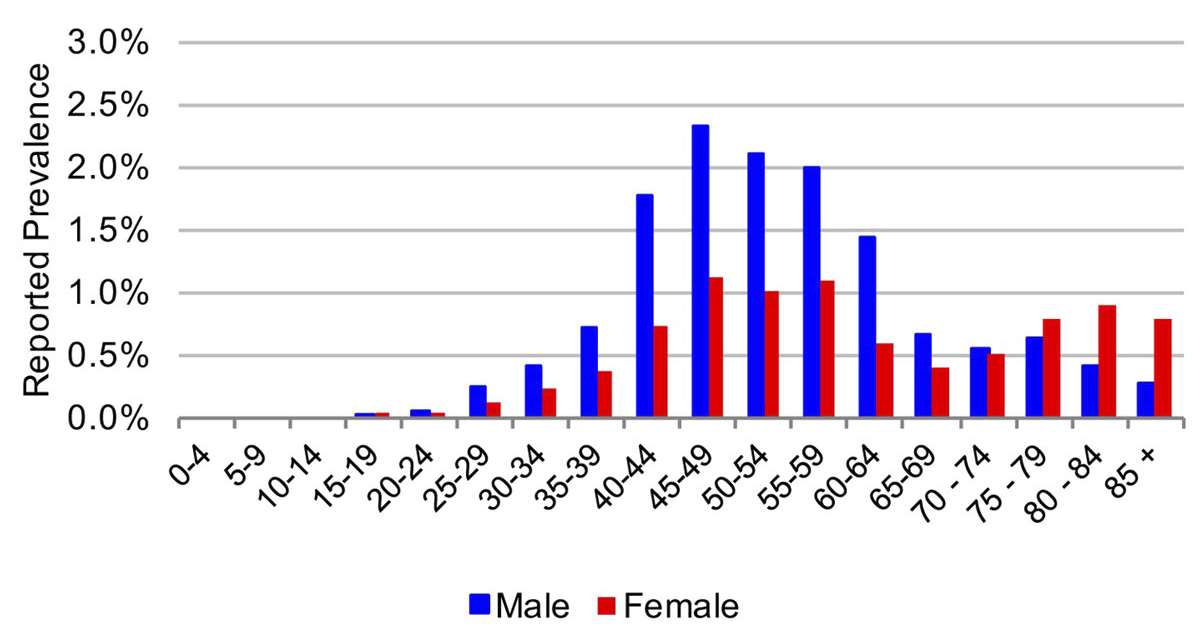
Figure 1 Age and sex distribution of hepatitis C cases in the canton of St Gallen in 2015.
DOI: https://doi.org/10.4414/smw.2019.14694
In 2016, the World Health Organization (WHO) identified viral hepatitis as a pandemic becuase of its associated substantial public health and economic burdens [1]. Affecting communities and health systems across the world, viral hepatitis was responsible for more deaths than the human immunodeficiency virus (HIV) and tuberculosis in the previous year [1]. Hepatitis C virus (HCV) infection is a leading cause of liver cirrhosis, decompensated cirrhosis and hepatocellular carcinoma (HCC), often requiring expensive therapies and procedures such as liver transplants, or resulting in premature death. Chronic hepatitis C (CHC) affects more than 71 million people worldwide and was responsible for approximately 400,000 deaths in 2015 [1, 2]. Though HCV inflicts undue health and economic damage on countries, the opportunity for public health strategies to tackle the disease burden has been revived. Given the development of newer, more efficacious treatment regimens such as direct-acting antivirals (DAAs), strategies can now be created that address the elimination, rather than the management, of HCV globally.
In high-income countries such as Switzerland, with a relatively low prevalence and expanded access to DAA therapy, to cure and eliminate HCV is possible. Given this situation, a multi-stakeholder network launched the Swiss Hepatitis Strategy [3], with the goal of reducing morbidity and mortality caused by viral hepatitis C and eliminate it by 2030. The strategy outlined a reduction of the number of new infections, of chronic HCV infections, and of liver transplants and HCC cases caused by HCV. Though a recent situational analysis estimated an overall anti-HCV prevalence of 0.7% and, after adjustment, reported 36,000–43,000 viraemic cases (estimated prevalence 0.49%) in 2015 [4] (roughly three times that of HIV [4]), only a minority of patients has been cured thus far. Since 1995, approximately 4000 people have died in connection with HCV in Switzerland, about five times as many deaths as from HIV or hepatitis B virus (HBV) [4].
Previous modelling work has evaluated the forecasted impact of increased access to more efficacious therapies [5–7]. Considering more than 40 countries throughout Europe and Asia, these studies showed that, although significant reductions in disease burden are plausible, strategies to improve diagnosis and treatment capacity are needed in order to do so [5–7]. More recent modelling studies have quantified the estimated diagnostic and treatment expansion necessary to achieve the Global Health Sector Strategy goals for elimination of viral hepatitis by 2030 [8, 9]. These studies highlighted impediments on the pathway to care and analysed the trade-offs between segments of the patient population to be targeted – considerations countries must account for in order to achieve elimination. Particularly, although increased treatment coverage and sustained virological response (SVR) in later stage liver disease have the greatest short-term impact on reducing morbidity and mortality, treatment of early stage fibrosis (F0–F1) is necessary to achieve reductions in total viraemic infections and prevent ongoing transmission [9]. These reports are helpful in the initial planning and development of national HCV policies, but modelling of elimination strategies amongst specific populations or sub-regions is limited. Development of possible microelimination scenarios breaking down national elimination goals into individual population segments [10] enables policy makers to understand current disease landscapes on a hospital-based or regional level.
In late 2017, Switzerland took a first step towards elimination by lifting treatment restrictions to allow ≥F0 patients to receive treatment. Alongside this change was an increase in the number of patients starting treatment, which exceeded the 7% threshold that puts Switzerland on track for elimination [11]. However, high treatment levels can be difficult to sustain, especially if there is an insufficient pool of patients diagnosed and linked to care. Thus, there is a growing need to identify HCV management strategies in the cantons throughout Switzerland.
This study aimed to identify scenarios that could achieve the Swiss Hepatitis Strategy by 2030 at the regional level. In this analysis, we modelled the current epidemiological landscape of HCV and identified the increases in diagnosis and treatment ratesthat might be necessary to achieve the strategy goals in eastern (region of St Gallen), western (region of Geneva) and northern (region of Zurich) Switzerland.
Three regions in Switzerland were included in this analysis: St Gallen, a region in eastern Switzerland containing the cantons of Appenzell Innerrhoden, Appenzell Ausserrhoden and St Gallen. It has approximately 570,000 inhabitants (at the end of 2016) [12], representing almost 10% of the Swiss population. Geneva, a region in southern Switzerland including the canton of Geneva, is the second most populous city in the country, with a resident population (at the end of 2016) of approximately 489,500 [12]. Zurich, a region in northern Switzerland including the canton of Zurich, is the largest city in the country, with a population of 1.5 million inhabitants [12].
An Excel-based Markov disease burden model was developed to forecast the current and future prevalence of HCV infection stratified by fibrosis stage, and of associated liver-related morbidity and mortality. A comprehensive description of this model has been published previously [2, 13–15]. A disease progression model was constructed to estimate the HCV-infected population by liver disease stage, from 1950 to 2030. The model estimates the annual number of acute infections that progress to chronic HCV infection, after accounting for spontaneous clearance of the virus. The progression of these new cases by age and sex was followed, with adjustments for mortality and cure. Three models were populated with published data or expert feedback and calibrated with canton-specific assumptions to forecast the future burden of HCV by sequelae through to 2030. The three models were based on canton-specific data and outcome values were fitted to these historical parameters. A full description of the model can be found in appendices 1 and 2 .
The St Gallen model was seeded with data gathered from the Cantonal Hospital of St Gallen (KSSG). Electronic medical records from 1 January 2004 and 31 December 2016 of all patients with a positive anti-HCV screening test (acute or chronic HCV infection) presenting at the hospital were analysed retrospectively. Patient characteristics such as age, year of hepatitis C diagnosis, diagnosis of acute hepatitis C, stage of liver fibrosis (liver elastography and/or liver biopsies were used to define the individual fibrosis score according to METAVIR [16]), treatment regimen and outcome (SVR12), co-infections (HIV, HBV, hepatitis A virus [HAV]), HCC and liver transplantation due to HCV were assessed. This information was used as input data (treatment numbers, treatment outcomes, genotype distribution, age distribution, HCC and liver transplantation due to HCV, actual stage of liver fibrosis) for the St Gallen model (table 1). The study was approved by the ethical committee in eastern Switzerland, with the project-ID 2017-00693.
Table 1 Inputs by cantonal model.
| St Gallen | Source | Geneva | Source | Zurich | Source | |
|---|---|---|---|---|---|---|
| Viraemic prevalence rate (2015) | 0.49% (0.45–0.55%) |
[4] | 0.65% (0.59–0.71%) |
Calculated | 0.73% (0.68–0.81%) |
Calculated |
| Total diagnosed viraemic | 2,025 (2013*) |
[17] | 2095 (2015*) |
[17] | 6470 (2015*) |
[17] |
| Annual newly diagnosed | 85 (2013*) |
[17] | 96 (2015*) |
[17] | 184 (2015*) |
[17] |
| Annual treated (2016) | 113 | [18] | 191 | [18] | 515 | [18] |
| HCV liver transplants | 1 (2015*) |
KSSG | 6 (2016*) |
University Hospital, Geneva | 3 (2016*) |
University Hospital, Zurich |
HCV = hepatitis C virus; KSSG = Kantonsspital St. Gallen / Cantonal Hospital St Gallen * Denotes year of estimate
Genotype distribution and liver transplant data from the University Hospital of Geneva were used. Liver cancer data by histology, including HCC, were collected from 1990 to 2013 from the Geneva Liver Tumour Registry (table 2). Ethical approval was not necessary in Geneva as only aggregated totals were used.
