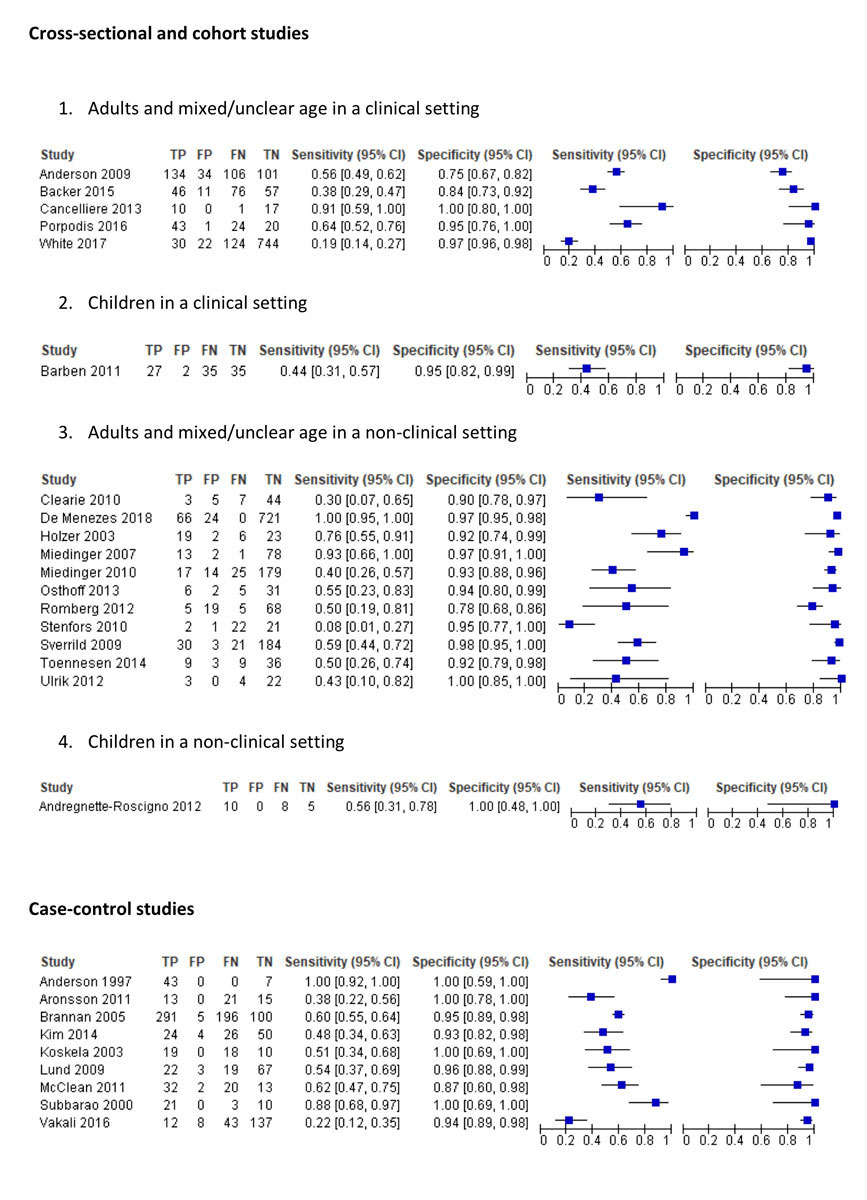
Figure 3 Forest plot of the sensitivity and specificity of the mannitol test showing several subgroups of the included cross-sectional studies (n = 18) and case-controlled studies (n = 9).
TP = true positive; FP = false positive; FN = false negative; TN = true negative; CI = confidence interval
When all case-control studies were excluded, cross sectional and cohort studies conducted in a clinical setting showed a range from 19% (95% CI 14–27%) to 91% (95% CI 59–100%) for sensitivity and from 75% (95% CI 67–82%) to 100% (95% CI 80–100%) for specificity [10, 19, 21, 32, 33, 36]. Cross-sectional and cohort studies conducted in a nonclinical setting showed a range from 8% (95% CI 1–27%) to 100% (95% CI 95–100%) for sensitivity and from 78% (95% CI 68–86%) to 100% (95% CI 85–100%) for specificity [8, 9, 12, 16, 18–20, 26, 27, 30, 34, 35]. In all studies that considered patients who had stopped asthma medication appropriately prior to testing, sensitivity and specificity ranged from 22% (95% CI 12–35%) to 91% (95% CI 59–100%) and from 75% (95% CI 67–82%) to 100% (95% CI 80–100%), respectively [10, 20, 23, 25, 27, 33–35]. In the studies that did not stop asthma medication appropriately, sensitivity and specificity ranged from 8% (95% CI 1–27%) to 100% (95% CI 95–100%) and from 78% (95% CI 68–86%) to 100% (95% CI 85–100%). [9, 13, 15, 17, 18, 22, 24–28, 32–36]
We found a high level of heterogeneity among the included studies and explored reasons for this by assessment of the different study designs and methods used, population characteristics such as atopy status and smoking, and risk of bias. We explored populations further by dividing them into subgroups, showing forest plots as well as giving the range of accuracy. There is no evidence that accuracy of the mannitol tests differs according to the populations examined.
