Nature and duration of symptoms among callers to the coronavirus hotline in the canton of Fribourg, Switzerland
Jean-Pierre Zellweger, Catherine Plancherel, Frédéric Poulet, Raphaël M. Zellweger, Erina Panchaud, Jeanette Muntwyler, Grégoire Seitert
Summary
AIMS OF THE STUDY: As the COVID-19 pandemic rages around the world, studies describing the disease mostly focus on severely ill patients in hospital or intensive care settings. Much less is known about COVID-19 in other patient groups. Our study addresses this knowledge gap by describing symptoms and risk factors in individuals who called the coronavirus hotline in the canton of Fribourg, during the peak of the COVID-19 outbreak in Switzerland.
METHODS: From 18 March to 15 June 2020, the Food Safety and Veterinary Office of the canton of Fribourg organised and supervised a hotline, enabling people with questions or suffering from COVID 19 symptoms to talk to health professionals and receive advice.
RESULTS: Between 18 March and 15 June 2020, the three physicians on duty received 430 calls from 410 people. Twenty people called the hotline twice. The average duration of symptoms at the time of the call was 6.8 days. Based on symptom severity and current guidelines, 113 people (27%) were referred to a facility where a diagnostic test could be performed, 111 (26%) were referred for clinical examination and 34 (8%) were referred directly to an emergency department. In total, 151 people were tested and 40% of them had a positive polymerase chain-reaction test. Amongst those tested, the proportion of positive results was higher in people aged ≥65 years, people reporting known contact, fever, or duration of symptoms ≥5 days. Multivariate analysis of risk factors adjusted for clinical symptoms showed a significant relationship only for age ≥65 (odds ratio [OR] 3.16), symptoms for ≥5 days (OR 3.27) and history of contact (OR 2.90). The hotline doctors re-contacted 36 people who tested positive and 20 people who tested negative, aged between 19 and 91 years, 18 of whom had been hospitalised. Forty-two of 56 (75%) people still had symptoms 30 days after the onset of the illness. The average duration of the illness was 47 days in the patients hospitalised, 38 days in the outpatients and 23 days in the people who tested negative.
CONCLUSIONS: Within our collective of callers, apart from fever, clinical symptoms were not clearly indicative of test positivity. The course of COVID-19 may be long, the majority of patients still have symptoms 1 month after the onset of the disease, even if tested negative. Many people who tested negative at the onset of the disease may have suffered from COVID-19 without being diagnosed as such.
IntroductionThe current SARS-CoV-2 pandemic has disrupted the world like no other infectious disease in the past century and has presented a number of health problems to the medical community. Some problems were similar to those that arise during seasonal influenza epidemics, and some were new.
In reaction to the sudden increase in calls to the emergency clinics of the hospitals and medical institutions, the local health authorities of several Swiss cantons, including Fribourg, organised a hotline enabling people with questions, fearing that they may have been contaminated or suffering from symptoms suggestive of COVID-19, to talk to health professionals and receive advice without sending all people to an emergency unit of a hospital.
In an attempt to better understand the specifics of the COVID-19 infection among the hotline callers, a retrospective analysis was performed by a group of physicians who received telephone calls from people with symptoms suggestive of COVID-19 during the initial period of the outbreak. The aims were to identify the risk factors and symptoms associated with a positive diagnostic tests, to describe the similarities and differences between the people with a positive and a negative diagnostic test, and to compare the outcome of the illness in people with a positive test (hospitalised or not) and with a negative test result. Whereas most studies to date related exclusively to hospitalised patients with severe disease, only a few focused on the outcome and duration of disease in patients who had not been in intensive care. Our study addressed this important knowledge gap. The study was intended as an aid to clinicians who may meet people with acute or late symptoms of COVID-19.
Method
From 13 March to 15 June 2020, the Food Safety and Veterinary Office of the canton of Fribourg organised and supervised a public hotline on behalf of the Cantonal SARS-CoV2 Control Board and the emergency department of the regional hospital in Fribourg. The staff was composed of nurses or medical students (level 1) who received the calls, registered the demographic information and gave first advice according to the current guidelines from the Federal Office of Public Health (FOPH). Initially, urgent calls were transmitted to the emergency department of the local hospital. Because of the rapid increase in the number of calls (656 on 16 March), three independent medical doctors and several psychologists were hired from 18 March (level 2) to decrease the work load of the staff in the hospital. In the event of medical questions or a possible emergency, the callers could be connected to a physician or to a psychologist via a call-back mechanism. The physicians spoke with the callers and conducted a semi-structured interview to assess the nature, duration and severity of symptoms and the presence of known risk factors, and offered advice. The physicians could refer the callers directly to one of the services that took a diagnostic nasopharyngeal swab (increasing from one to five during the acute phase of the outbreak), or to an emergency department (from one to four) in the event of severe symptoms requiring immediate medical attention, or provide callers with information on home isolation measures. The clinical internists hired by the SARS-CoV2 control board took turns on duty daily from 08:00 to 20:00.
The demographics and risk factors, and the nature and duration of symptoms of those who spoke with one of the physicians were recorded in a common file. The test results were transmitted by the laboratory of the local hospital to the cantonal medical officer and to the control board. The indications for the polymerase chain-reaction (PCR) diagnostic test were those of the FOPH and cantonal doctor’s office guidelines and varied over time, being very restrictive at the beginning of the epidemic owing to the limited availability of the diagnostic tests, then becoming progressively broader at the end of the period, after the introduction of the contact tracing procedure and the improvement in availability of the diagnostic test.
Three months after the telephone call to the hotline, the three medical doctors re-contacted those who tested positively (not all could be reached) and a random group of those who tested negatively (control group) to assess the duration, nature and severity of symptoms in the weeks following the first call, using a similar semi-structured interview. All people contacted were informed of the purpose of the call and gave oral informed consent for the anonymous recording of the information and its use for educational purposes for medical practitioners. Due to the emergency situation, no approval from an ethical committee was sought.
The following demographic factors and symptoms were included in the analysis: age, sex, being a healthcare worker, having a risk factor (age ≥65 years, diabetes, immune deficiency or high blood pressure), history of contact with a known case, having symptoms for ≥5 days, presence of cough, dyspnoea, chest pain, fever, pharyngitis and anosmia. The test used to determine statistical significance is indicated in the legend of the respective tables and figures. The multiple correspondence analysis (MCA) was performed using all the demographic factors and symptoms quoted above. Statistical analysis was performed with R version 4.0.0. using following packages: dplyr, ggplot2, tableone, UpsetR, FactoMineR and factoextra.