Table 2 Annual number of reported liver cancer cases.
| Year | Cases in Geneva | Cases in Zurich |
|---|---|---|
| 1988 | 73 | |
| 1989 | 61 | |
| 1990 | 28 | 73 |
| 1991 | 36 | 67 |
| 1992 | 39 | 74 |
| 1993 | 36 | 62 |
| 1994 | 25 | 66 |
| 1995 | 35 | 73 |
| 1996 | 38 | 85 |
| 1997 | 43 | 74 |
| 1998 | 36 | 81 |
| 1999 | 40 | 77 |
| 2000 | 35 | 82 |
| 2001 | 49 | 94 |
| 2002 | 47 | 97 |
| 2003 | 64 | 85 |
| 2004 | 46 | 98 |
| 2005 | 62 | 85 |
| 2006 | 40 | 79 |
| 2007 | 47 | 101 |
| 2008 | 46 | 86 |
| 2009 | 55 | 99 |
| 2010 | 47 | 148 |
| 2011 | 49 | 109 |
| 2012 | 62 | 132 |
| 2013 | 69 | 121 |
| 2014 | 132 |
The genotype distribution was based on Switzerland cohort data, which gave an estimate that approximately 29% of cases had genotype 3 and 26% genotypes 1a and 1b [19, 20]. Liver transplant data from the University Hospital Zurich were used. Zurich liver cancer data from 1988–2014 was collected (table 2). Ethical approval was not necessary in Zurich as only aggregated totals were used.
Regional HCV prevalence by canton was not available and had to be estimated from available data – national HCV prevalence, reported liver cancer cases by canton, and reported HCV diagnoses (after adjusting for treatment and mortality) by canton [4, 17]. Expert consensus was used to check for consistency of the prevalence estimates across all three regions. Attempts to use the reported numbers of all and newly diagnosed cases by canton to estimate regional prevalence was not successful as they were influenced by the number of centres treating HCV and the level of testing in each region.
The anti-HCV prevalence in St Gallen was estimated to be the same as the published viraemic HCV prevalence in Switzerland: 0.49% (95% confidence interval [CI] 0.45–0.54) [4]. Geneva and Zurich have more diverse immigrant populations, which made extrapolation from national data more difficult. The reported liver cancer cases in each region were used to back-calculate the total number of HCV infections required to result in the reported cancer cases. The prevalence estimates generated (table 1) were higher than the national prevalence, as expected.
The total number of HCV infections was estimated by multiplying the regional prevalence and the population of the cantons in each region. For St Gallen, the 2015 population of the cantons Appenzell Innerrhoden, Appenzell Ausserrhoden and St Gallen was used [17]. Notification data, provided by the Swiss Federal Office of Public Health, was used to form the age and gender distribution of the model, yearly number of new HCV infections and diagnosed cases.
The total number of HCV diagnosed cases in the cantons Appenzell Innerrhoden, Appenzell Ausserrhoden and St Gallen, Geneva and Zurich (1990–2016) and the yearly number of new HCV infections were based on the cases reported to the Swiss Federal Office of Public Health [17], after accounting for mortality and cure. The reported chronic cases were stratified into 1-year age cohorts and were aged forward each year, taking into account reported background mortality in the individual age cohorts, as well as reported treatment and cure rates based on rates of SVR.
The annual number of treated patients in St Gallen was collected in the study described above. All specialised care of HCV patients in the cantons of St Gallen and Appenzell is centralised in the cantonal hospital. The annual number of treated patients for Geneva and Zurich was scaled from national estimates using IMS Health data (2004–2015) [18]. Geneva and Zurich account for 10% and 30% of total Swiss HCV infections, respectively. It was assumed that they accounted for the same proportion of the HCV treated population. Since all patients, regardless of fibrosis score, were eligible for treatment from 2018, and Geneva and Zurich are the two largest cantons in Switzerland, it was believed that a larger proportion of patients linked to care would be captured in these regions.
Two scenarios were developed to evaluate the disease burden in St Gallen, Geneva and Zurich: Base 2016 and Swiss Hepatitis Strategy. The inputs by scenario can be found in tables 3a and 3b .
Table 3a Inputs by scenario for St Gallen, Geneva, and Zurich 2015–2030: Base 2016.
| 2015 | 2016 | 2017 | 2018-2030 | |
|---|---|---|---|---|
| St Gallen | ||||
| Treated | 107 | 113 | 113 | 113 |
| Newly diagnosed | 85 | 85 | 85 | 85 |
| Fibrosis stage | ≥F2 | ≥F2 | ≥F2 | ≥F0 |
| Treated age | 20–85+ | 20–85+ | 20–85+ | 20–85+ |
| SVR | 96% | 96% | 96% | 96% |
| Geneva | ||||
| Treated | 232 | 191 | 191 | 191 |
| Newly diagnosed | 96 | 96 | 96 | 96 |
| Fibrosis stage | ≥F2 | ≥F2 | ≥F2 | ≥F0 |
| Treated age | 20–85+ | 20–85+ | 20–85+ | 20–85+ |
| SVR* | 97% | 97% | 97% | 97% |
| Zurich | ||||
| Treated | 626 | 515 | 515 | 515 |
| Newly diagnosed | 184 | 184 | 184 | 184 |
| Fibrosis stage | ≥F2 | ≥F2 | ≥F2 | ≥F0 |
| Treated age | 20–85+ | 20–85+ | 20–85+ | 20–85+ |
| SVR* | 97% | 97% | 97% | 97% |
| SVR = sustained viral response * Although SVR by genotype was the same across all regions, the average SVR was different because of the different genotype distribution in the three cantons. |
||||
Table 3b Inputs by scenario for St Gallen, Geneva, and Zurich 2015–2030: Swiss Hepatitis Strategy*.
| 2018 | 2019 | 2020 | 2022 | 2025 | |
|---|---|---|---|---|---|
| St Gallen | |||||
| Treated | 250 | 350 | 430 | 160 | 150 |
| Newly diagnosed | 100 | 100 | 130 | 130 | 130 |
| Fibrosis stage | ≥F0 | ≥F0 | ≥F0 | ≥F0 | ≥F0 |
| Treated age | 20–85+ | 20–85+ | 20–85+ | 20–85+ | 20–85+ |
| SVR | 97% | 97% | 97% | 97% | 97% |
| Geneva | |||||
| Treated | 230 | 240 | 240 | 240 | 240 |
| Newly diagnosed | 140 | 140 | 140 | 140 | 140 |
| Fibrosis stage | ≥F0 | ≥F0 | ≥F0 | ≥F0 | ≥F0 |
| Treated age | 20–85+ | 20–85+ | 20–85+ | 20–85+ | 20–85+ |
| SVR | 97% | 97% | 97% | 97% | 97% |
| Zurich | |||||
| Treated | 750 | 850 | 850 | 800 | 800 |
| Newly diagnosed | 200 | 250 | 300 | 350 | 350 |
| Fibrosis stage | ≥F0 | ≥F0 | ≥F0 | ≥F0 | ≥F0 |
| Treated age | 20–85+ | 20–85+ | 20–85+ | 20–85+ | 20–85+ |
| SVR | 97% | 97% | 97% | 97% | 97% |
SVR = sustained viral response * The inputs are the same as the base 2016 scenario for years 2015-2017. Numbers are rounded values.
The Base 2016 scenario assumes that the current standard of care will continue in the individual cantons up to 2030. For St Gallen, treatment was restricted to those with liver fibrosis grade ≥F2 and aged 18 to 84 years to represent the treatment restrictions across Switzerland up to the end of 2017. This was expanded to ≥F0 in 2018 to represent the updated treatment guidelines in Switzerland, which expanded eligibility to all patients regardless of fibrosis stage at the end of 2017. SVR was set to an average of 96% to represent patients receiving DAAs. Under this scenario, approximately 110 patients are treated annually and 90 patients newly diagnosed each year up to 2030.
Similar fibrosis and age restrictions and SVR rates were applied to the Base 2016 scenario for Geneva and Zurich. In 2016, those ≥F2 and aged over 18 years were eligible for treatment; however, beginning in 2018, this was relaxed to ≥F0. The SVR rates by genotype that were applied in the St Gallen model were also incorporated into the models for Geneva and Zurich. An estimated 190 patients and 520 patients were treated in Geneva and Zurich, respectively, in 2016. This number was expected to stay constant over time; 140 and 180 patients were diagnosed each year in Geneva and Zurich, respectively, up to 2030.
Under the Swiss Hepatitis Strategy, diagnosis and treatment was increased in order to achieve a 30% reduction in new infections, total viraemic infections, liver transplants and HCC cases by 2020 and a further 90% reduction by 2030. The outcomes by scenario were then compared.
In 2015, the estimated viraemic prevalence in St Gallen was 0.5% (0.5–0.6%), corresponding to 2800 (2600–3,100) chronic HCV cases. In Geneva and Zurich, the estimated prevalence was slightly higher, with an estimated 0.7% (0.6–0.7%) viraemic prevalence, or 3300 (3000–3600) cases in Geneva and 0.7% (0.7–0.8%) viraemic prevalence, or 10,800 (9900–11,900) infections in Zurich in the same year. Of these, approximately 70% of the viraemic population was found to be between the ages of 30 and 64. The distribution by age and sex can be seen in figures 1–6 . In St Gallen, the median age was 52 years, whereas in Geneva it was slightly older at 54 years. According to hospital-based data, in both cantons there were more male patients, who represented more than 60% of cases (65% in St Gallen vs 61% in Geneva). More than half of all patients had ever been treated in both St Gallen (52.2%) and Geneva (58.5%). Of these, 83% were cured in St Gallen, whereas in Geneva almost three-quarters of patients were cured. This hospital-specific data could not be provided for Zurich at the time of publication. The canton-specific cascade of care for 2017 is represented in figures 7–9 . The distribution of viraemic cases under the Base 2016 scenario is summarised in figures 10–12 .

Figure 1 Age and sex distribution of hepatitis C cases in the canton of St Gallen in 2015.