Reasons for the heterogeneity in sensitivity and specificity of the mannitol test have previously been discussed and may be false negative and false positive mannitol tests. One reason for the heterogeneity in sensitivity and specificity may be current asthma treatment and lack of current asthma inflammation, which may lead to false negative results as the mannitol test just shows how many participants have active asthma at the time of assessment. A positive result for mannitol indicates the presence of inflammatory cells and a sufficient concentration of mediators to cause bronchoconstriction. A negative test result indicates that one of these elements is missing, such as is the case in treated asthmatic patients with inhaled corticosteroids (ICS), β2 agonists or leukotriene inhibitors [38, 39]. An example for this circumstance is the study of Brannan et al., who assessed that the sensitivity of the mannitol test to identify asthma was 59.8%, which rose to 88.9% when those asthmatics taking ICS, who were negative to mannitol challenge, were excluded [13]. In a later analysis of the adult data from this study, Brannan et al. reported that 49% of the asthmatic patients taking ICS daily were negative to mannitol [40]. This is confirmed by our analysis in which sensitivity was higher in studies in which asthma medication was stopped for an appropriately long time according to Anderson et al. than in those studies in which this was not the case [10].
Another case in which a mannitol test can be negative is when a trigger for asthma is taken away and no asthma inflammation is present, which has been seen for reduced exposure to house dust mites in house dust mite allergic patients [41]. Overall, the fact that the specificity was high demonstrates that there are few false positive tests. A reason for false positive results could be current smoking, which has been previously examined by Stolz et al. [42] Current smokers were excluded in several of the examined studies, a possible reason for the few false positive tests.
To a lesser degree, different reference standards may have caused heterogeneity. Even though the GINA guidelines give advice on the best diagnostic approach in asthma, a single gold-standard test does not exist [1]. We therefore accepted various reference standards.
The ROC space plot indicated that there are some studies that are clearly separate from the main group and which we would like to discuss in more detail.
In this context we need to discuss those studies in which the mannitol test was not only the index test but was also included in the reference standard, as this may lead to an overestimation of sensitivity and specificity. Looking at the studies by Miedinger [12], De Menezes [31] and Cancelliere [33], we find support for this assumption as all three show exceptionally high sensitivity. As we only included studies that combined clinical symptoms with the mannitol test result to make a diagnosis, we still assume that these are truly asthmatic patients. However, incorporation bias is of concern and we did acknowledge the risk of bias to be high (see appendix 4). An exclusion of the studies including mannitol in their reference standard may have caused an inclusion bias, as a a certain phenotype of asthma may have been selected.
In contrast, the studies by Clearie [22], Stenfors [30], Vakali [35] and White [32] showed lower sensitivity than the other studies. The studies by Clearie, Stenfors and Vakali were all conducted in nonclinical settings. They included athletes. In the study by Clearie, the fact that the elite swimmers continued to take inhaled corticosteroids before testing could be another reason for the sensitivity of only 30%, as corticosteroid treatment has been shown to inhibit indirect bronchial hyperresponsiveness, as mentioned previously [13]. In the study by Stenfors [30], the slection of asthmatic athletes were probably biased towards subjects with mild and/or well-controlled asthma, as only 21% of them had bronchial hyperresponsiveness, 29% had experienced shortness of breath post-exercise and 15% had an asthma attack in the last 12 months. Additionally, 37% were taking anti-inflammatory medicine, including steroids. As the mannitol protocol is fixed, it is not possible to administrate additional doses to elucidate a response in those with mild asthma, which may cause false negative results and thus low sensitivity in this situation [38]. In the study by White [32], the use of ICS might be a reason for the low sensitivity of 19%. However, even after participants with a negative mannitol test who were using ICS were excluded, there was no real change in sensitivity. A reason for the low sensitivity in this study might be the fact that all participants seem to have only mild asthma, showing normal pulmonary function and only a few reporting the use of ICS in the past 12 months.
The mannitol test showed high specificity diagnosing asthma in all studied populations independent of age group or study setting.