ResultsInitial data (first call)
Between 13 March and 15 June 2020, the staff of the hotline received 8037 calls, of which 430 were transmitted to the three physicians on duty. Twenty people called the hotline twice. For those who called twice, both calls were recorded because the nature and severity of symptoms were different on the second call. In most cases, the reason for the call was the inability to consult a family doctor quickly or to find out if a diagnostic test had to be performed, and if so when and where. In five cases, either the request was not due to a health problem but for general medical information, or the individuals did not respond to the doctor’s call and no demographic or clinical details could be recorded. The study flow chart is shown in figure 1.
Figure 1: Study flow chart, including follow-up.
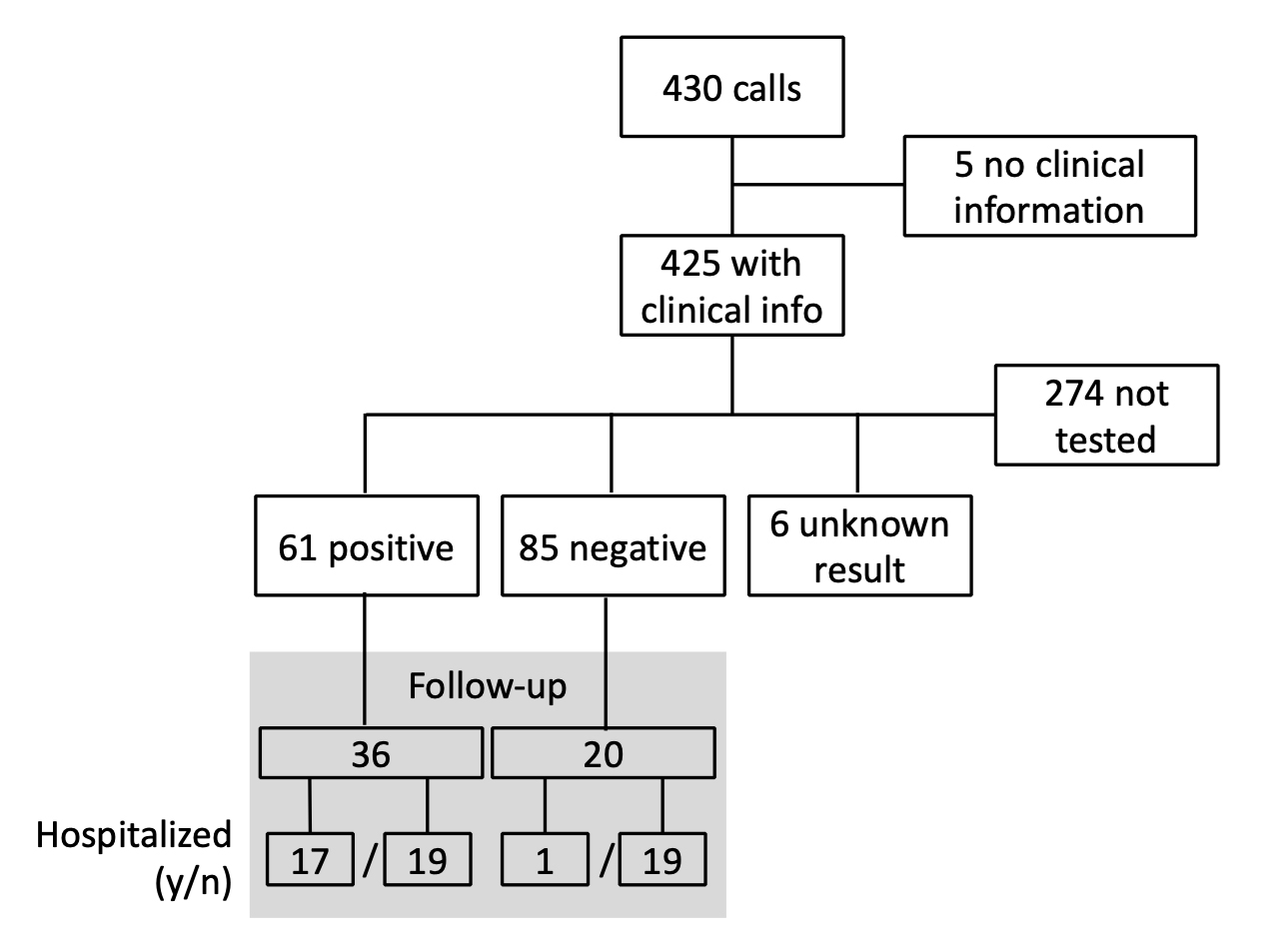
After assessment of risk factors and symptom severity, 113 people (27%) were referred to a facility where a diagnostic test could be performed, 111 (26%) were referred for clinical examination to a facility where the final decision to perform a diagnostic test was made according to severity criteria, and 34 (8%) were referred directly to an emergency department. In 166 cases, callers were advised to remain in isolation while waiting for the evolution of symptoms and call back in the event of symptom worsening. One person was tested twice with two negative test results. People who were tested later without calling the hotline again were not registered in the database. In six cases, the result of the test is unknown.
The distribution of demographic and risk factors is reported in table 1. In people who were tested according to the guidelines, results were positive in two thirds of people over the age of ≥65, half of the men and one third of the women, one third of the healthcare workers, half of the people with a risk factor and more than half of the contacts. No positive test result was observed in people under the age of 20 years.
Table 1: Demographic characteristics and test results of the people who called the coronavirus hotline between 18 March and 15 June 2020.
Total
Test
Test result*
Not tested
%
Tested
%
Positive
%
Negative
%
Total
425
274
64
151
36
61
40
84
56
0–20 years
33
23
67
10
33
0
0
10
100
21–65 years
327
215
66
111
34
41
37
64
58
>65 years
65
35
48
30
52
20
67
10
33
Men
169
101
58
68
42
33
48
32
48
Women
256
173
67
83
33
28
34
52
63
Health care workers
82
37
45
45
55
15
33
28
62
Risk factors
181
106
56
75
44
36
48
35
47
Known contacts
68
36
53
32
47
18
56
10
31
* Six cases with unknown test results are omitted
The distribution of clinical symptoms for individuals who were not tested, and individuals with a positive or a negative test result, is presented in table 2. The most common complaints, by decreasing order of frequency, were cough, dyspnoea, chest pain, fever, pharyngitis (including ageusia), and anosmia. Half of the people had had symptoms for ≥5 days at the time of calling. A statistical difference between the groups was observed only for fever and for a symptom duration ≥5 days, which were more frequent in people testing positive, and for pharyngitis, which was more frequent in people testing negative.