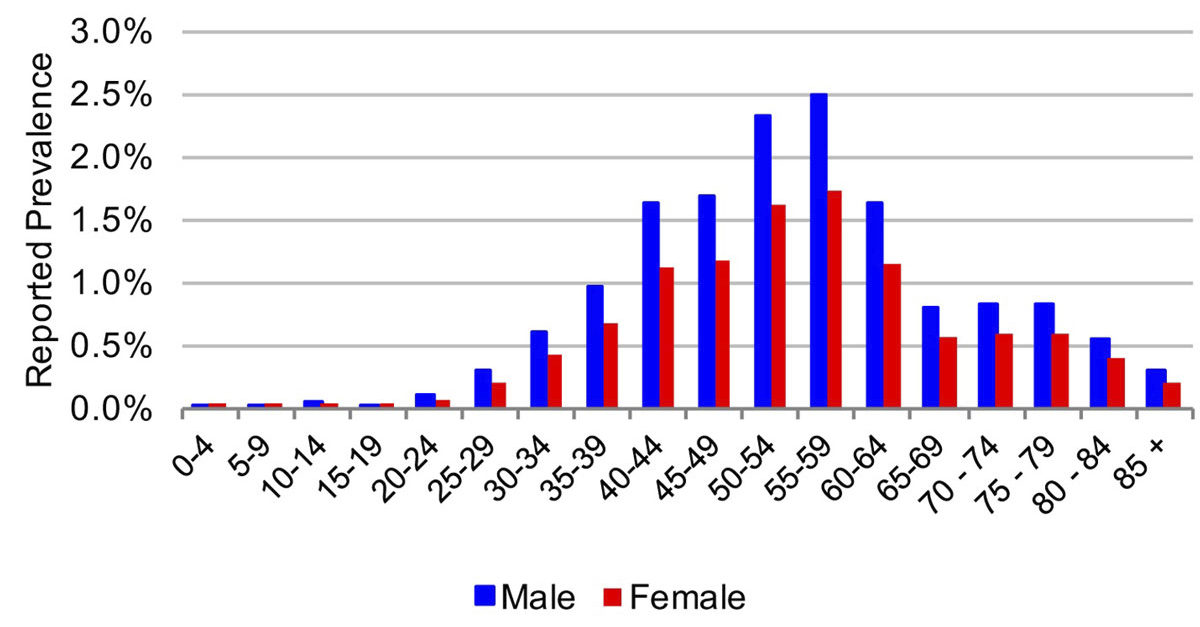
Figure 2 Age and sex distribution of hepatitis C cases in the canton of Geneva in 2015.
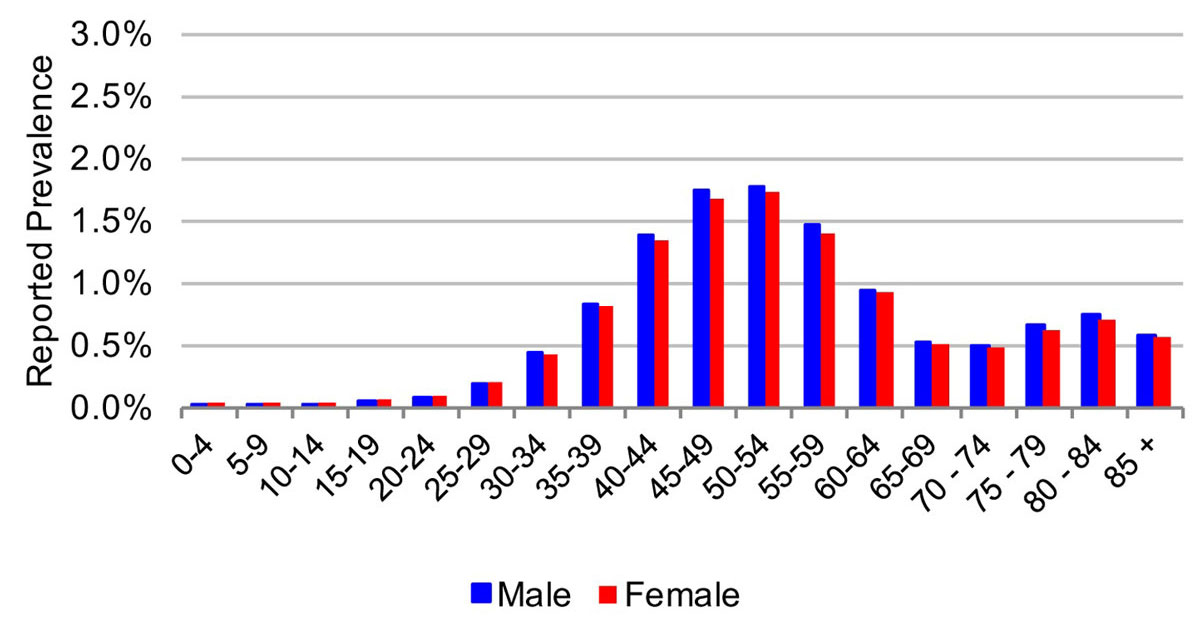
Figure 3 Age and sex distribution of hepatitis C cases in the cantons of St Gallen, Geneva and Zurich in 2015.
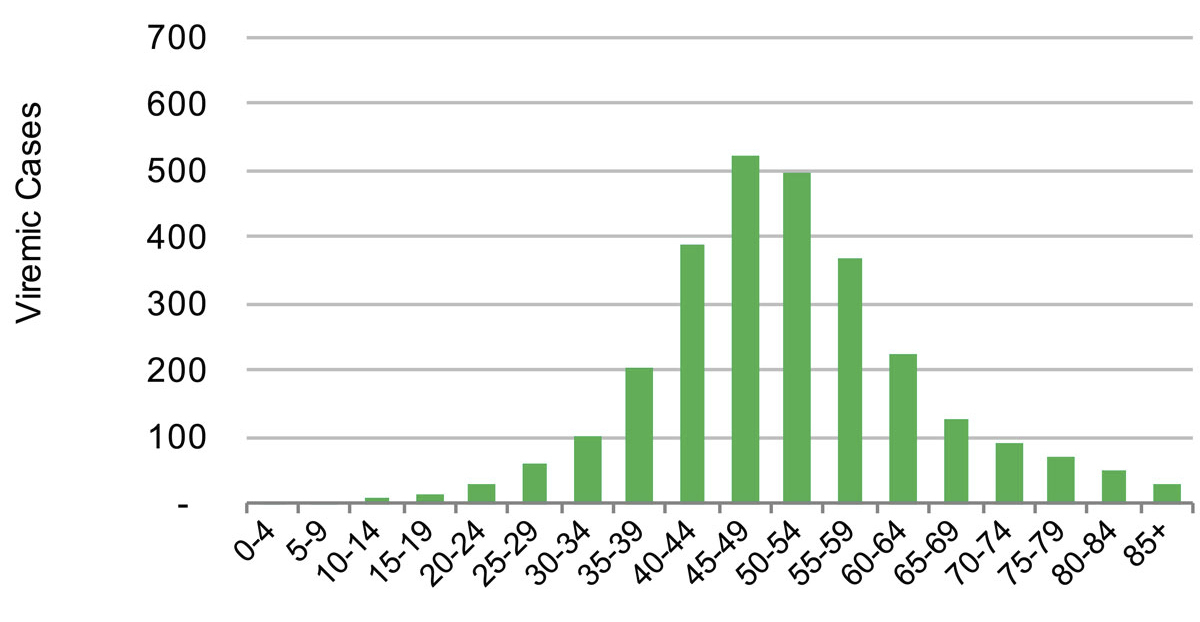
Figure 4 Distribution of hepatitis C cases by five year age cohort in the canton of St Gallen in 2015.
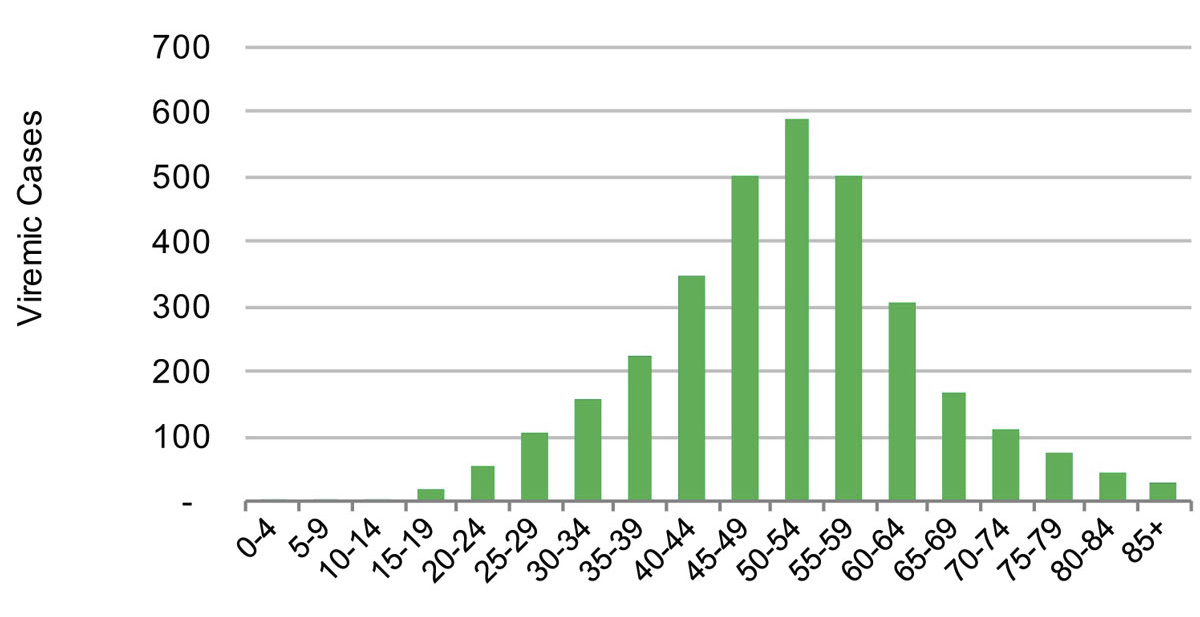
Figure 5 Distribution of hepatitis C cases by five year age cohort in the canton of Geneva in 2015.