Due to its heterogeneity, we cannot postulate a certain phenotype of asthma in which mannitol is specifically useful. Previous studies reported a higher specificity and correlation with eosinophilic asthma [38, 43–46], which we cannot confirm because of the present heterogeneity of the studies. What we do see is the strength of the mannitol test to confirm asthma owing to the high specificity showed in our systematic review. This, as well as the high practicability of the mannitol test, could make it a useful diagnostic test in certain population groups such as athletes in a nonclinical setting.
As our systematic review focused on the mannitol test and we did not include a review on methacholine test, a direct comparison with a direct test such as methacholine is therefore not possible. From the literature it is known that the methacholine test shows a high sensitivity and a high negative predictive value and therefore seems to be a good test to exclude asthma. It therefore, also due to the low sensitivity of the mannitol test emphasised in our review, remains the test of choice to exclude asthma in patients with symptoms that suggest asthma, but are caused by another condition [38].
This was the first systematic review assessing the accuracy of the mannitol test in diagnosing asthma. In 2011, a systematic review assessed the accuracy of the mannitol test, but the diagnosis differed from our systematic review as this review concentrated on the diagnosis of exercise-induced bronchoconstriction, and the objective differed as it focussed on comparing eucapnic voluntary hyperpnoea and mannitol with standard exercise challenge testing [47]. Three studies were detected of which only one was also included in our systematic review [10]. Comparable to our results they found that there are only a few studies that assessed the accuracy of the mannitol test, and that heterogeneity was high [47]. Several reviews have discussed the advantages and disadvantages of indirect challenge tests, including mannitol, but no systematic review has been performed with an assessment of the risk of bias of publications [38, 39, 48, 49].
We conducted this systematic review according to a prespecified protocol, using a comprehensive literature search strategy and multiple reviewers, which strengthened the analysis by avoiding publication bias and selection bias. Another strength was the application of the Quality Assessment of Diagnostic Accuracy Studies instrument [14].
Our systematic review has limitations. There is a possibility of publication bias as we may have missed some studies despite systematic screening. There is also the risk of study selection bias, which we aimed to overcome by using two independent reviewers. Another limitation was that not all studies were designed as accuracy studies and methodological issues limited the generalisability of the results. Even though the general quality of the included studies was good, one concern was the blinding of the assessors, which was poorly reported. Studies followed a strict, predefined protocol of the mannitol test, and it was therefore deemed unlikely that a lack of blinding would have caused bias. In the studies where the methacholine test was part of the reference standard, the lack of blinding was not an important concern, as the methacholine test is an objective test. Because of selection bias, we plotted case-controlled studies separately. Our main concern about the case-controlled design was selection bias, as patients who have difficult-to-diagnose asthma are usually not included. This may lead to an overestimation of sensitivity and specificity.
The 27 studies included in the systematic review showed very heterogeneous results concerning the accuracy of the mannitol test in the diagnosis of asthma. This may have been caused by different study methods that resulted in false negative or false positive tests, as well as different study settings, populations and reference standards. This between-study heterogeneity hindered the formation of a conclusive statement on the accuracy of the mannitol test and there needs to be further research. In future studies, factors that may influence sensitivity and specificity such as smoking, stopping of asthma medications and current asthma symptoms, FEV1 and atopy status (information about seasonal allergies and time of assessment) should be considered and clearly stated.
Because of the high specificity the mannitol test showed in our systematic review, it seems to be a good test to confirm a diagnosis of asthma. This, and the advantage of a standardised protocol with an easy and safe test procedure, can make it a good diagnostic tool also in a nonclinical setting. To exclude asthma, however, methacholine seems to remain the test of choice as the literature shows high sensitivity, whereas in our review we could often only show low sensitivity and heterogeneous results for the mannitol test.
As bronchial provocation tests can be especially useful in patients with an intermediate probability of asthma diagnosis, further studies are needed that include subjects with asthma symptoms but an intermediate probability of asthma diagnosis. In these studies, a longitudinal follow-up would be useful in order to verify the diagnosis and establish an appropriate reference standard.
The search strategy included the term (mannitol) combined with (asthma OR bronchial* OR bronchoconstrict*). In Embase the terms were: ‘mannitol’/exp OR ‘mannitol’/syn AND (‘asthma’/exp OR ‘asthma’/syn OR bronchial* OR ‘bronchoconstriction’/exp OR ‘bronchoconstriction’/syn), in PubMed/Medline they were: mannitol AND (asthma OR bronchial* OR bronchoconstrict*). For our search there was no language restriction imposed.