Table 2: Prevalence of symptoms across stratified by test category. Proportion with symptom in brackets (%).
Risk factor or symptom
Overall
(n =419)
Not tested
(n = 274)
Tested, result negative
(n = 84)
Tested, result positive
(n = 61)
p-value*
Cough (%-yes)
337 (80.4)
224 (81.7)
62 (73.8)
51 (83.6)
0.202
Dyspnoea (%-yes)
266 (63.5)
172 (62.8)
49 (58.3)
45 (73.8)
0.149
Chest pain (%-yes)
224 (53.4)
141 (51.5)
51 (60.7)
32 (52.5)
0.34
Fever (%-yes)
199 (47.5)
121 (44.2)
38 (45.2)
40 (65.6)
0.009
Pharyngitis (%-yes)
143 (34.1)
91 (33.2)
37 (44.0)
15 (24.6)
0.045
Anosmia (%-yes)
38 (9.1)
21 (7.7)
9 (10.7)
8 (13.1)
0.348
Symptoms ≥5 days (%-yes)
212 (50.6)
137 (50.0)
32 (38.1)
43 (70.5)
0.001
Six individuals with unknown test results were excluded from the analysis.
* From Fisher’s exact test
Subsequently, we focused on tested individuals only, to understand the association between the presence of a symptom or risk factor and test positivity. Among the individuals who were tested, the distribution of positive and negative test results stratified by clinical symptoms and risk factors is presented in figure 2. Among those tested, the proportion of positive test results was higher among people aged ≥65 years, in people reporting known contact, fever, or with symptoms lasting ≥5 days (fig. 2).
Figure 2: Proportion of positive and negative tests by the presence or absence of different risk factors and symptoms. Statistical significance based on the chi-square test is indicated as follow: * for p <0.05, ** for p <0.01, *** for p <0.001. HCW = healthcare worker
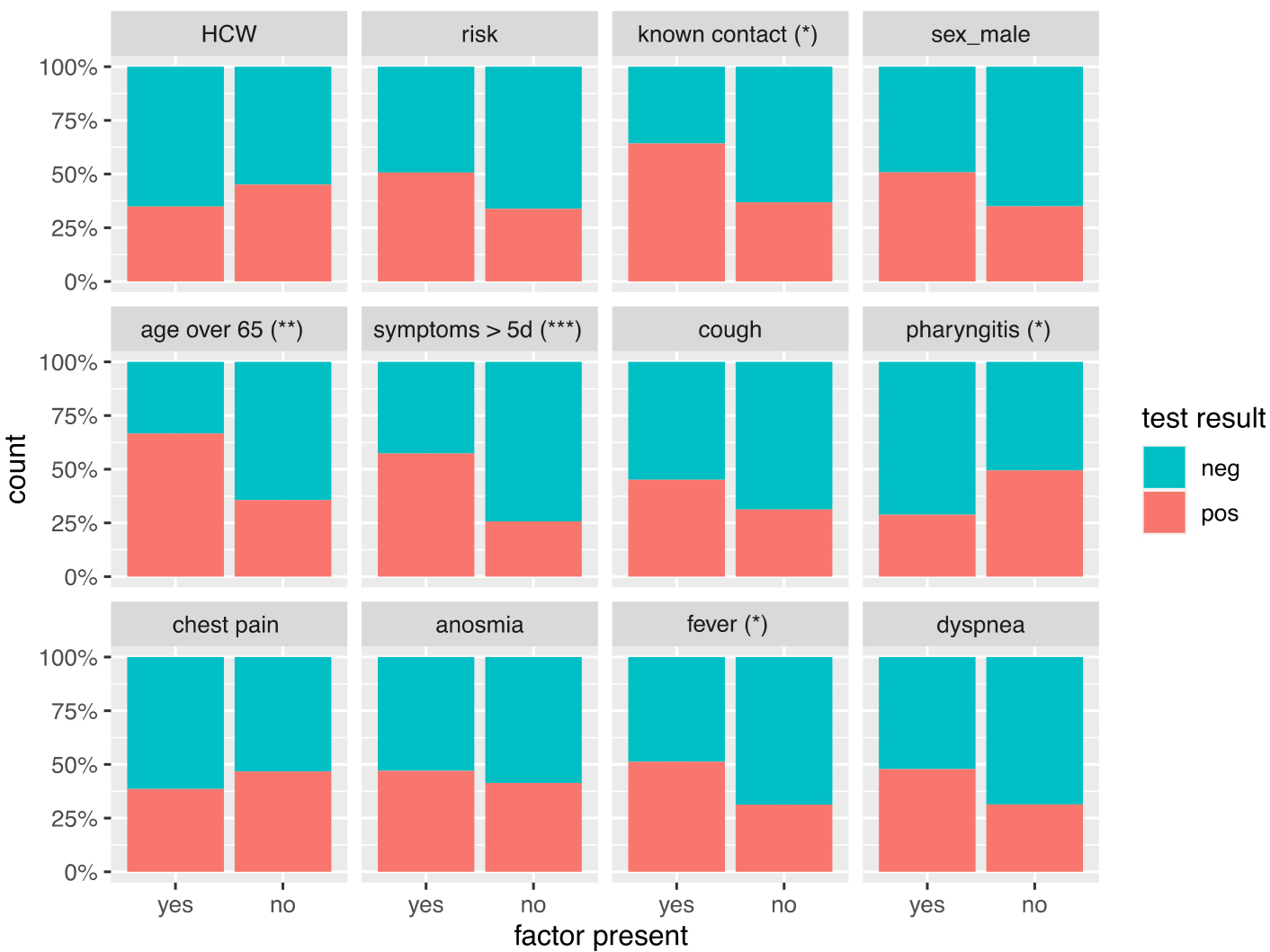
Univariate logistic regression analysis showed that test positivity was associated with age ≥65 years, symptoms lasting ≥5 days, contact with a known case, fever and presence of risk factors. After adjustment for clinical symptoms, test positivity was still significantly associated with age ≥65 (odds ratio [OR] 3.16), duration of symptoms of ≥5 days (OR 3.27) and history of contact (OR 2.90) (table 3).
Table 3: Logistic regression analysis, univariable (crude, top) and adjusted for clinical symptoms (bottom).