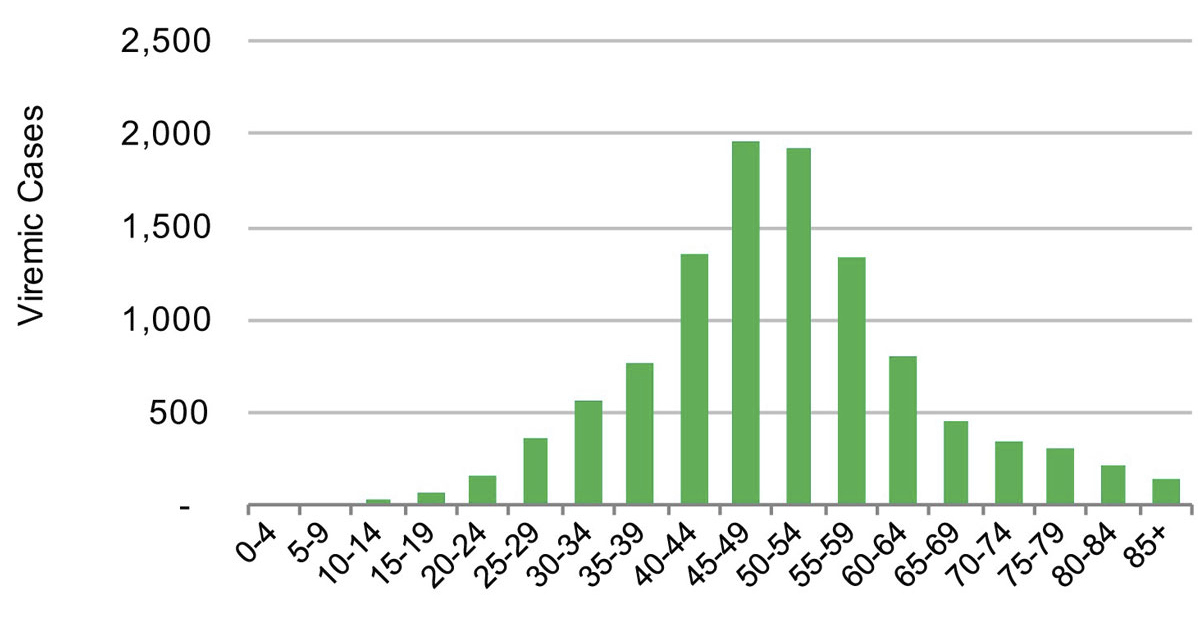
Figure 6 Distribution of hepatitis C cases by five year age cohort in the canton of Zurich in 2015.
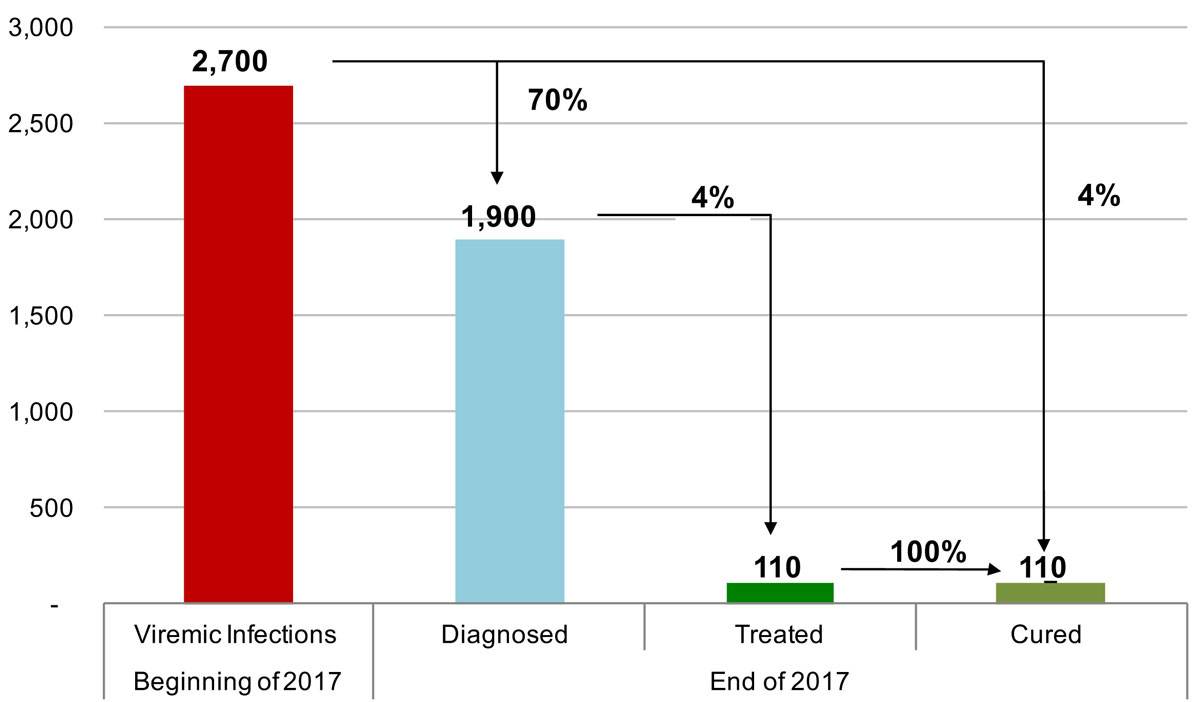
Figure 7 The hepatitis C cascade of care, including total viremic infections, diagnosed, treated and cured, in the canton of St Gallen for 2017.
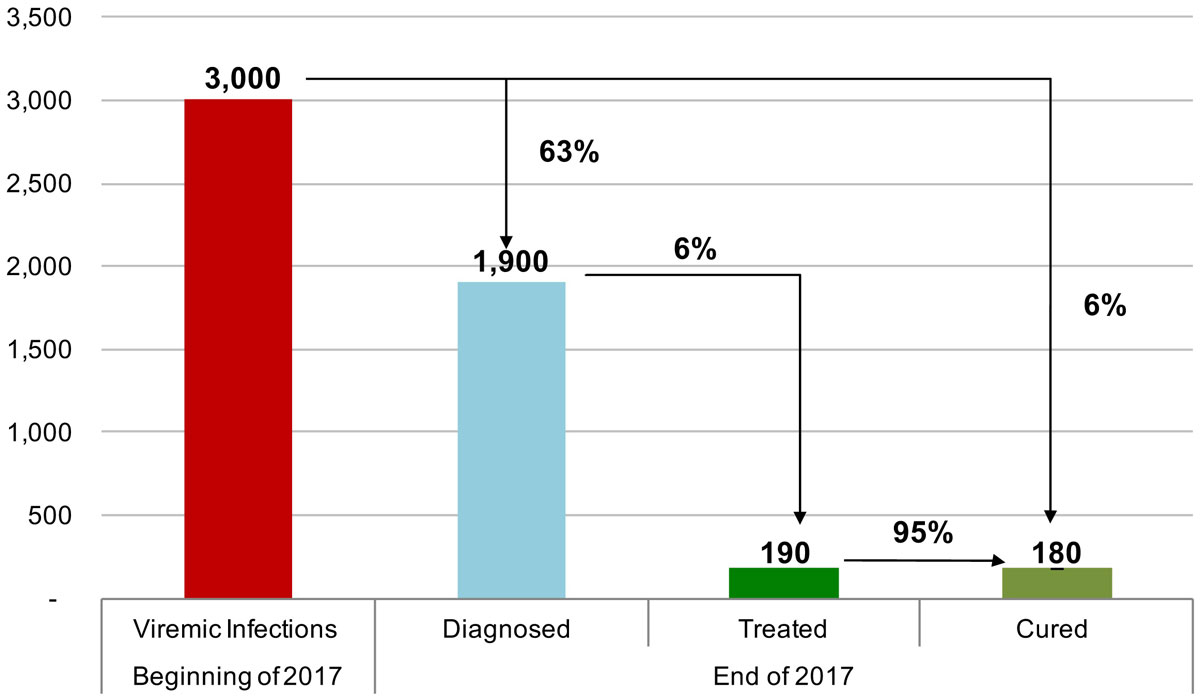
Figure 8 The hepatitis C cascade of care, including total viremic infections, diagnosed, treated and cured, in the canton of Geneva for 2017.
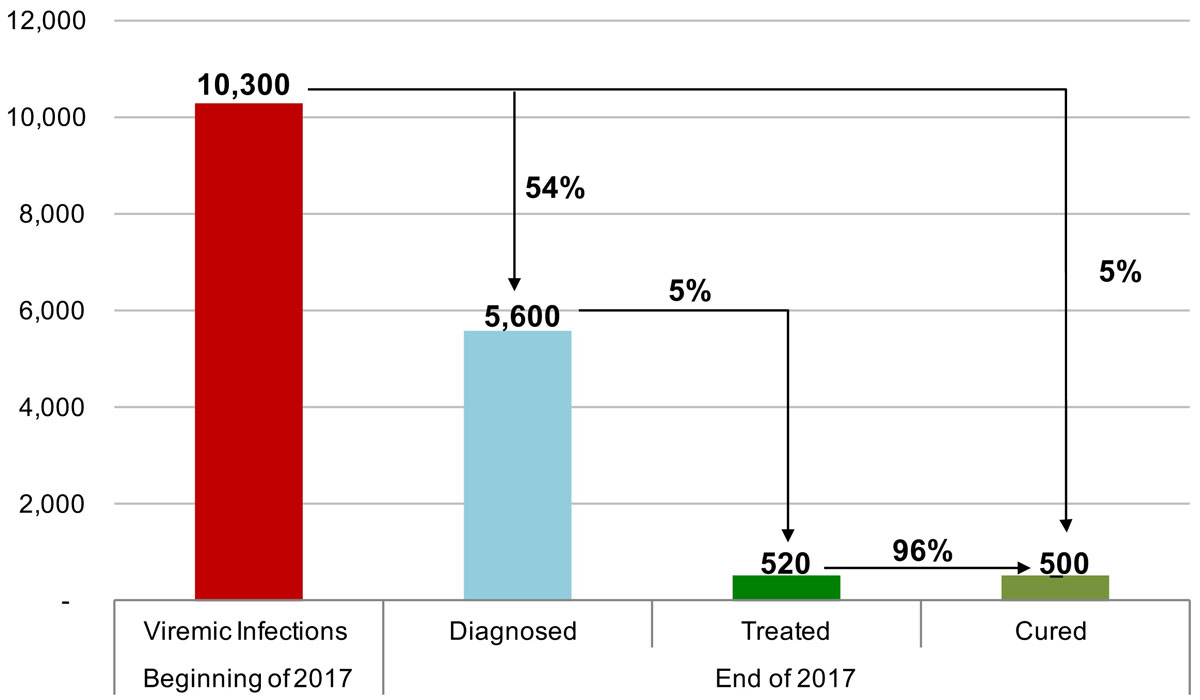
Figure 9 The hepatitis C cascade of care, including total viremic infections, diagnosed, treated and cured, in the canton of Zurich for 2017.