The methodological quality of the selected studies was graded independently and in duplicate by two reviewers with the Quality Assessment of Diagnostic Accuracy Studies tool (QUADAS-2, an improved, redesigned tool since the original QUADAS tool), a validated tool for the quality assessment of diagnostic accuracy studies [14]. The QUADAS-2 tool includes 4 domains (patient selection, index test, reference standard, and flow and timing). Each domain is assessed in terms of risk of bias, and the first 3 domains also in terms of concerns regarding applicability. Some signalling questions are included to help judge the risk of bias. We added some signalling questions to the predefined QUADAS-2 form, which seemed to be important to us judging our included studies. Disagreements were resolved by consensus. If consensus was not achieved, a third reviewer had the decisive vote.
The two reviewers extracted data on characteristics of studies and patients, the index test and the reference standard as well as test results. Wherever possible, we recorded for each study: Title, author, year of publication, country and journal, conflict of interest and project funding, study aim, study design (cohort study, case-control study, cross-sectional or later follow-up, prospective or retrospective), study population (age, prevalence of asthma, severity of symptoms, co morbidity, smoking status, gender, professional sportsmen, history of asthma, atopy), Stop of anti-asthmatic therapy prior to the index test, Patient selection (consecutive, non-consecutive, random- sample, inclusion- and exclusion criteria), technical details of Mannitol bronchial provocation and reference tests regarding standardisation (protocol, definition for positive/negative test result), performance of the index test (sensitivity and specificity), number of individuals eligible and no of individuals who underwent the tests, number of individuals undergoing either the index and the reference test missing one or the other, time interval between the index and the reference test, side effects from undergoing either the Mannitol bronchial provocation test or reference standard, number of individuals in whom the test was terminated prematurely or was not analysable, reasons for exclusion from test or analysis, inter-observer variability and test reproducibility, reported results (Sensitivity, Specificity, true positive, true negative, false positive, false negative, positive predictive value, negative predictive value), Data for two-by-two table.
| Anderson, 1997[15] | Anderson, 2009[10] | Brannan, 2005[13] | Clearie, 2010[22] | Holzer, 2003[23] | Koskela, 2003[25] | Lund, 2009[16] | Miedinger, 2007[12] | Miedinger, 2010[8] | Sverrild, 2009[9] | Aronsson, 2011[28] | Subbarao, 2000[26] | Barben, 2011[19] | Stenfors, 2010[30] | McClean, 2011[29] | Romberg, 2012[17] | Andregnette-Roscigno, 2012[24] | Ulrik, 2012[18] | Kim, 2014[27] | Toennesen, 2014[20] | De Menezes, 2018[31] | White, 2017[32] | Cancelliere 2013[33] | Osthoff, 2013[34] | Backer, 2015[21] | Porpodis, 2016[36] | Vakali, 2016[35] | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PATIENT SELECTION | |||||||||||||||||||||||||||
| Consecutive or random sample enrolment? | unclear | unclear | unclear | unclear | unclear | yes | unclear | no | yes | yes | unclear | unclear | unclear | unclear | unclear | unclear | unclear | unclear | unclear | unclear | no | no | yes | yes | yes | yes | unclear |
| Case-control design avoided? | no | yes | no | yes | yes | no | no | yes | yes | yes | no | no | yes | yes | no | yes | yes | yes | no | yes | yes | yes | yes | yes | yes | yes | no |
| Inappropriate exclusions avoided? | no | no | yes | yes | yes | yes | yes | yes | yes | yes | no | no | yes | yes | no | unclear | unclear | no | no | yes | no | yes | yes | no | no | unclear | no |
| Risk of Bias | HIGH | LOW | HIGH | LOW | LOW | HIGH | HIGH | LOW | LOW | LOW | HIGH | HIGH | LOW | LOW | HIGH | UNCLEAR | LOW | LOW | HIGH | LOW | LOW | LOW | LOW | LOW | LOW | LOW | HIGH |
| Applicability concerns | HIGH | HIGH | LOW | HIGH | HIGH | LOW | HIGH | HIGH | HIGH | LOW | HIGH | HIGH | LOW | HIGH | HIGH | HIGH | LOW | HIGH | HIGH | HIGH | HIGH | LOW | LOW | HIGH | LOW | LOW | HIGH |
| INDEX TEST (MANNITOL) | |||||||||||||||||||||||||||
| Description of Index Test | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes |
| Results interpreted without knowing results of the reference standard (Blinding) | no | yes | yes | unclear | unclear | unclear | unclear | unclear | yes | yes | no | unclear | no | unclear | unclear | yes | yes | unclear | unclear | yes | unclear | unclear | no | yes | yes | unclear | unclear |