OR
95% CI
p-value
Crude
Age ≥65
3.61
1.58–8.74
0.0031
Sex male
1.92
0.98–3.77
0.0569
Healthcare worker
0.65
0.31–1.35
0.2566
Risk
2.02
1.04–3.97
0.0402
Known contact
3.10
1.33–7.56
0.0099
Symptoms ≥5 days
3.88
1.94–8.00
0.0002
Cough
1.81
0.80–4.32
0.1635
Pharyngitis
0.41
0.20–0.84
0.0171
Chest pain
0.71
0.37–1.39
0.3219
Anosmia
1.26
0.45–3.50
0.6578
Fever
2.31
1.18–4.61
0.0162
Dyspnoea
2.01
0.99–4.19
0.0564
Adjusted for clinical symptoms
Age ≥65
3.16
1.33–7.90
0.0108
Symptoms ≥5 days
3.27
1.56–7.05
0.0020
Known contact
2.90
1.19–7.48
0.0220
CI = confidence interval; OR = odds ratio
After investigating the association between individual symptoms or risk factors and test positivity, we assessed the combinations of symptoms or risk factors observed in callers who were tested. Individuals with unknown test results were omitted from this analysis. The combinations of risk factors encountered most frequently in the tested individuals are shown in figure 3.
Figure 3: Demographic and risk factor combinations in individuals tested and having a known test result. Only the 10 most frequent combinations are shown. Presence of a factor is indicated by a black dot. The “intersection size” denotes the number of individuals with a certain combination of factors and is depicted above the various combinations. The “set size” is the total number of individuals with a certain risk factor and is indicated on the left of the graph. The red portion of the bars indicate individuals with a positive test result. HCW = healthcare worker.
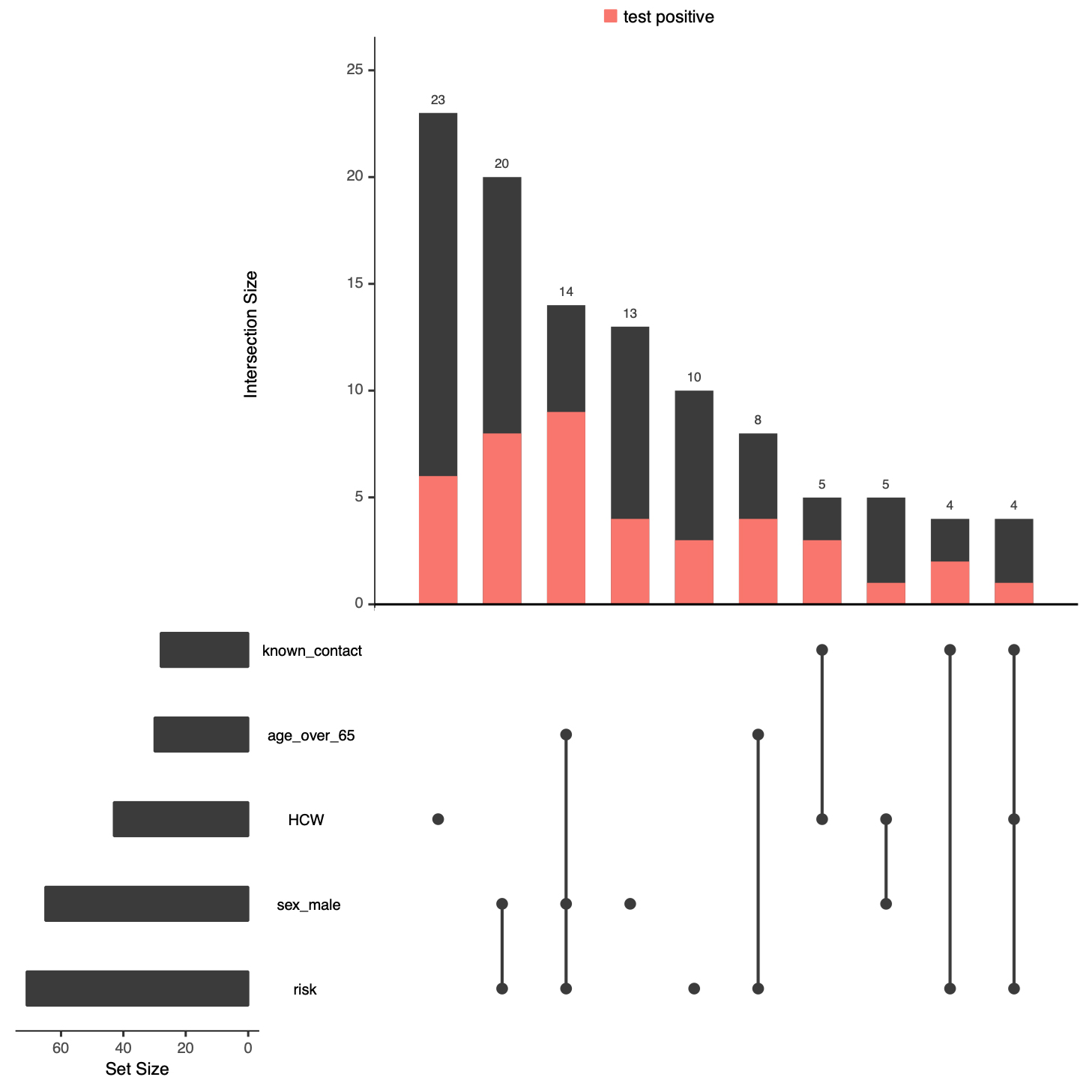
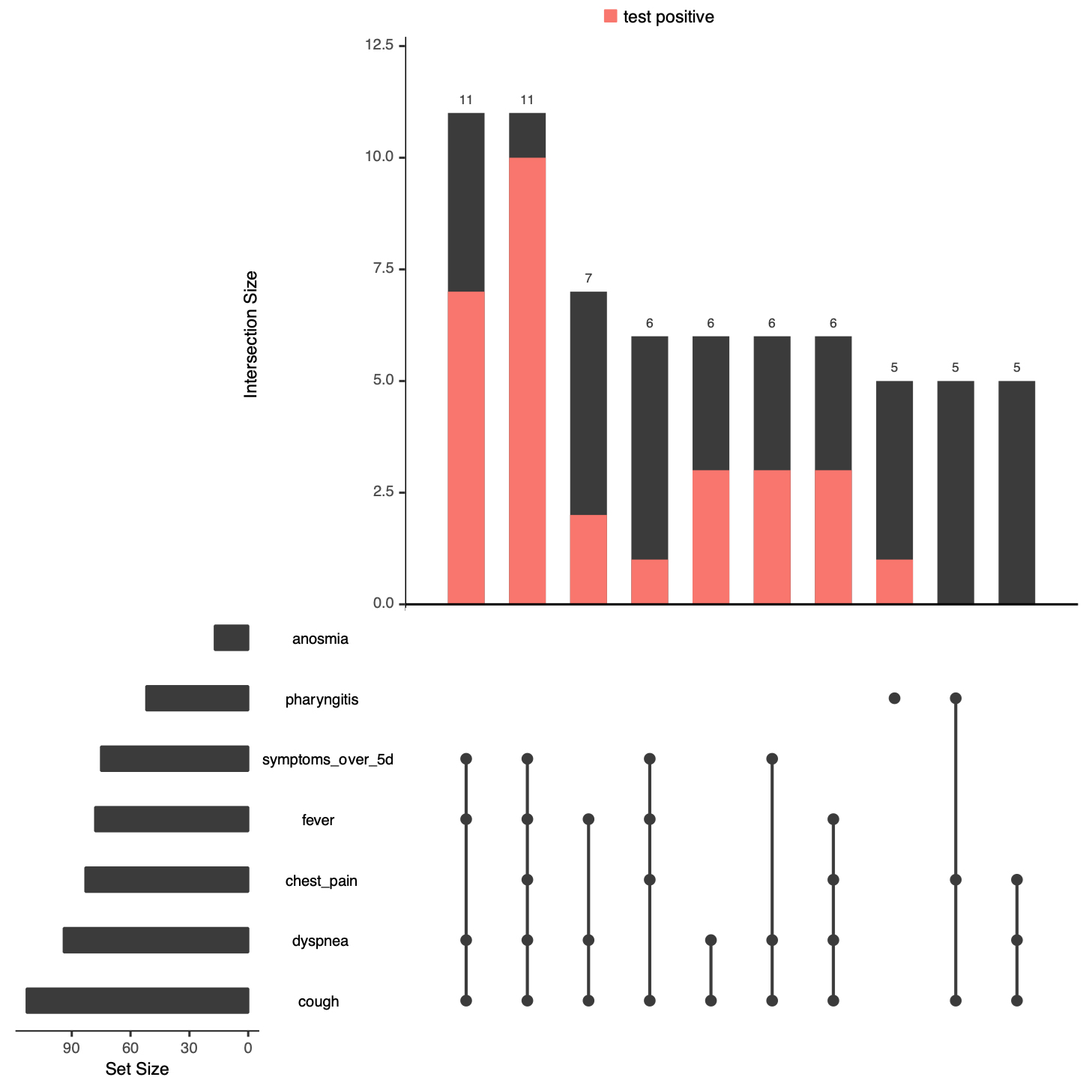