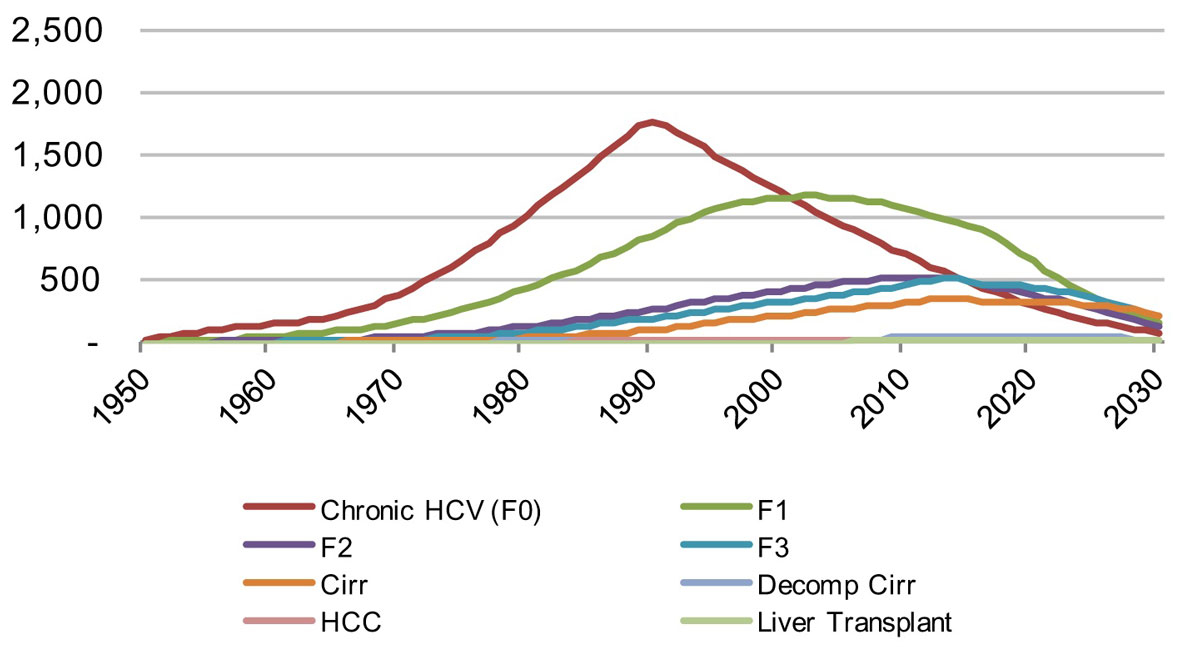
Figure 10 Distribution of viremic infections by disease stage in the canton of S. Gallen, from 1950–2030 under the Base 2016 scenario.
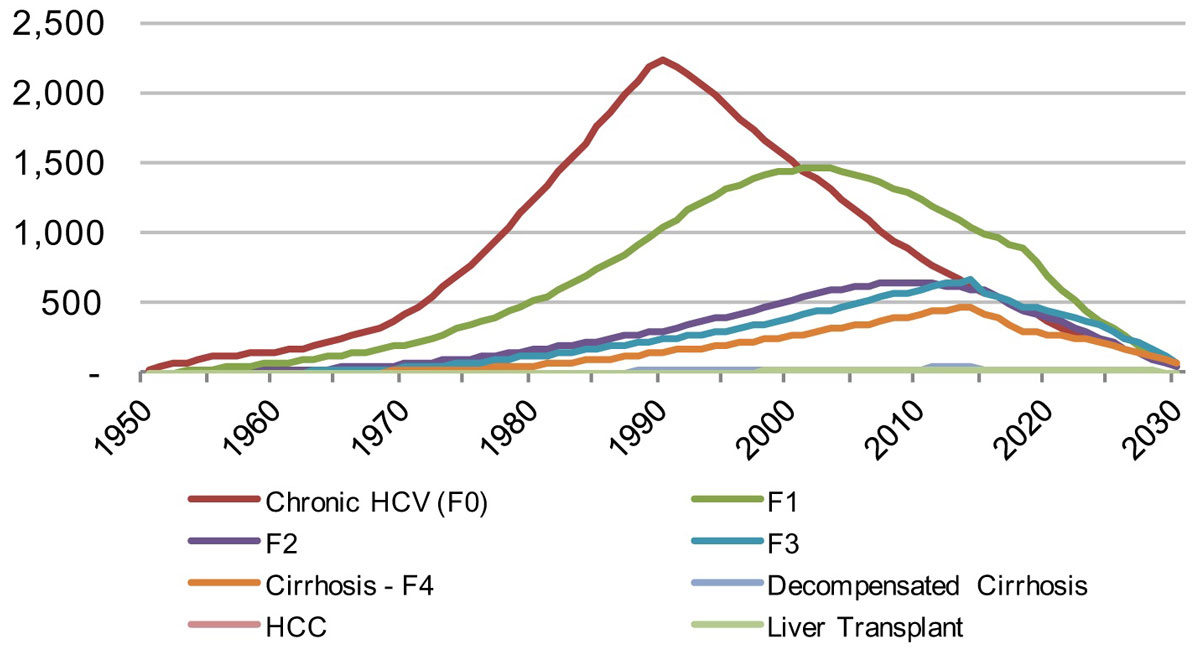
Figure 11 Distribution of viremic infections by disease stage in the canton of Geneva from 1950–2030 under the Base 2016 scenario.
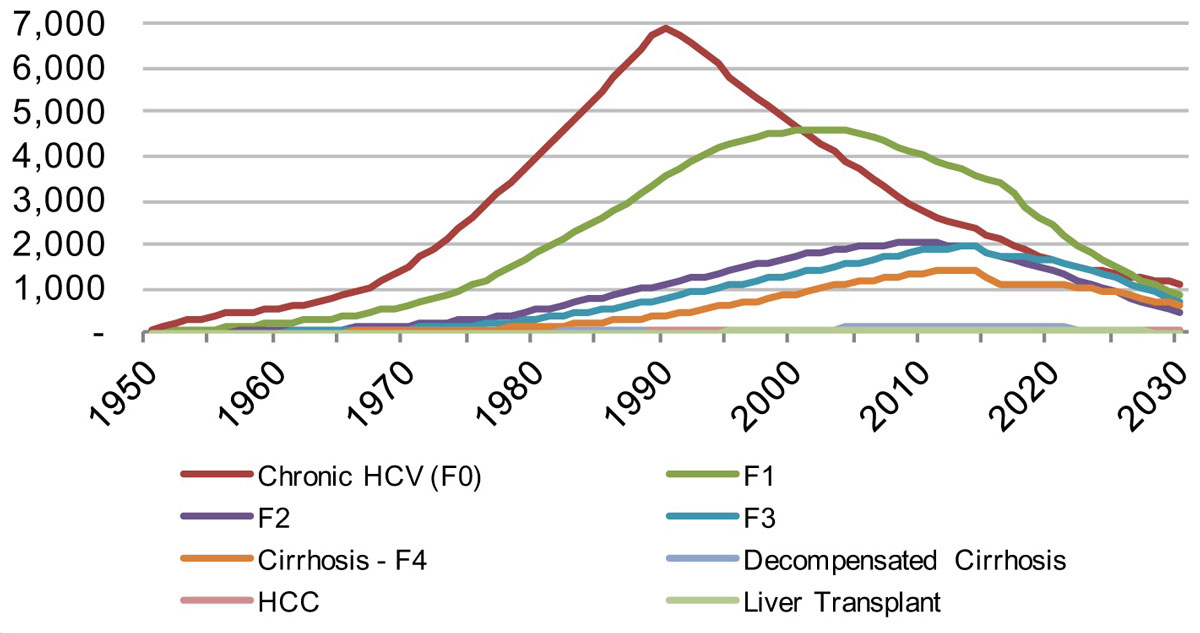
Figure 12 Distribution of viremic infections by disease stage in the canton of Zurich from 1950–2030 under the Base 2016 scenario.
The liver-related morbidity and mortality by scenario and canton can be seen in figures 13–15 . Given the number of patients treated with DAA therapy, decreases in liver-related morbidity and total viraemic infections are expected over the next 15 years. Under the Base 2016 scenario, the St Gallen model forecasts a reduction of liver-related death by about 10% in 2020 and by 35% in 2030, whereas liver cancer due to HCV would decline by more than 10% in 2020 and close to 40% in 2030. The model also estimates a reduction of chronic infections by 20% in 2020 and 70% in 2030. Decompensated cirrhosis cases are also forecasted to decline, by 10% in 2020 and by 35% by 2030. By retaining a rate of 95 newly diagnosed patients per year, 90% of all infected patients would be detected in 2030.
In Geneva, the model estimates a more rapid decline in liver-related morbidity and mortality. There would be a reduction of liver-related death by 35% in 2020 and 85% in 2030, whereas liver cancer due to HCV would be reduced by 30% in 2020 and 80% in 2030. There is also a forecasted decline of chronic infections by 30% in 2020 and approximately 90% in 2030. Decompensated cirrhosis cases are forecasted to decline by 45% by 2020 and 90% by 2030. By retaining a rate of 96 newly diagnosed patients per year, almost all infected patients would be detected in 2030.