| If a threshold was used, was it pre-specified? | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes |
| Risk of Bias | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW |
| Applicability concerns | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW |
| REFERENCE STANDARD | |||||||||||||||||||||||||||
| Description of Reference standard | yes | yes | yes | no | no | yes | no | yes | yes | yes | yes | yes | yes | no | no | yes | yes | yes | yes | yes | yes | no | yes | no | yes | yes | no |
| Results interpreted without knowing results of the Mannitol test (Blinding) | yes | yes | yes | yes | yes | yes | yes | no | no | yes | yes | yes | no | yes | yes | unclear | unclear | no | yes | no | no | unclear | no | unclear | yes | yes | yes |
| Adequate Reference standard | yes | yes | yes | unclear | yes | yes | unclear | yes | yes | yes | yes | yes | yes | unclear | unclear | yes | yes | yes | yes | yes | yes | unclear | yes | unclear | yes | yes | unclear |
| Mannitol Test was not part of the Reference standard (no incorporation bias) | yes | yes | yes | yes | yes | yes | yes | no | no | yes | yes | yes | no | unclear | unclear | yes | yes | no | yes | no | no | yes | no | unclear | yes | yes | unclear |
| Risk of Bias | LOW | LOW | LOW | UNCLEAR | LOW | LOW | UNCLEAR | HIGH | HIGH | LOW | LOW | LOW | HIGH | UNCLEAR | UNCLEAR | LOW | LOW | HIGH | LOW | HIGH | HIGH | UNCLEAR | HIGH | UNCLEAR | LOW | LOW | UNCLEAR |
| Applicability concerns | LOW | LOW | LOW | UNCLEAR | LOW | LOW | UNCLEAR | LOW | LOW | LOW | LOW | LOW | LOW | UNCLEAR | UNCLEAR | HIGH | LOW | LOW | LOW | LOW | LOW | UNCLEAR | LOW | UNCLEAR | LOW | LOW | UNCLEAR |
| FLOW AND TIMING | |||||||||||||||||||||||||||
| Appropriate time intervall between Mannitol test and reference standard (>24h and <1week) | unclear | unclear | yes | unclear | unclear | no | unclear | yes | unclear | unclear | unclear | yes | yes | unclear | unclear | unclear | yes | unclear | unclear | unclear | no | unclear | no | yes | unclear | no | unclear |
| Therapeutic intervention avoided between Index test and reference standard | yes | yes | no | unclear | unclear | yes | unclear | yes | unclear | yes | unclear | yes | yes | unclear | no | yes | unclear | no | unclear | yes | no | no | no | yes | unclear | unclear | unclear |
| Did all patients receive a reference standard | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | no | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes |
| Did all patients receive the same reference standard | yes | yes | yes | unclear | unclear | yes | unclear | yes | unclear | yes | unclear | yes | no | unclear | unclear | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes | yes |
| Were all patients included in the analysis | yes | unclear | no | no | yes | no | yes | no | no | no | no | no | no | yes | yes | no | yes | yes | yes | yes | no | yes | yes | yes | yes | yes | yes |
| Were reasons reported for stopping a test | not applicable | yes | yes | yes | not applicable | not applicable | not applicable | yes | yes | yes | yes | yes | yes | not applicable | not applicable | not applicable | not applicable | not applicable | not applicable | not applicable | yes | not applicable | not applicable | not applicable | not applicable | not applicable | not applicable |
| Risk of Bias | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | LOW | UNCLEAR | UNCLEAR | LOW | LOW | LOW | LOW | HIGH | HIGH | HIGH | LOW | LOW | LOW | LOW |
The authors would like to thank Gill Worthy for her support in data analysis and figure creation.
This study was supported by the Allergiestiftung Ulrich Müller-Gierok.
Prior to this study, David Miedinger and Jörg Leuppi received free mannitol bronchial provocation test kits from Pharmaxis Ltd. to perform clinical studies investigating patients with asthma/COPD or individuals in the workforce.
1GINA Board. Global Strategy for Asthma management and prevention [Internet]. 2015 [cited 2015 Dec 1]. Available from: http://www.ginasthma.org/local/uploads/files/GINA_Report_2015.pdf
2Fanta CH. Diagnosis of asthma in adolescents and adults. UpToDate. 2015. Available from: https://www.uptodate.com/contents/diagnosis-of-asthma-in-adolescents-and-adults.
3 Leuppi JD , Brannan JD , Anderson SD . Bronchial provocation tests: the rationale for using inhaled mannitol as a test for airway hyperresponsiveness. Swiss Med Wkly. 2002;132(13-14):151–8.