A total of 23 healthcare workers without any other risk factor were tested, but only 6 of them tested positively. The next most frequent combination of risk factors was being male and having a risk factor, which was followed by being male, having a risk factor and being ≥65. Similarly, the combinations of symptoms frequently seen in tested individuals are depicted in figure 4. The most frequent combinations of clinical symptoms in the tested population were cough, dyspnoea, fever, and a duration of symptoms of ≥5 days, with or without chest pain.
Figure 4: Clinical symptom combinations in individuals tested and having a known test result. See explanations in figure 2. Anosmia was not present in the 10 most frequent clinical symptom combinations depicted on the graph.
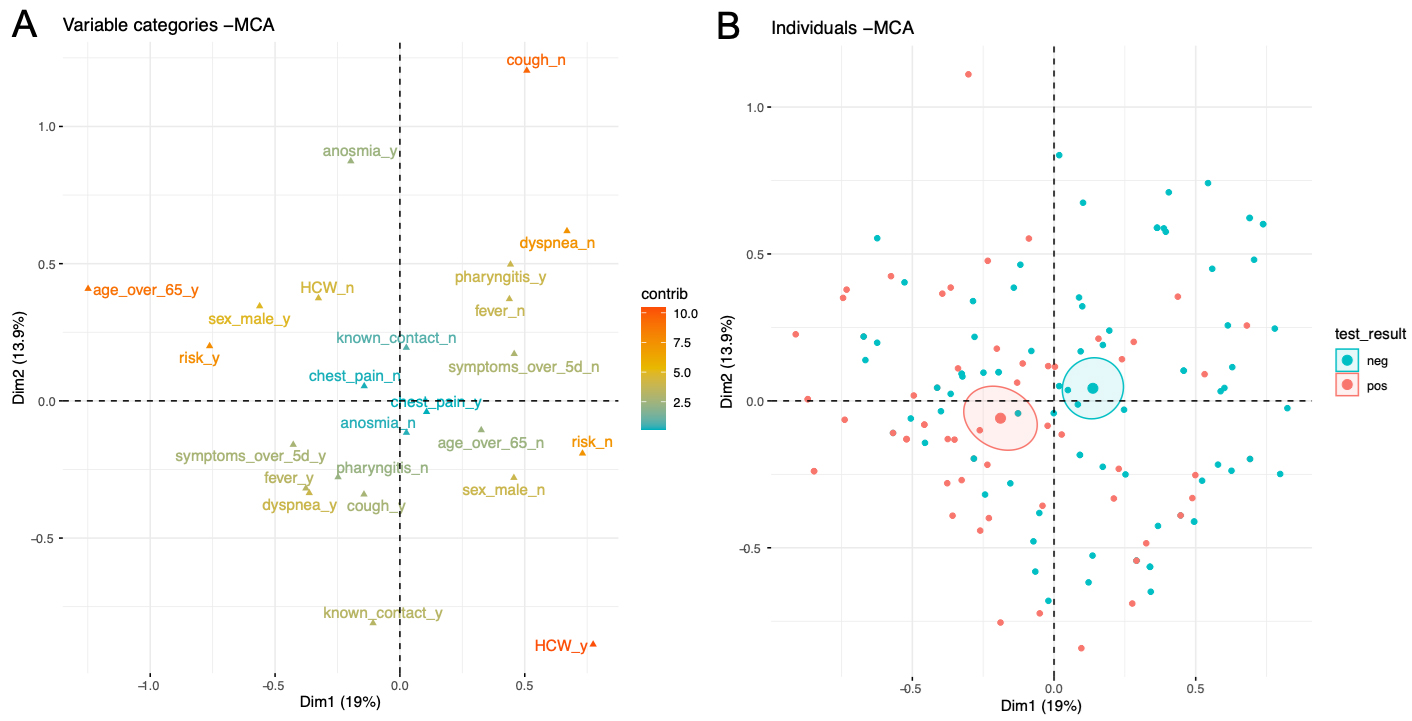
Finally, multiple correspondence analysis (MCA) was performed with all risk factors and symptoms of tested individuals with a known test result (fig. 5). MCA is an exploratory dimensional reduction technique that allows the graphic representation of multidimensional datasets to help uncover potential patterns in individuals. The variable plot (fig. 5A) represents the variables and their contribution to the variance of the individuals. The closer two variables are located, the more correlated they are. The individual plot (fig. 5B) shows that individuals with a positive (red) and negative (blue) test are mostly overlapping on the MCA plane. This indicates that these two groups were largely similar with respect to the risk factors and symptoms analysed during this study. Note that the centroid of the group of individuals having a positive test (red) is shifted slightly to the left of the group of individuals with a negative test (blue). This confirms, for example, that age ≥65 years (far-left on the variable graph) is more frequently observed in individuals who tested positive, as already seen in the logistic regression.