In Zurich, an even greater impact is seen on the estimated HCV prevalence and associated liver-related morbidity and mortality. There would be a reduction of liver-related death by 15% in 2020 and 60% in 2030, whereas liver cancer due to HCV would be reduced by 15% in 2020 and 60% in 2030. There is also a forecasted decline of chronic infections of 20% in 2020 and 60% in 2030. Decompensated cirrhosis cases were estimated to drop by 20% by 2020 and 70% by 2030. By retaining a rate of 180 newly diagnosed patients per year, almost all infected patients would be detected in 2030.
In order to achieve these goals for St Gallen, fibrosis restrictions were relaxed to ≥F0 in 2018 in order to increase the pool of patients eligible for treatment. Treated age and SVR remained the same as in the Base 2016 scenario (18–84 years, 97%); however, the number of patients treated was increased to 350 in 2020 (in 2016 only 107 patients were treated at the Cantonal Hospital St Gallen). Furthermore, the number of new diagnoses increased from 90 patients annually to 130 from 2020. With these increases during the following 10 years, a modelled reduction of total chronicly infected patients by 40% in 2020 and 100% in 2030 can be achieved. More, the model shows a reduction in HCC due to HCV of 30% in 2020 and 95% in 2030; liver related deaths could be reduced by 30% in 2020 and 100% in 2030. Lastly, decompensated cirrhosis cases are forecasted to decline by 30% in 2020 and 100% in 2030. In total, approximately 2700 chronic hepatitis C patients have to be treated during 2016–2030 in eastern Switzerland to achieve the Swiss Hepatitis Strategy goals.
In Geneva, similar increases in treatment and newly diagnosed cases were necessary. The number of treated patients was increased to 2015 treatment levels (235 patients treated annually) in 2018 and maintained up to 2030. The number diagnosed will need to increase to 140 individuals annually from 2018. Treatment was also extended to those ≥F0 in 2018. SVR remained constant.
With 240 treated patients per year during the following 10 years, a modelled reduction of all chronicly infected patients by 35% in 2020 and 100% in 2030 can be achieved. The estimated number of HCC cases due to HCV is forecasted to decline by of 40% in 2020 and 100% in 2030, whereas decompensated cirrhosis cases are estimated to decrease by 60% by 2020 and 100% by 2030. Liver related deaths could be reduced by 45% in 2020 and 100% in 2030.
In Zurich, the number of patients that are needed to be treated will peak at 850 patients annually in 2019 and 2020. This then declines to 800 patients annually up to 2030. The number diagnosed will need to increase to 350 individuals annually by 2022. Treatment was also expanded to those ≥F0 in 2018. SVR remained constant.
Given these increases in diagnosed and treated cases, the total number of chronicly infected patients, HCC cases due to HCV, decompensated cirrhosis cases and liver related deaths will all decrease drastically, by 95% by 2030.
As shown in table 4, achieving the Swiss Hepatitis Strategy targets in the three cantons will have significant impact on morbidity and mortality in the HCV-infected population. In St Gallen, by 2030 approximately 65 new cases of HCC and 50 new cases of decompensated cirrhosis could be averted and 95 lives could be saved. In Geneva, 25 and 20 new cases of HCC and decompensated cirrhosis, respectively, could be averted by 2030. Additionally, 30 lives could be saved. In Zurich, the largest reduction in morbidity and mortality could be seen. More than 175 new cases of HCC and more than 140 new cases of decompensated cirrhosis could be averted over the next 12 years. Two hundred and sixty lives could be saved by implementing this scenario.
Table 4 Hepatocellular carcinoma and decompensated cirrhosis cases averted, and lives saved from 2015–2030 under the Swiss Hepatitis Strategy, by canton.
| St Gallen | Geneva | Zurich | |
|---|---|---|---|
| Hepatocellular carcinoma cases averted | 65 | 25 | 180 |
| Decompensated cirrhosis cases averted | 50 | 20 | 140 |
| Lives saved | 95 | 30 | 260 |
Values are rounded
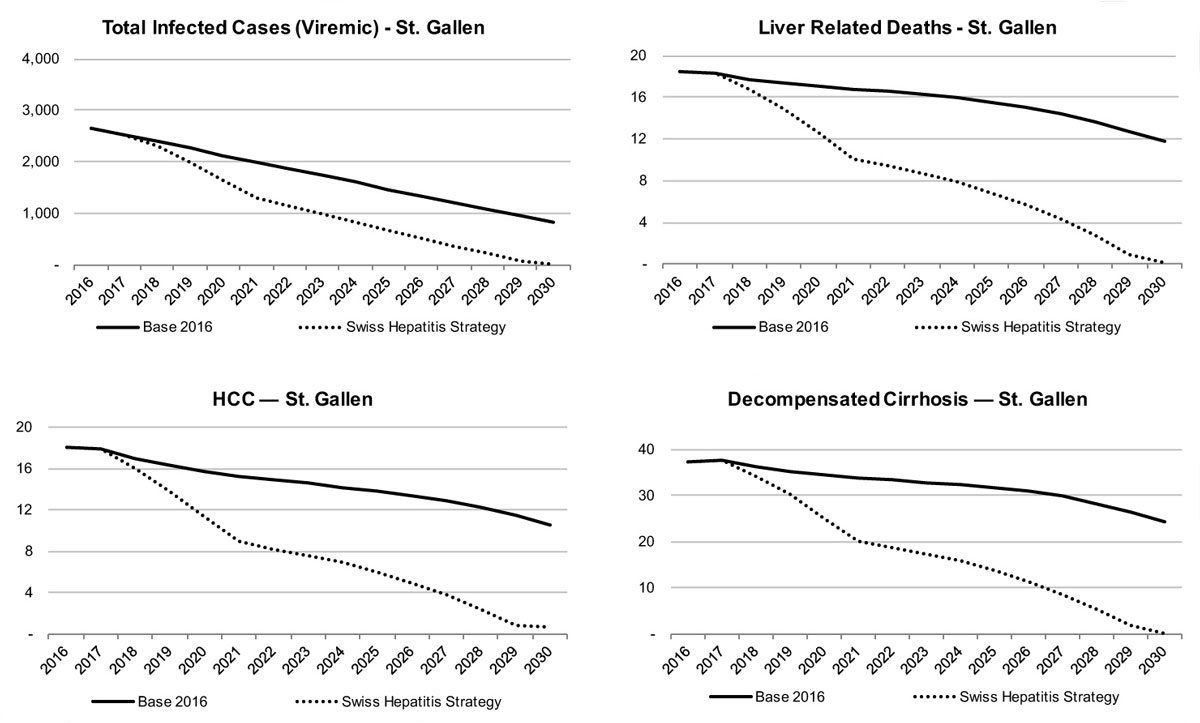
Figure 13 Total viraemic infections, liver related morbidity and mortality by scenario for the canton of St Gallen.
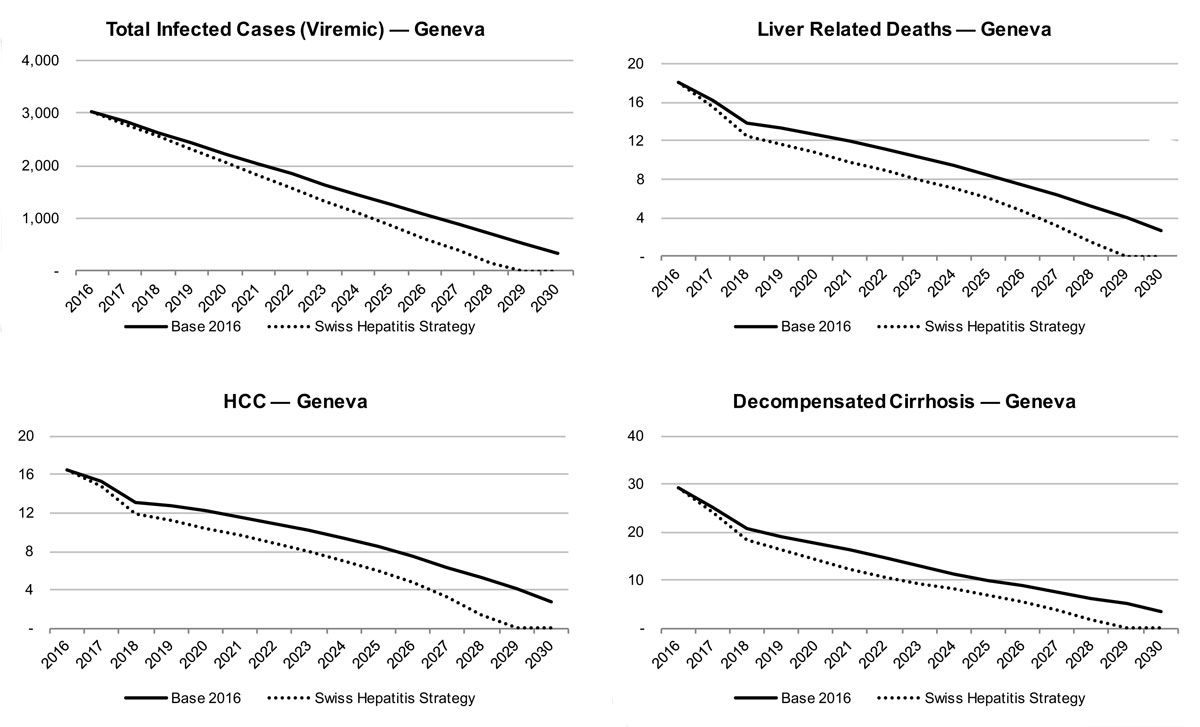
Figure 14 Total viraemic infections, liver related morbidity and mortality by scenario for the canton of Geneva.