4 Coates AL , Wanger J , Cockcroft DW , Culver BH , Carlsen K-H , Diamant Z , et al., Bronchoprovocation Testing Task Force: Kai-Håkon Carlsen. ERS technical standard on bronchial challenge testing: general considerations and performance of methacholine challenge tests. Eur Respir J. 2017;49(5):1601526. doi:.https://doi.org/10.1183/13993003.01526-2016
5 Porsbjerg C , Rasmussen L , Thomsen SF , Brannan JD , Anderson SD , Backer V . Response to mannitol in asymptomatic subjects with airway hyper-responsiveness to methacholine. Clin Exp Allergy. 2007;37(1):22–8. doi:.https://doi.org/10.1111/j.1365-2222.2006.02614.x
6 Anderson SD . Provocative challenges to help diagnose and monitor asthma: exercise, methacholine, adenosine, and mannitol. Curr Opin Pulm Med. 2008;14(1):39–45. doi:.https://doi.org/10.1097/MCP.0b013e3282f197f6
7Pharmaxis Ltd. Product Information Aridol [Internet]. 2014 [cited 2016 Apr 1]. p. 1–7. Available from: http://www.aridol.info/assets/pdf/20140626_Aridol_PI_AUS.pdf
8 Miedinger D , Mosimann N , Meier R , Karli C , Florek P , Frey F , et al. Asthma tests in the assessment of military conscripts. Clin Exp Allergy. 2010;40(2):224–31. doi:.https://doi.org/10.1111/j.1365-2222.2009.03387.x
9 Sverrild A , Porsbjerg C , Thomsen SF , Backer V . Diagnostic properties of inhaled mannitol in the diagnosis of asthma: a population study. J Allergy Clin Immunol. 2009;124(5):928–32.e1. doi:.https://doi.org/10.1016/j.jaci.2009.06.028
10 Anderson SD , Charlton B , Weiler JM , Nichols S , Spector SL , Pearlman DS ; A305 Study Group. Comparison of mannitol and methacholine to predict exercise-induced bronchoconstriction and a clinical diagnosis of asthma. Respir Res. 2009;10(1):4. doi:.https://doi.org/10.1186/1465-9921-10-4
11 Rundell KW , Slee JB . Exercise and other indirect challenges to demonstrate asthma or exercise-induced bronchoconstriction in athletes. J Allergy Clin Immunol. 2008;122(2):238–46, quiz 247–8. doi:.https://doi.org/10.1016/j.jaci.2008.06.014
12 Miedinger D , Chhajed PN , Tamm M , Stolz D , Surber C , Leuppi JD . Diagnostic tests for asthma in firefighters. Chest. 2007;131(6):1760–7. doi:.https://doi.org/10.1378/chest.06-2218
13 Brannan JD , Anderson SD , Perry CP , Freed-Martens R , Lassig AR , Charlton B ; Aridol Study Group. The safety and efficacy of inhaled dry powder mannitol as a bronchial provocation test for airway hyperresponsiveness: a phase 3 comparison study with hypertonic (4.5%) saline. Respir Res. 2005;6(1):144. doi:.https://doi.org/10.1186/1465-9921-6-144
14 Whiting PF , Rutjes AWS , Westwood ME , Mallett S , Deeks JJ , Reitsma JB , et al.; QUADAS-2 Group. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;155(8):529–36. doi:.https://doi.org/10.7326/0003-4819-155-8-201110180-00009
15 Anderson SD , Brannan J , Spring J , Spalding N , Rodwell LT , Chan K , et al. A new method for bronchial-provocation testing in asthmatic subjects using a dry powder of mannitol. Am J Respir Crit Care Med. 1997;156(3):758–65. doi:.https://doi.org/10.1164/ajrccm.156.3.9701113
16 Lund TK , Pedersen L , Anderson SD , Sverrild A , Backer V . Are asthma-like symptoms in elite athletes associated with classical features of asthma? Br J Sports Med. 2009;43(14):1131–5. doi:.https://doi.org/10.1136/bjsm.2008.054924
17 Romberg K , Tufvesson E , Bjermer L . Extended diagnostic criteria used for indirect challenge testing in elite asthmatic swimmers. Respir Med. 2012;106(1):15–24. doi:.https://doi.org/10.1016/j.rmed.2011.09.011
18 Ulrik CS , Svenningsen C . High prevalence of asthma in Danish elite canoe- and kayak athletes. Dan Med J. 2012;59(4):A4405.