Figure 5: Multiple correspondence analysis (MCA) of all individuals tested and with a known test result. The variable plot (A) represents the variables and their contribution to the variance of the individuals in colour. The closer two variables are on the plane, the more they are correlated. In B, the individual plot shows all individuals coloured by test negativity (blue) or positivity (red). The large symbol represents the centroid of the cloud of points, and the ellipse represents the 95% confidence interval around the centroid.
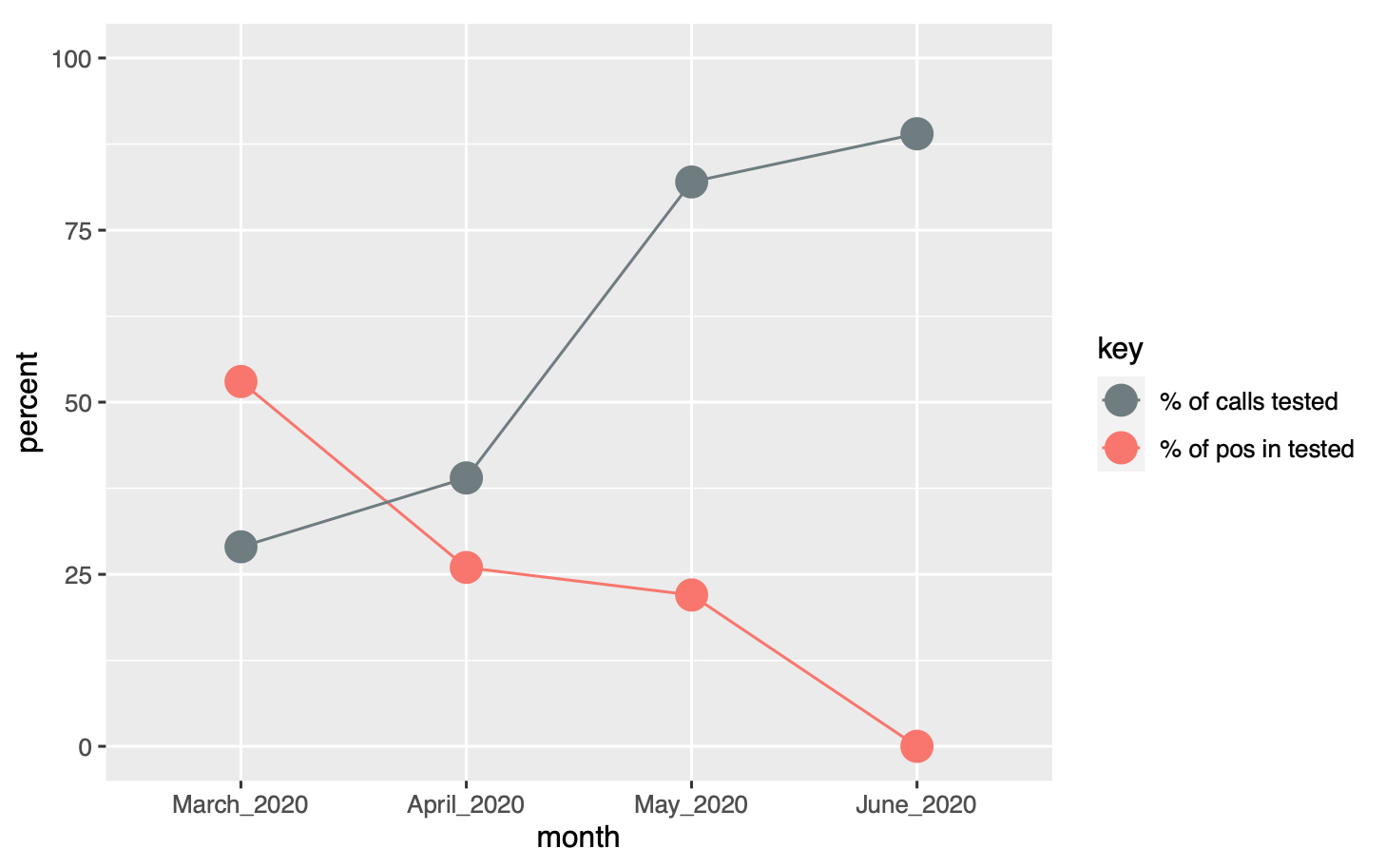
The proportion of people tested among all people calling varied over time, from 29% in March 2020 to 89% in May, following the change in official recommendations (broadening of the indication for testing). Conversely, the proportion of positive results among those tested decreased from 46% in March to 4% in May (fig. 6).
Figure 6: Proportion of calls tested (grey) and proportion of positive results (red) over time (March–June 2020).
Case follow-up
Three months after the initial call, the medical doctors re-contacted one by one all persons with a positive test result and a random sample of persons with a negative test result. Persons who failed to answer the call twice were not considered for follow-up. They could reach 36 people with a positive PCR test result (one of them had been tested after the initial call), and 20 people with a negative test result. Seventeen people with a positive test result and one person with a negative test result had been hospitalised. The mean duration of the illness was 47 days for people hospitalised, 38 days for the non-hospitalised patients and 23 days in people with a negative test result, the difference probably reflecting the difference in illness severity. Forty-two people (75%) still had symptoms 30 days after the initial onset of disease. Several people mentioned the persistence of anosmia or agueusia after 1 month and a weight loss of up to 12 kg (table 4).