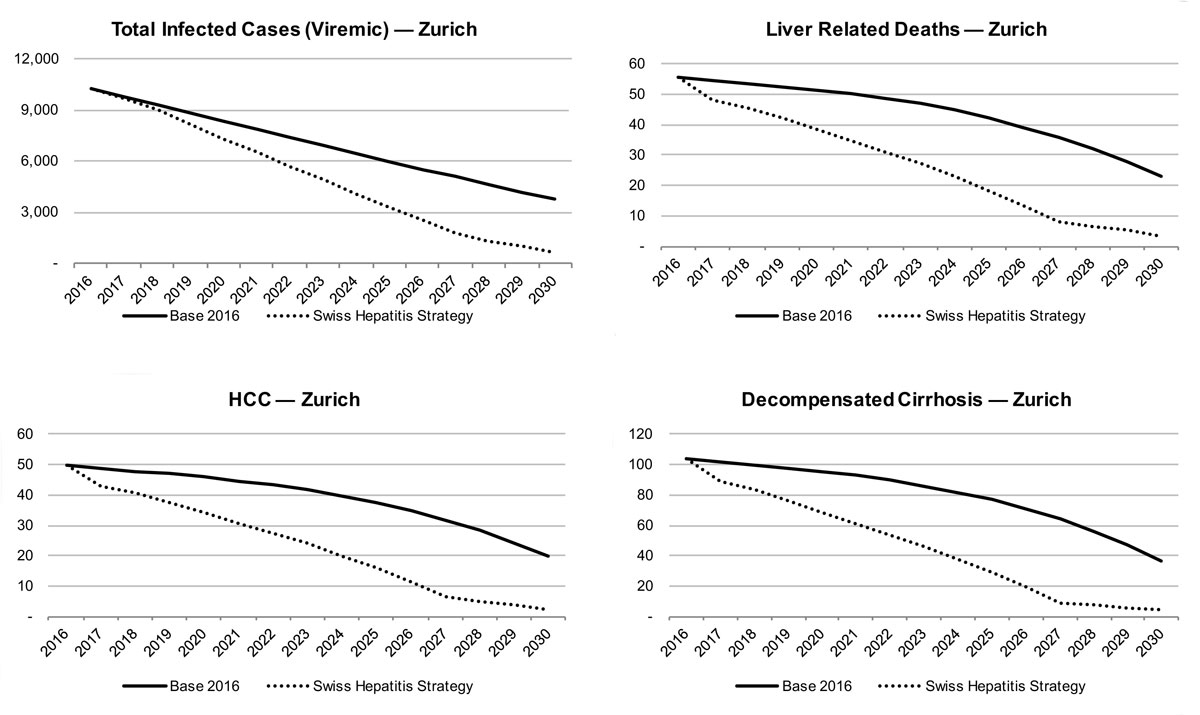
Figure 15 Total viraemic infections, liver related morbidity and mortality by scenario for the canton of Zurich.
This analysis presented the epidemiological burden of hepatitis C in various cantons throughout Switzerland and identified potential scenarios for achieving the Swiss Hepatitis Strategy goals by 2030. Our study estimated a higher prevalence of HCV in Geneva and Zurich than in Switzerland overall. Similar studies have highlighted this difference in infected cases between urban and rural regions. A study from France, for example, noted an increased risk of HCV infection among Parisians as compared with the overall French population [21]. Thus, targeted, evidence-based screening and treatment strategies are needed, as the epidemiological burden is quite diverse across cantons.
The current standard of care in St Gallen, Geneva and Zurich is sufficient to reduce the chronic hepatitis population and HCV-related impact on liver related morbidity and mortality. This, however, is based on the assumption that current trends in numbers of diagnosed and treated patients will be maintained. If efforts are not in place to sustain such a standard of care, reductions in late stage morbidity and mortality would falter. In Georgia, for example, which initatied the world’s first programme to eliminate hepatitis C by 2020, the estimated number of persons starting treatment peaked in September 2016, but has since declined, indicating that interventions to increase access to HCV testing, care and treatment are needed if the country is to achieve its national targets [22]. Similar challenges were faced in Egypt, where additional, outside assistance was needed in order to sustain its formal screening strategy [23]. The healthcare system in Switzerland has been responsive to these issues, with treatment access extended to all patients regardless of fibrosis score in 2017. Although this is a positive step, identifying available patients and linking them to treatment still remains a bottleneck in these regions. Increases in both treatment and diagnosis rates are necessary if St Gallen, Geneva and Zurich are to achieve the Swiss Hepatitis Strategy targets set out in 2014. In Geneva, maintaining the previous standard of care (235 treated patients per year by 2018) would be required to achieve such targets. However, the number of patients diagnosed annually would also need to increase if the strategic goals are to be met. In Zurich, treatment would need to increase by 100 patients and the annual number of diagnosed patients would need to double. A recent Swiss study analysed the age distribution of HCV cases in the country in order to develop better screening strategies. It was found that screening those born between 1951 and 1985 would capture almost 75% of all infections in Switzerland [24]. St Gallen, Geneva and Zurich, which diagnose less than 70% of their viraemic population, could consider such tailored screening strategies in order to capture enough patients to treat in order meet the Swiss strategy goals.
In 2014, the World Health Organization (WHO) established the Global Health Sector Strategy (GHSS) goals for elimination of hepatitis globally by 2030, like the Swiss Hepatitis Strategy, [25]. As of 2017, treatment restrictions were removed across Switzerland, such that all patients were eligible for treatment regardless of fibrosis score. In cantons such as St Gallen and Geneva, where almost half of patients seen in the hospital setting were ≥F2, this policy shift was necessary if the Swiss Hepatitis Strategy and WHO Goals are to be achieved. Other countries and modelling efforts have evaluated the possibility of elimination by 2030 by targeting high-risk populations, such as people who inject drugs (PWID). In Iceland, unique in its geographic isolation and relatively small chronically infected population (880–1300 people), an HCV transmission model was used to estimate the treatment and testing scale up necessary among PWID to achieve the WHO HCV elimination targets [26]. The study evlauted strategies similar to those used in this cantonal analysis, and also showed that additional testing and harm reduction efforts are required [26]. The study also suggested targeted community based strategies that could be used to achieve local elimination of HCV [26]. Efforts such as these regional analyses highlight the importance of tailored, context-specific programmes and could be used by other cities with comparable epidemiological landscapes to define what scale ups in prevention and treatment are required in order to achieve elimination targets.As long as national strategic plans for HCV are warranted on a global level, the feasibility, funding, and implementation of such plans must be considered. Thus, smaller scale policy initiatives targeting specific populations or localities are a tangible step towards achieving global elimination of HCV. This analysis sets forth pragmatic and defined strategies, in three regions across Switzerland, which could be used as models for larger intervention programmes or similar analyses across the country.
Though the prospect of achieving the GHSS goals in Switzerland can be daunting, it is clear from this study that achieving these goals in subregions is plausible, if certain programmatic changes are implemented. Increasing the proportion of diagnosed cases is a key constraint in these cantons. Screening programmes in the “baby boomer” population have previously been estimated to be the most successful in identifying new cases [27] and also proven to be cost effective [28]. In St Gallen and Geneva, where the mean age of patients was the early-to-mid fifties, such screening strategies could be one potential avenue for identifying patients to diagnose and link to care. These policies could then be adapted to other cantons throughout Switzerland or to cities with a comparable age distribution in the HCV-infected population, in order to meet needs in specific treatment settings [10]. Similar modelling conducted in Australia has shown the need to improve the cascade of care from infection to treatment and cure and, in particular, ensure priority populations have access to testing, care and DAA therapy [29]. Such strategies could be implemented by improving follow up in primary care services, such as general practitioner-initiated screening [30, 31].
Although not considered in this analysis, there is also a need for robust harm reduction programmes to supplement these screening and treatment strategies. As the majority of new infections in Europe occur amongst PWID and men who have sex with men (MSM) and engage in high-risk behaviour, the potential for reinfection in these cohorts could partly counteract the benefits achieved by increased treatment. The prevalence of HCV has been decreasing amongst PWID in Switzerland [32], as more than 85% of injection drug users were found to be engaged in harm reduction programmes [33]. However, in the absence of these behavioural interventions, the number of secondary infections would increase regardless of treatment [33]. Additionally, two recent studies in Switzerland considered the impact of increased treatment on the reduction in new infections amongst HIV-positive MSM [34, 35]. It was found that increased treatment initation reduced HCV incident infections by 50% [35] and that intensive interventions could boost long-term efforts for prevention of high-risk behaviours [34]. Thus, systematic population based screening, scale up of treatment, and increased access to harm reduction programmes are necessary for sustained reduction of new HCV infections and elimination of the disease [35].
A number of limitations to the study may affect the forecasts of the model. Without a national surveillance programme, uncertainty in the prevalence estimates will remain. To date, no general population prevalence studies have been conducted in the regions. However, all estimates have been presented with 95% uncertainty intervals to capture such uncertainty in the outcomes. Similarly, though notification data were used to estimate the total number of linked-to-care patients in the cantons, underreporting, especially in high-risk groups, may occur. If this is the case, then policies would need to capture larger numbers of diagnosed patients moving forward. Lastly, no treatment registry exists in Switzerland and thus the true numbers of treated patients in Geneva and Zurich are unknown. As they represent large cities with more prevalent migrant populations, we assumed a proportional distribution of treatment in these regions. Further analysis to identify the number of patients linked to care is warranted.
Elimination of chronic HCV infection in western, eastern, and northern Switzerland by 2030 is possible, but not with the current HCV detection or treatment rate. Intensified HCV screening and increased DAA access are necessary to meet the Swiss Hepatitis Strategy elimination goals over the next 15 years.
The appendices are available in a separate file at https://smw.ch/en/article/doi/smw.2019.14694/.
The authors acknowledge the Swiss Federal Office of Public Health, particularly Jean-Luc Richard, for providing Swiss data and further analysis regarding notification data.
This analysis was supported through Gilead Sciences. Gilead Sciences did not have any influence over the design, implementation, outcomes or the publication of the results. Urs Rusch has receivet a project grant from Gilead. H. Razavi and S. Blach are employees of the CDA foundation. P. Bruggmann has served as advisor and speaker for, and has received project and research grants from Merck, AbbVie, Gilead, BMS and Mundipharma. B. Müllhaupt served as an advisor and/or speaker for Gilead, AbbVie, Astra, Intercept, Bayer, BMS and received research support from Gilead. F. Negro served as an advisor for Gilead, AbbVie, Merck and has received research support from Gilead. D. Semela has received consulting and speaker fees from AbbVie, Gilead and MSD.
1World Health Organization. Global hepatitis report 2017. Geneva, Switzerland: WHO; 2017.