19 Barben J , Kuehni CE , Strippoli MP , Schiller B , Hammer J , Trachsel D ; Swiss Paediatric Respiratory Research Group. Mannitol dry powder challenge in comparison with exercise testing in children. Pediatr Pulmonol. 2011;46(9):842–8. doi:.https://doi.org/10.1002/ppul.21453
20 Toennesen LL , Porsbjerg C , Pedersen L , Backer V . Predictors of airway hyperresponsiveness in elite athletes. Med Sci Sports Exerc. 2015;47(5):914–20. doi:.https://doi.org/10.1249/MSS.0000000000000496
21 Backer V , Sverrild A , Porsbjerg C . FENO and AHR mannitol in patients referred to an out-of-hospital asthma clinic: a real-life study. J Asthma. 2014;51(4):411–6. doi:.https://doi.org/10.3109/02770903.2013.878953
22 Clearie KL , Williamson PA , Vaidyanathan S , Short P , Goudie A , Burns P , et al. Disconnect between standardized field-based testing and mannitol challenge in Scottish elite swimmers. Clin Exp Allergy. 2010;40(5):731–7. doi:.https://doi.org/10.1111/j.1365-2222.2010.03461.x
23 Holzer K , Anderson SD , Chan HK , Douglass J . Mannitol as a challenge test to identify exercise-induced bronchoconstriction in elite athletes. Am J Respir Crit Care Med. 2003;167(4):534–7. doi:.https://doi.org/10.1164/rccm.200208-916OC
24 Andregnette-Roscigno V , Fernández-Nieto M , Arochena L , García Del Potro M , Aguado E , Sastre J . Methacholine is more sensitive than mannitol for evaluation of bronchial hyper-responsiveness in youth athletes with exercise-induced bronchoconstriction. Pediatr Allergy Immunol. 2012;23(5):501–3. doi:.https://doi.org/10.1111/j.1399-3038.2012.01293.x
25 Koskela HO , Hyvärinen L , Brannan JD , Chan HK , Anderson SD . Responsiveness to three bronchial provocation tests in patients with asthma. Chest. 2003;124(6):2171–7. doi:.https://doi.org/10.1378/chest.124.6.2171
26 Subbarao P , Brannan JD , Ho B , Anderson SD , Chan HK , Coates AL . Inhaled mannitol identifies methacholine-responsive children with active asthma. Pediatr Pulmonol. 2000;29(4):291–8. doi:.https://doi.org/10.1002/(SICI)1099-0496(200004)29:4<291::AID-PPUL9>3.0.CO;2-A
27 Kim MH , Song WJ , Kim TW , Jin HJ , Sin YS , Ye YM , et al. Diagnostic properties of the methacholine and mannitol bronchial challenge tests: a comparison study. Respirology. 2014;19(6):852–6. doi:.https://doi.org/10.1111/resp.12334
28 Aronsson D , Tufvesson E , Bjermer L . Comparison of central and peripheral airway involvement before and during methacholine, mannitol and eucapnic hyperventilation challenges in mild asthmatics. Clin Respir J. 2011;5(1):10–8. doi:.https://doi.org/10.1111/j.1752-699X.2009.00183.x
29 McClean MA , Htun C , King GG , Berend N , Salome CM . Cut-points for response to mannitol challenges using the forced oscillation technique. Respir Med. 2011;105(4):533–40. doi:.https://doi.org/10.1016/j.rmed.2010.10.013
30 Stenfors N . Self-reported symptoms and bronchial hyperresponsiveness in elite cross-country skiers. Respir Med. 2010;104(11):1760–3. doi:.https://doi.org/10.1016/j.rmed.2010.07.014
31 de Menezes MB , Ferraz E , Brannan JD , Martinez EZ , Vianna EO . The efficacy and safety of mannitol challenge in a workplace setting for assessing asthma prevalence. J Asthma. 2018;55(12):1278–85. doi:.https://doi.org/10.1080/02770903.2017.1418887
32 White EC , de Klerk N , Hantos Z , Priston M , Hollams EM , James A , et al. Mannitol challenge testing for asthma in a community cohort of young adults. Respirology. 2017;22(4):678–83. doi:.https://doi.org/10.1111/resp.12955
33 Cancelliere N , Bobolea I , López-Carrasco V , Barranco P , López-Serrano C , Quirce S . Comparative study of bronchial hyperresponsiveness to methacholine and mannitol in the initial diagnosis of asthma. J Investig Allergol Clin Immunol. 2013;23(5):361–2.