Table 4: Demographic characteristics, type and duration of symptoms in 56 patients with positive (inpatient and outpatient) and negative test results.
Positive test
Negative test* (n = 20)
Inpatient
(n = 17)
Outpatient
(n = 19)
Age
46–91
26–83
19–73
Sex (m/f)
9/8
8/11
5/15
Healthcare workers
2
7
11
Contact
5
7
2
Risk
13
9
8
Hospitalised
17
0
1
Mean symptom duration (d)
47
38
23
Symptoms at 1 month
15 (88%)
15 (79%)
12 (60%)
Fatigue
12 (71%)
13 (68%)
12 (60%)
Cough
4 (24%)
3 (16%)
2 (10%)
Muscle pain
6 (35%)
3 (16%)
3 (15%)
Dyspnoea
10 (59%)
9 (47%)
4 (20%)
Anosmia/agueusia
2 (12%)
4 (21%)
2 (10%)
* Includes data for 19 outpatients, and 1 patient who was hospitalised and reported symptoms for 52 days.
Discussion
Between 18 March and 15 June 2020, the three clinicians on duty for the coronavirus hotline answered 430 calls, each corresponding to a telephone consultation. Most of the calls came from people with symptoms very suggestive of COVID-19 (fever, cough, muscle pain, dyspnoea). One third of callers were referred to a medical testing or examination facility and 40% of them tested positive by PCR. In 8% of cases, emergency hospitalisation was deemed necessary.
The proportion of positive diagnostic test results tended to increase with the age of those tested and with the duration of symptoms. No positive test was observed in people under 20 years of age. The proportion of positive test results was higher in subjects with fever, symptoms for ≥5 days, age ≥65 and a history of contact with a known case. The overall test positivity rate was also higher during the initial period of the outbreak, when testing was selectively reserved for those at risk or more severely affected, and tended to decrease as the indication for testing expanded to groups of people at lower risk (e.g., asymptomatic contacts of sick people).
Of all callers, healthcare workers, people at risk, known contacts, the elderly, people whose symptoms had lasted ≥5 days, and people with fever, were tested more often than others. Interestingly, healthcare workers were tested very often but rarely tested positive. This suggests that the risk of transmission of SARS-CoV2 to healthcare workers in our study population was not higher than for the general population, probably due to the adequate preventive measures implemented in healthcare settings. This contrasts with reports from London [1] and The Netherlands [2], where higher rates of infection were observed in healthcare workers than in the general population.
Apart from fever, individual clinical symptoms were only poorly indicative of test positivity. This is consistent with a recent study in which an attempt to define the most appropriate diagnostic attitude during telephone or video-assisted consultations for suspicion of COVID-19 emphasised the importance of assessing dyspnoea, but recognised the difficulty of an objective evaluation of most symptoms [3].
Individuals with no indication for the test had symptoms as frequently (at least with respect to cough and dyspnoea) as those tested. It is therefore possible that a number of these people did indeed suffer from COVID-19 in an initial stage, but that the diagnosis was not made (for reasons of logistics, availability of tests or according to FOPH instructions). It could be that some people were tested too early or too late to detect the virus in the nasopharyngeal smear, that the sampling technique was inadequate or that the test itself remained falsely negative. The actual number of cases with true COVID-19 in the local population is therefore most likely much higher than the number of positive tests, and a significant proportion of those who tested negative were actually infected with coronavirus and may have transmitted the disease to others.
The people who called the hotline suffered from symptoms that were sometimes severe and had a long evolution, even in those whose diagnostic test remained negative. The disease was characterised in our collective by a much slower clinical evolution than, for example, the evolution of the seasonal influenza, but was close to that of Epstein-Barr virus infection.
The long duration of COVID-19 symptoms has been observed in other studies. Most of the studies published to date related to hospitalised patients and analysed risk factors associated with adverse disease outcome (intubation, ventilation or death) [4–6]. Few studies have examined the outcome and duration of disease in patients who have not been in intensive care. A recent study of 1420 patients with a positive PCR test, of whom less than 10% had been hospitalised, reported an average duration of illness of 11.5 ± 5.7 days, but some symptoms such as fatigue, headache, anosmia and breathing difficulties lasted more than 30 days in a large proportion of patients [7]. A study of 179 patients hospitalised in Rome for documented SARS-CoV2 infection, 72% of whom had suffered from pneumonia, showed that 88% still had symptoms 60 days after the onset of illness, most often fatigue and dyspnoea [8]. In a group of 117 patients who returned home after hospitalisation in Fribourg (site of the current study), 63% still had symptoms 1 month after the onset of the disease [9], which is consistent with our observations. Studies carried out in China and New York have shown that the duration and severity of symptoms is greater in the elderly [10] and that clinical evolution can occur in two stages, with rapid worsening occurring approximately 1 week after the onset of symptoms of an initially benign disease [11, 12].
One of the difficulties of the study was that the onset of the coronavirus epidemic coincided with the end of the seasonal influenza epidemic and the peak of the epidemic occurred during the pollen season. It was therefore sometimes difficult for the physicians on duty to distinguish, on the basis of a telephone call, the nature and origin of the symptoms. In addition, assessing the severity of symptoms, such as dyspnoea, was not always easy, since most calls were made in a context of anxiety and in people whose mother tongue was often other than French.
The study has several weaknesses. The first is the fact that the criteria used to direct callers to a test facility varied over time (very restrictive at the beginning and then progressively broader), so that the test indications and the pre-test probability were not the same at the beginning and at the end of the hotline’s activity. There was no verification of the evolution in untested individuals, some of whom could have been carriers of SARS-Cov2, but did not meet the criteria for testing. Comparison of the clinical course of those who tested positive with those who tested negative reinforces this hypothesis. Finally, there has been no systematic monitoring of serological progression in positive individuals or in individuals with long-term symptoms. In cases where a check-up was carried out by the treating physicians the result is not known. Therefore, the true disease status of individuals with negative test results (or not tested) could not be ascertained.
For administrative reasons, the medical doctors in charge were not able to compare observations with the observations made in other Swiss cantons with a similar organisation, and could not compare the demographic characteristics of the study population with the local population of the canton. The study population may therefore not be representative of the local population. On the other hand, as most medical offices and hospitals redirected the calls to the hotline for further management, we assume that we received a largely representative sample of the population with symptoms suggestive of COVID-19.