2 Blach S , Zeuzem S , Manns M , Altraif I , Duberg A-S , Muljono DH , et al.; Polaris Observatory HCV Collaborators. Global prevalence and genotype distribution of hepatitis C virus infection in 2015: a modelling study. Lancet Gastroenterol Hepatol. 2017;2(3):161–76. doi:.https://doi.org/10.1016/S2468-1253(16)30181-9
3Swiss Hepatitis Strategy 2014-2030. Time to Act Now! Process Paper - A Living Document. http://www.hepatitis-schweiz.ch/files/Dokumente/Process_Paper_2nd_version_final.pdf. 2015.
4Swiss Federal Office of Public Health. [Situation Analysis of Hepatitis B and C in Switzerland.] Bern: Federal Office of Public Health; 2017.
5 Wedemeyer H , Duberg AS , Buti M , Rosenberg WM , Frankova S , Esmat G , et al. Strategies to manage hepatitis C virus (HCV) disease burden. J Viral Hepat. 2014;21(Suppl 1):60–89. doi:.https://doi.org/10.1111/jvh.12249
6 Gane E , Kershenobich D , Seguin-Devaux C , Kristian P , Aho I , Dalgard O , et al. Strategies to manage hepatitis C virus (HCV) infection disease burden - volume 2. J Viral Hepat. 2015;22(s1, Suppl 1):46–73. doi:.https://doi.org/10.1111/jvh.12352
7 Alfaleh FZ , Nugrahini N , Matičič M , Tolmane I , Alzaabi M , Hajarizadeh B , et al. Strategies to manage hepatitis C virus infection disease burden - volume 3. J Viral Hepat. 2015;22(Suppl 4):42–65. doi:.https://doi.org/10.1111/jvh.12474
8 Razavi H , Robbins S , Zeuzem S , Negro F , Buti M , Duberg A-S , et al.; European Union HCV Collaborators. Hepatitis C virus prevalence and level of intervention required to achieve the WHO targets for elimination in the European Union by 2030: a modelling study. Lancet Gastroenterol Hepatol. 2017;2(5):325–36. doi:.https://doi.org/10.1016/S2468-1253(17)30045-6
9 Chen DS , Hamoudi W , Mustapha B , Layden J , Nersesov A , Reic T , et al. Strategies to manage hepatitis C virus infection disease burden-Volume 4. J Viral Hepat. 2017;24(Suppl 2):44–63. doi:.https://doi.org/10.1111/jvh.12759
10 Lazarus JV , Wiktor S , Colombo M , Thursz M ; EASL International Liver Foundation. Micro-elimination - A path to global elimination of hepatitis C. J Hepatol. 2017;67(4):665–6. doi:.https://doi.org/10.1016/j.jhep.2017.06.033
11Polaris Observatory. [Internet] Cited 2017 September 1. Available from: http://polarisobservatory.org/.
12Federal Statistical Office FSO. Switzerland's population 2016. Neuchâtel: FSO;2017.
13 Razavi H , Waked I , Sarrazin C , Myers RP , Idilman R , Calinas F , et al. The present and future disease burden of hepatitis C virus (HCV) infection with today’s treatment paradigm. J Viral Hepat. 2014;21(Suppl 1):34–59. doi:.https://doi.org/10.1111/jvh.12248
14 Sibley A , Han KH , Abourached A , Lesmana LA , Makara M , Jafri W , et al. The present and future disease burden of hepatitis C virus infections with today’s treatment paradigm - volume 3. J Viral Hepat. 2015;22(Suppl 4):21–41. doi:.https://doi.org/10.1111/jvh.12476
15 Hatzakis A , Chulanov V , Gadano AC , Bergin C , Ben-Ari Z , Mossong J , et al. The present and future disease burden of hepatitis C virus (HCV) infections with today’s treatment paradigm - volume 2. J Viral Hepat. 2015;22(Suppl 1):26–45. doi:.https://doi.org/10.1111/jvh.12351
16 Bedossa P , Poynard T ; The METAVIR Cooperative Study Group. An algorithm for the grading of activity in chronic hepatitis C. Hepatology. 1996;24(2):289–93. doi:.https://doi.org/10.1002/hep.510240201
17Swiss Federal Office of Public Health (FOPH). Swiss mandatory notifications for hepatitis C per canton. FOPH/CD/EPI/RIC 29.08.2016. Berne: Swiss Federal Office of Public Health (FOPH); 2016.
18 IMS Health International. 2015;Q1:
19Swiss hepatitis C cohort study report-March 31, 2012. [cited 3/7/2013]; Available from: http://www.swisshcv.ch/pdf/Report_SCCS_March_31_2012.pdf.
20Swiss hepatitis C cohort study. Study Population; Some salient features 2014. [cited 2014 4/15/2014]; Available from: http://www.swisshcv.ch/study-population.html.
21 Meffre C , Le Strat Y , Delarocque-Astagneau E , Dubois F , Antona D , Lemasson JM , et al. Prevalence of hepatitis B and hepatitis C virus infections in France in 2004: social factors are important predictors after adjusting for known risk factors. J Med Virol. 2010;82(4):546–55. doi:.https://doi.org/10.1002/jmv.21734
22 Nasrullah M , Sergeenko D , Gvinjilia L , Gamkrelidze A , Tsertsvadze T , Butsashvili M , et al. The Role of Screening and Treatment in National Progress Toward Hepatitis C Elimination - Georgia, 2015-2016. MMWR Morb Mortal Wkly Rep. 2017;66(29):773–6. doi:.https://doi.org/10.15585/mmwr.mm6629a2
23Omran H. World Bank to lend Health ministry $200m for hepatitis C screening: minister, in Daily News Egypt. 2018: Egypt.
24 Bruggmann P , Negro F , Bihl F , Blach S , Lavanchy D , Müllhaupt B , et al. Birth cohort distribution and screening for viraemic hepatitis C virus infections in Switzerland. Swiss Med Wkly. 2015;145:w14221. doi:.https://doi.org/10.4414/smw.2015.14221
25World Health Organization. Global Health Sector Strategy on Viral Hepatitis, 2016–2021 Towards Ending Viral Hepatitis. Geneva: World Health Organization; 2016.
26 Scott N , Ólafsson S , Gottfreðsson M , Tyrfingsson T , Rúnarsdóttir V , Hansdottir I , et al. Modelling the elimination of hepatitis C as a public health threat in Iceland: A goal attainable by 2020. J Hepatol. 2018;68(5):932–9. doi:.https://doi.org/10.1016/j.jhep.2017.12.013
27 Müllhaupt B , Bruggmann P , Bihl F , Blach S , Lavanchy D , Razavi H , et al. Modeling the Health and Economic Burden of Hepatitis C Virus in Switzerland. PLoS One. 2015;10(6):e0125214. doi:.https://doi.org/10.1371/journal.pone.0125214
28 Rein DB , Smith BD , Wittenborn JS , Lesesne SB , Wagner LD , Roblin DW , et al. The cost-effectiveness of birth-cohort screening for hepatitis C antibody in U.S. primary care settings. Ann Intern Med. 2012;156(4):263–70. doi:.https://doi.org/10.7326/0003-4819-156-4-201202210-00378
29 Scott N , Doyle JS , Wilson DP , Wade A , Howell J , Pedrana A , et al. Reaching hepatitis C virus elimination targets requires health system interventions to enhance the care cascade. Int J Drug Policy. 2017;47:107–16. doi:.https://doi.org/10.1016/j.drugpo.2017.07.006
30 Scott N , Doyle JS , Wilson DP , Wade A , Howell J , Pedrana A , et al. Reaching hepatitis C virus elimination targets requires health system interventions to enhance the care cascade. Int J Drug Policy. 2017;47:107–16. doi:.https://doi.org/10.1016/j.drugpo.2017.07.006
31 Snow K , Scott N , Clothier HJ , MacLachlan JH , Cowie B . Limited provision of diagnostic services to Victorians living with hepatitis C antibodies, 2001-2012: a multi-level modelling analysis. Aust N Z J Public Health. 2017;41(2):193–8. doi:.https://doi.org/10.1111/1753-6405.12560
32 Dubois-Arber F , Balthasar H , Huissoud T , Zobel F , Arnaud S , Samitca S , et al. Trends in drug consumption and risk of transmission of HIV and hepatitis C virus among injecting drug users in Switzerland, 1993-2006. Euro Surveill. 2008;13(21):18881. doi:.https://doi.org/10.2807/ese.13.21.18881-en
33 Bruggmann P , Blach S , Deltenre P , Fehr J , Kouyos R , Lavanchy D , et al. Hepatitis C virus dynamics among intravenous drug users suggest that an annual treatment uptake above 10% would eliminate the disease by 2030. Swiss Med Wkly. 2017;147:w14543.
34 Salazar-Vizcaya L , Kouyos RD , Fehr J , Braun D , Estill J , Bernasconi E , et al.; Swiss HIV Cohort Study. On the potential of a short-term intensive intervention to interrupt HCV transmission in HIV-positive men who have sex with men: A mathematical modelling study. J Viral Hepat. 2018;25(1):10–8. doi:.https://doi.org/10.1111/jvh.12752
35Braun DL, Hampel BH, Nguyen H, Flepp M, Stoeckle M, Beguelin C, et al. A treatment as prevention trial to eliminate HCV in HIV+ MSM: The Swiss HCVREE Trial. Conference on Retroviruses and Opportunistic Infections. 2018: Boston, Massachusetts.
UR and SR contributed equally.
This analysis was supported through Gilead Sciences. Gilead Sciences did not have any influence over the design, implementation, outcomes or the publication of the results. Urs Rusch has receivet a project grant from Gilead. H. Razavi and S. Blach are employees of the CDA foundation. P. Bruggmann has served as advisor and speaker for, and has received project and research grants from Merck, AbbVie, Gilead, BMS and Mundipharma. B. Müllhaupt served as an advisor and/or speaker for Gilead, AbbVie, Astra, Intercept, Bayer, BMS and received research support from Gilead. F. Negro served as an advisor for Gilead, AbbVie, Merck and has received research support from Gilead. D. Semela has received consulting and speaker fees from AbbVie, Gilead and MSD.