34 Osthoff M , Michel F , Strupler M , Miedinger D , Taegtmeyer AB , Leuppi JD , et al. Bronchial hyperresponsiveness testing in athletes of the Swiss Paralympic team. BMC Sports Sci Med Rehabil. 2013;5(1):7. doi:.https://doi.org/10.1186/2052-1847-5-7
35 Vakali S , Vogiatzis I , Florou A , Giavi S , Zakynthinos S , Papadopoulos NG , et al. Exercice-induced bronchoconstriction among athletes: Assessment of bronchial provocation tests. Respir Physiol Neurobiol. 2017;235:34–9. doi:.https://doi.org/10.1016/j.resp.2016.09.010
36 Porpodis K , Domvri K , Kontakiotis T , Fouka E , Kontakioti E , Zarogoulidis K , et al. Comparison of diagnostic validity of mannitol and methacholine challenges and relationship to clinical status and airway inflammation in steroid-naïve asthmatic patients. J Asthma. 2017;54(5):520–9. doi:.https://doi.org/10.1080/02770903.2016.1238926
37 Rutjes AWS , Reitsma JB , Vandenbroucke JP , Glas AS , Bossuyt PMM . Case-control and two-gate designs in diagnostic accuracy studies. Clin Chem. 2005;51(8):1335–41. doi:.https://doi.org/10.1373/clinchem.2005.048595
38 Leuppi JD . Bronchoprovocation tests in asthma: direct versus indirect challenges. Curr Opin Pulm Med. 2014;20(1):31–6. doi:.https://doi.org/10.1097/MCP.0000000000000009
39 Anderson SD , Brannan JD . Bronchial provocation testing: the future. Curr Opin Allergy Clin Immunol. 2011;11(1):46–52. doi:.https://doi.org/10.1097/ACI.0b013e3283423183
40 Brannan JD , Perry CP , Anderson SD . Mannitol test results in asthmatic adults receiving inhaled corticosteroids. J Allergy Clin Immunol. 2013;131(3):906–7. doi:.https://doi.org/10.1016/j.jaci.2012.08.028
41 Katial RK , Covar RA . Bronchoprovocation testing in asthma. Immunol Allergy Clin North Am. 2012;32(3):413–31. doi:.https://doi.org/10.1016/j.iac.2012.06.002
42 Stolz D , Anderson SD , Gysin C , Miedinger D , Surber C , Tamm M , et al. Airway reactivity to inhaled mannitol in cigarette smokers: a longitudinal study. Respir Med. 2007;101(7):1470–6. doi:.https://doi.org/10.1016/j.rmed.2007.01.008
43 Sverrild A , Bergqvist A , Baines KJ , Porsbjerg C , Andersson CK , Thomsen SF , et al. Airway responsiveness to mannitol in asthma is associated with chymase-positive mast cells and eosinophilic airway inflammation. Clin Exp Allergy. 2016;46(2):288–97. doi:.https://doi.org/10.1111/cea.12609
44 Leuppi JD , Salome CM , Jenkins CR , Koskela H , Brannan JD , Anderson SD , et al. Markers of airway inflammation and airway hyperresponsiveness in patients with well-controlled asthma. Eur Respir J. 2001;18(3):444–50. doi:.https://doi.org/10.1183/09031936.01.00058601
45 Anderson WJ , Lipworth BJ . Relationship of mannitol challenge to methacholine challenge and inflammatory markers in persistent asthmatics receiving inhaled corticosteroids. Hai. 2012;190(5):513–21. doi:.https://doi.org/10.1007/s00408-012-9396-6
46 Wood LG , Powell H , Gibson PG . Mannitol challenge for assessment of airway responsiveness, airway inflammation and inflammatory phenotype in asthma. Clin Exp Allergy. 2010;40(2):232–41. doi:.https://doi.org/10.1111/j.1365-2222.2009.03371.x
47 Stickland MK , Rowe BH , Spooner CH , Vandermeer B , Dryden DM . Accuracy of eucapnic hyperpnea or mannitol to diagnose exercise-induced bronchoconstriction: a systematic review. Ann Allergy Asthma Immunol. 2011;107(3):229–34.e8. doi:.https://doi.org/10.1016/j.anai.2011.06.013
48 Anderson SD . Indirect challenge tests: Airway hyperresponsiveness in asthma: its measurement and clinical significance. Chest. 2010;138(2, Suppl):25S–30S. doi:.https://doi.org/10.1378/chest.10-0116
49 Parkerson J , Ledford D . Mannitol as an indirect bronchoprovocation test for the 21st century. Ann Allergy Asthma Immunol. 2011;106(2):91–6. doi:.https://doi.org/10.1016/j.anai.2010.11.010
Both authors contributed equally to the manuscript.
PK and EHSK contributed equally to the manuscript.
This study was supported by the Allergiestiftung Ulrich Müller-Gierok.
Prior to this study, David Miedinger and Jörg Leuppi received free mannitol bronchial provocation test kits from Pharmaxis Ltd. to perform clinical studies investigating patients with asthma/COPD or individuals in the workforce.