The fact that many people with symptoms highly suggestive of COVID-19 have tested negatively has been noted in several studies [13, 14]. The reason may be that testing is sometimes done very early in the course of the disease, or the sample is not collected properly, or the tests do not have optimal sensitivity. In cases where there is a high pre-test probability of COVID-19, and when a decision for isolation has to be taken, repeat testing after a few days or serological testing for antibodies is justified and useful.
Conclusions
Demographic and clinical data analysis, as well as follow-up, of people who called the coronavirus hotline because of suspicious symptoms of COVID-19 shows that certain anamnestic criteria or risk factors such as age, duration of symptoms beyond 5 days and the notion of prior contact are statistically associated with the positivity of diagnostic tests by PCR, but that symptoms alone are not very discriminatory. The follow-up of those tested has shown that the course of COVID-19 may be long, that the majority of patients still have symptoms 1 month after the onset of the disease, and that many people who tested negatively at the onset of the disease most likely suffered from COVID-19 without being diagnosed as such.
Take-home messages
- Persons calling the hotline during the acute phase of the SARS-CoV2 outbreak complained of a variety of symptoms, which were not clearly associated with the test positivity.
- The only risk factors correlated with test positivity by logistic regression after adjustment for clinical symptoms were age ≥65 years, history of contact with a known case of COVID-19 and duration of symptoms ≥5 days.
- The proportion of test positivity among health care workers was not higher than in the global population
- The average duration of illness was 47 days in people who tested positive and were hospitalised, 38 days in people who tested positive but were not hospitalised and 23 days in people who tested negative. Globally, 75% of people still complained of health trouble 30 days after the beginning of disease.
- Due to the restrictive use of diagnostic tests during the acute phase of the outbreak, many people with symptoms were not tested but may have suffered from COVID-19 and have transmitted the disease.
Jean-Pierre Zellweger, Pulmonary physician, Villars-sur-Glâne, Switzerland, jean-pierre.zellweger[at]hin.ch
Catherine Plancherel, Internist, Villars-sur-Glâne, Switzerland
Frédéric Poulet, Internist, Fribourg, Switzerland
Raphaël M. Zellweger, Biologist, International Vaccine Institute, Seoul, South Korea
Erina Panchaud, Food Safety and Veterinary Office, Fribourg, Switzerland
Jeanette Muntwyler, Food Safety and Veterinary Office, Fribourg, Switzerland
Grégoire Seitert, Food Safety and Veterinary Office, Fribourg, Switzerland
References
- Houlihan CF, Vora N, Byrne T, Lewer D, Kelly G, Heaney J, et al.; Crick COVID-19 Consortium; SAFER Investigators. Pandemic peak SARS-CoV-2 infection and seroconversion rates in London frontline health-care workers. Lancet. 2020;396(10246):e6–7. doi:https://doi.org/10.1016/S0140-6736(20)31484-7.
- Kluytmans-van den Bergh MFQ, Buiting AGM, Pas SD, Bentvelsen RG, van den Bijllaardt W, van Oudheusden AJG, et al. Prevalence and Clinical Presentation of Health Care Workers With Symptoms of Coronavirus Disease 2019 in 2 Dutch Hospitals During an Early Phase of the Pandemic. JAMA Netw Open. 2020;3(5):e209673. doi:https://doi.org/10.1001/jamanetworkopen.2020.9673.
- Greenhalgh T, Koh GCH, Car J. Covid-19: a remote assessment in primary care. BMJ. 2020;368:m1182. doi:https://doi.org/10.1136/bmj.m1182.
- Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–9. doi:https://doi.org/10.1001/jama.2020.1585.
- Goyal P, Choi JJ, Pinheiro LC, Schenck EJ, Chen R, Jabri A, et al. Clinical Characteristics of Covid-19 in New York City. N Engl J Med. 2020;382(24):2372–4. doi:https://doi.org/10.1056/NEJMc2010419.
- Argenziano MG, Bruce SL, Slater CL, Tiao JR, Baldwin MR, Barr RG, et al. Characterization and clinical course of 1000 patients with coronavirus disease 2019 in New York: retrospective case series. BMJ. 2020;369:m1996. doi:https://doi.org/10.1136/bmj.m1996.
- Lechien JR, Chiesa-Estomba CM, Place S, Van Laethem Y, Cabaraux P, Mat Q, et al.; COVID-19 Task Force of YO-IFOS. Clinical and epidemiological characteristics of 1420 European patients with mild-to-moderate coronavirus disease 2019. J Intern Med. 2020;288(3):335–44. doi:https://doi.org/10.1111/joim.13089.
- Carfì A, Bernabei R, Landi F; Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent Symptoms in Patients After Acute COVID-19. JAMA. 2020;324(6):603–5. doi:https://doi.org/10.1001/jama.2020.12603.
- Pellaud C, Grandmaison G, Pham Huu Thien HP, Baumberger M, Carrel G, Ksouri H, et al. Characteristics, comorbidities, 30-day outcome and in-hospital mortality of patients hospitalised with COVID-19 in a Swiss area - a retrospective cohort study. Swiss Med Wkly. 2020;150:w20314. doi:https://doi.org/10.4414/smw.2020.20314.
- Liu K, Chen Y, Lin R, Han K. Clinical features of COVID-19 in elderly patients: A comparison with young and middle-aged patients. J Infect. 2020;80(6):e14–8. doi:https://doi.org/10.1016/j.jinf.2020.03.005. PubMed</jrn>
- Gandhi RT, Lynch JB, Del Rio C. Mild or Moderate Covid-19. N Engl J Med. 2020;383(18):1757–66. doi:https://doi.org/10.1056/NEJMcp2009249.
- Lescure FX, Bouadma L, Nguyen D, Parisey M, Wicky PH, Behillil S, et al. Clinical and virological data of the first cases of COVID-19 in Europe: a case series. Lancet Infect Dis. 2020;20(6):697–706. doi:https://doi.org/10.1016/S1473-3099(20)30200-0.
- Chenal R, Ho LKS, Kokkinakis I, Selby K, Greub G, d’Acremont V, et al. Détection du SARS-CoV2 par RT-PCR sur frottis naso-pharyngé. Swiss Med Forum. 2020;20:415–9.
- Woloshin S, Patel N, Kesselheim AS. False Negative Tests for SARS-CoV-2 Infection - Challenges and Implications. N Engl J Med. 2020;383(6):e38. doi:https://doi.org/10.1056/NEJMp2015897.
